Scalable Synthesis of N-Alkyl-3-Piperidones for Pharmaceutical Intermediates
Scalable Synthesis of N-Alkyl-3-Piperidones for Pharmaceutical Intermediates
The pharmaceutical industry continuously demands robust, scalable, and cost-effective pathways for constructing nitrogen-containing heterocycles, which serve as the backbone for countless active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN101638378A, which discloses a highly efficient synthetic method for N-alkyl substituted-3-piperidones. This technology addresses the critical bottlenecks of traditional synthesis by utilizing a strategic ring-opening approach starting from readily available cyclopropyl methyl ketone. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this methodology offers a compelling value proposition through its operational simplicity and high atom economy. The process circumvents the complexities of earlier routes, delivering high-purity products suitable for long-term storage and downstream medicinal chemistry applications without the burden of extensive chromatographic purification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-alkyl-3-piperidones has relied heavily on classical strategies such as Dieckmann condensation or the modification of pyridine derivatives, both of which present substantial hurdles for industrial scale-up. As illustrated in prior art strategies, such as the conversion of pyridine derivatives shown below, these conventional pathways often involve expensive starting materials and harsh reaction conditions that compromise overall yield.
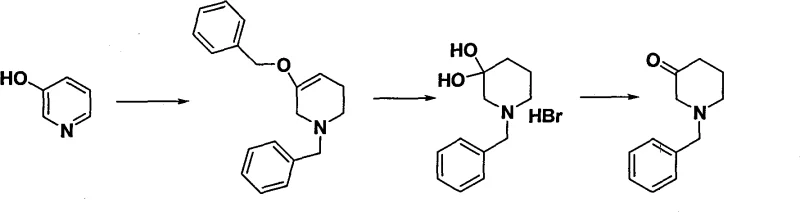
Furthermore, literature reports indicate that while some methods possess versatility regarding substituent groups, the yields for the critical azanylization and cyclization steps are generally low, creating a bottleneck in mass production. The reliance on column chromatography for purification in many laboratory-scale protocols further exacerbates cost issues, making these routes economically unviable for generating multi-kilogram quantities required for clinical trials. The inability to easily remove impurities without sophisticated separation techniques limits the applicability of these older methods in a GMP-compliant manufacturing environment, necessitating a shift towards more pragmatic synthetic designs.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent leverages a concise four-step sequence that transforms cheap cyclopropyl methyl ketone into the target piperidone scaffold with remarkable efficiency. This strategy fundamentally alters the economic landscape of cost reduction in pharmaceutical intermediate manufacturing by replacing costly precursors with bulk chemicals that are universally accessible in the fine chemical market. The route is characterized by moderate reaction conditions and a streamlined workup procedure that eschews column chromatography entirely, relying instead on recrystallization and vacuum distillation. This simplification not only accelerates the production timeline but also drastically reduces the environmental footprint associated with solvent waste, aligning perfectly with modern green chemistry initiatives and supply chain sustainability goals.
Mechanistic Insights into Ring-Opening and Cyclization Strategy
The core innovation of this synthesis lies in the clever manipulation of ring strain and protecting group chemistry to drive the formation of the six-membered piperidine ring. The process initiates with the ring-opening of bromoethyl cyclopropyl ketone using a hydrogen halide, typically hydrogen bromide, which effectively linearizes the three-membered ring into a 1,5-dibromo-2-pentanone intermediate. This step is crucial as it installs the necessary leaving groups at the terminal positions while maintaining the ketone functionality, setting the stage for subsequent intramolecular cyclization. The high yield of this opening step, reported at 97% in embodiments, underscores the thermodynamic favorability of relieving the cyclopropane ring strain under acidic conditions.
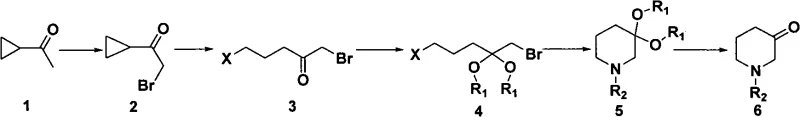
Following the ring opening, the ketone moiety is protected as a ketal using ethylene glycol and an acid catalyst like p-toluenesulfonic acid. This protection is mechanistically vital to prevent unwanted side reactions, such as self-condensation or polymerization, during the subsequent nucleophilic substitution with the primary amine. The cyclization step involves the displacement of the bromide atoms by the amine nitrogen, closing the ring to form the piperidine core. Finally, acidic hydrolysis removes the ketal protecting group to reveal the ketone, yielding the final N-alkyl-3-piperidone. This logical progression ensures high regioselectivity and minimizes the formation of structural isomers, resulting in a product with exceptional purity profiles.
How to Synthesize N-Alkyl-3-Piperidones Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the reported 80% total yield. The protocol is designed to be robust, tolerating slight variations in temperature and stoichiometry while maintaining high conversion rates. For process chemists looking to implement this route, the elimination of chromatographic purification is a key operational advantage, allowing for direct isolation of intermediates via crystallization or distillation. The detailed standardized synthetic steps for implementing this high-efficiency route are provided in the guide below.
- Ring Opening: React bromoethyl cyclopropyl ketone with hydrogen halide (preferably HBr) at 50°C to obtain 1,5-dibromo-2-pentanone.
- Protection: Protect the keto carbonyl group using ethylene glycol and p-TsOH in benzene under reflux to form the ketal derivative.
- Cyclization: React the protected dibromide with a primary amine (e.g., benzylamine) at 80°C to form the piperidine ring structure.
- Deprotection: Hydrolyze the ketal protecting group using 1M HCl under reflux to yield the final N-alkyl-3-piperidone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers transformative benefits regarding cost stability and material availability. By anchoring the synthesis on cyclopropyl methyl ketone, a commodity chemical, manufacturers can decouple their production costs from the volatility associated with specialized, low-volume starting materials. This shift ensures a more predictable pricing structure for the final piperidone intermediates, facilitating better budget forecasting for long-term drug development projects. Furthermore, the simplified purification train reduces the dependency on high-purity silica gel and large volumes of elution solvents, directly lowering the operational expenditure (OpEx) per kilogram of product produced.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive raw materials and the avoidance of expensive transition metal catalysts or exotic reagents. By eliminating the need for column chromatography, the process significantly reduces solvent consumption and labor hours associated with purification, leading to substantial cost savings in the final product price. The high yield of each individual step cumulatively results in a superior overall mass balance, minimizing raw material waste and maximizing the output from every batch processed.
- Enhanced Supply Chain Reliability: Sourcing risks are mitigated because the starting materials are widely produced by multiple global suppliers, preventing single-source bottlenecks. The robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-optimization, ensuring continuity of supply even if one facility faces disruptions. This flexibility is critical for maintaining the uninterrupted flow of intermediates required for just-in-time API manufacturing schedules.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial constraints in mind, such as heat transfer and mixing efficiency in large reactors. The reduction in hazardous waste generation, particularly through the avoidance of silica waste from chromatography, simplifies regulatory compliance and waste disposal logistics. This alignment with environmental standards makes the route attractive for companies aiming to reduce their carbon footprint and adhere to strict ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production pipelines.
Q: What are the advantages of this synthesis over traditional Dieckmann condensation?
A: Traditional Dieckmann condensation methods often suffer from lower yields in the azanylization and cyclization steps, limiting their industrial application. This novel route utilizes a ring-opening strategy from cheap cyclopropyl methyl ketone, achieving a total yield of approximately 80% with simpler purification.
Q: Is this process suitable for large-scale manufacturing without chromatography?
A: Yes, the process is specifically designed for industrial scalability. It eliminates the need for column chromatography, relying instead on recrystallization and vacuum distillation, which significantly reduces solvent consumption and processing time for bulk production.
Q: What represents the key cost-saving factor in this synthetic route?
A: The primary cost driver is the use of commercially available and inexpensive cyclopropyl methyl ketone as the starting material, replacing expensive specialized precursors used in other strategies. Additionally, the high yield of each step minimizes raw material waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkyl-3-Piperidone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug discovery and development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in heterocyclic chemistry allows us to optimize this specific ring-opening route further, tailoring it to meet the unique volume and quality requirements of our global partners.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
