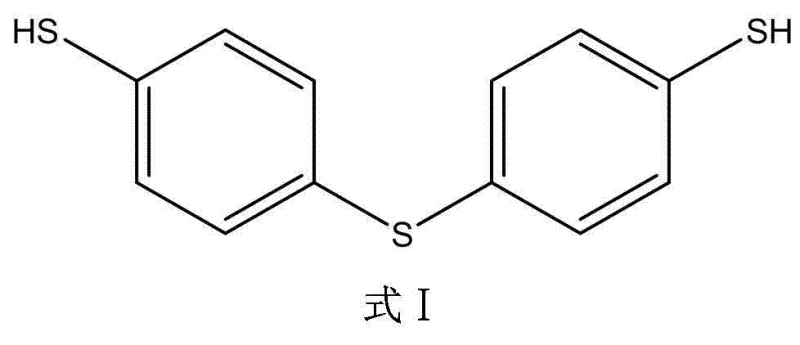
Scalable Synthesis of 4,4'-Thiobisbenzenethiol for High-Performance Pharmaceutical Intermediates
The global demand for high-purity sulfur-containing intermediates continues to surge, driven by the expanding pharmaceutical and agrochemical sectors that require robust building blocks for complex molecule assembly. Patent CN102093269B, published in early 2013, introduces a transformative synthesis method for 4,4'-thiobisbenzenethiol, a critical compound widely utilized in the manufacturing of fine chemicals, medicines, and dyes. This specific technical disclosure addresses long-standing inefficiencies in traditional production routes by optimizing reaction conditions and reagent selection to maximize yield while minimizing impurity profiles. For R&D Directors and Procurement Managers evaluating supply chain resilience, understanding the mechanistic advantages of this patent is essential for securing a reliable 4,4'-thiobisbenzenethiol supplier. The method described leverages a strategic esterification protection strategy followed by a controlled sulfur-bridge formation, ensuring that the final product meets the stringent purity specifications required for downstream API synthesis. By adopting this advanced methodology, manufacturers can significantly enhance the stability and reproducibility of their production lines, thereby reducing the risk of batch failures and ensuring consistent quality for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4,4'-dimercapto diphenyl sulfide has relied on cumbersome and environmentally taxing processes that pose significant challenges for modern supply chain heads. Prior art, such as the method reported in CN02114837.6, utilizes diphenyl sulfide and chlorosulfonic acid as starting materials, followed by a reduction step involving substantial quantities of zinc powder. This traditional approach suffers from severe drawbacks, including the high consumption of expensive metallic zinc and the generation of complex isomer mixtures that are difficult to separate, resulting in suboptimal product content. Furthermore, alternative routes documented in Japanese patents JP06032773 and JP04264064 employ triphenylphosphine for reduction, which introduces another layer of cost inefficiency due to the high price of phosphine reagents and the difficulty in removing phosphorus-containing byproducts. These legacy methods not only inflate the cost reduction in pharmaceutical intermediates manufacturing but also create substantial waste disposal burdens, making them increasingly unsustainable in the face of tightening environmental regulations and the need for greener chemical processes.
The Novel Approach
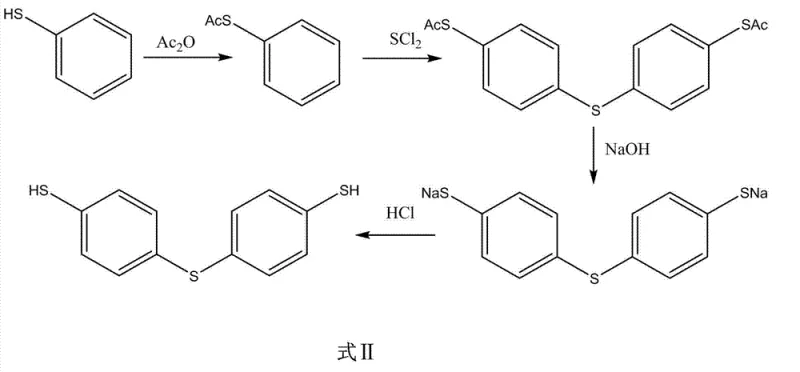
In stark contrast to these outdated techniques, the novel approach detailed in the patent utilizes a streamlined four-step sequence that begins with the esterification of thiophenol using acetic anhydride, effectively protecting the thiol group before the critical sulfur-bridge formation. This strategic protection allows for a highly controlled reaction with sulfur dichloride in the presence of an iodine catalyst at mild temperatures ranging from 10 to 15°C, which drastically reduces the formation of unwanted isomers and side products. The subsequent hydrolysis step employs sodium hydroxide followed by acidification with hydrochloric acid to liberate the free thiol groups, yielding a white solid that is easily purified through recrystallization. This methodology not only simplifies the operational complexity but also ensures a total molar yield exceeding 60%, demonstrating a clear pathway for the commercial scale-up of complex polymer additives and fine chemical intermediates. By eliminating the need for heavy metal reductants like zinc or expensive phosphines, this process offers a cleaner, more cost-effective alternative that aligns perfectly with the goals of a reliable agrochemical intermediate supplier seeking to optimize production economics.
Mechanistic Insights into Iodine-Catalyzed Sulfur Bridge Formation
The core innovation of this synthesis lies in the precise mechanistic control exerted during the etherification step, where iodine acts as a crucial catalyst to facilitate the coupling of the acetic acid thiophenol ester with sulfur dichloride. This catalytic system promotes the formation of the sulfur-sulfur-carbon bridge with high regioselectivity, ensuring that the substitution occurs primarily at the para-position of the aromatic ring, which is vital for maintaining the structural integrity required for downstream applications. The reaction conditions are meticulously optimized, with the temperature maintained strictly between 10 and 15°C to prevent thermal degradation and minimize the generation of polysulfide impurities that often plague high-temperature sulfurization reactions. For R&D teams focused on impurity谱 analysis, this low-temperature protocol is significant because it inherently limits the energy available for side reactions, thereby producing a cleaner crude product that requires less intensive purification. The use of acetic anhydride as a protecting group is also mechanistically sound, as it prevents the oxidation of the thiol group during the harsh sulfurization conditions, ensuring that the final hydrolysis step yields the desired dithiol with high fidelity.
Furthermore, the hydrolysis mechanism employed in the third step is designed to maximize recovery while ensuring the complete removal of acetyl protecting groups without damaging the sensitive sulfur bridge. By utilizing sodium hydroxide in a warming reflux reaction, the ester bonds are cleaved efficiently to form the sodium salt of the thiol, which is then converted to the free thiol upon the dropwise addition of hydrochloric acid solution. This acidification step is critical for precipitating the product as a white solid, allowing for easy filtration and separation from the aqueous phase containing inorganic salts. The careful control of the molar ratio between hydrochloric acid and sodium hydroxide ensures that the pH is adjusted precisely to the isoelectric point of the product, maximizing precipitation efficiency and minimizing product loss in the mother liquor. This level of mechanistic detail underscores the robustness of the process, providing a solid foundation for the production of high-purity OLED material precursors or pharmaceutical building blocks where trace impurities can be detrimental to final product performance.
How to Synthesize 4,4'-Thiobisbenzenethiol Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to achieve the reported yields and purity levels, making it an ideal candidate for technology transfer to commercial manufacturing facilities. The process begins with the esterification of thiophenol, where acetic anhydride is added dropwise to control the exotherm, followed by heating to 80-90°C to ensure complete conversion as monitored by GC analysis. The subsequent etherification step demands precise temperature control at 10-15°C during the addition of sulfur dichloride to maintain the selectivity of the iodine-catalyzed reaction, followed by a 2 to 4-hour insulation period to allow the reaction to reach completion. Finally, the hydrolysis and recrystallization steps utilize standard unit operations that are easily scalable, with the choice of recrystallization solvents such as benzene, toluene, or hexane allowing for flexibility based on availability and cost considerations. Detailed standardized synthesis steps see the guide below.
- Esterify thiophenol with acetic anhydride at 80-90°C to form the acetic acid thiophenol ester intermediate.
- React the ester with sulfur dichloride using iodine as a catalyst at 10-15°C to form the sulfur bridge.
- Hydrolyze the intermediate with sodium hydroxide followed by acidification with hydrochloric acid to obtain the crude solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patent-protected synthesis method offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line through optimized resource utilization and reduced waste management costs. The elimination of expensive and hazardous reagents like zinc powder and triphenylphosphine translates into significant cost savings in raw material procurement, while the simplified workup procedure reduces the labor and energy inputs required for purification. Moreover, the high selectivity of the reaction minimizes the formation of difficult-to-remove isomers, which reduces the need for extensive chromatographic purification and allows for more efficient use of production capacity. This efficiency gain is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling suppliers to respond more rapidly to market demands and secure long-term contracts with multinational corporations. The robustness of the process also enhances supply chain reliability, as the reagents involved are commodity chemicals with stable supply lines, mitigating the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The strategic replacement of high-cost reductants with a catalytic iodine system and acetic anhydride significantly lowers the variable cost per kilogram of the final product, allowing for more competitive pricing in the global market. By avoiding the use of stoichiometric amounts of zinc or phosphine, the process reduces the mass of waste generated per unit of product, which in turn lowers the environmental compliance costs associated with waste treatment and disposal. This economic efficiency is further amplified by the high yield of the process, which maximizes the output from a fixed amount of starting material, thereby improving the overall asset utilization of the manufacturing plant. Consequently, partners can expect substantial cost savings without compromising on the quality or purity of the delivered intermediates, making this route highly attractive for cost-sensitive applications in the agrochemical and dye industries.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as thiophenol, acetic anhydride, and sulfur dichloride ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with specialized reagents. This availability allows for better inventory planning and reduces the need for safety stock, freeing up working capital and improving the overall liquidity of the supply chain operation. Additionally, the mild reaction conditions reduce the stress on equipment, leading to lower maintenance requirements and longer campaign runs, which contributes to a more consistent and reliable delivery schedule for customers. For supply chain heads, this reliability is paramount, as it ensures the continuity of production for downstream APIs and fine chemicals, preventing costly delays and ensuring that market commitments are met consistently.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with unit operations such as esterification, filtration, and recrystallization being standard in the fine chemical industry and easily adaptable to large-scale reactors. The reduction in hazardous waste, particularly the absence of heavy metal sludge from zinc reduction, simplifies the environmental permitting process and aligns with the increasing global demand for green chemistry practices. This environmental advantage not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturer, making it a preferred partner for multinational companies with strict sustainability mandates. The ability to scale from pilot plant to commercial production with minimal process modification ensures that the technology can be deployed rapidly to meet growing market demand, securing a competitive edge in the fast-paced chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of 4,4'-thiobisbenzenethiol, providing clarity for stakeholders evaluating this technology for their production needs. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industrial applications. Understanding these details is crucial for making informed decisions about process adoption and supplier selection in the competitive landscape of fine chemical intermediates.
Q: What are the primary advantages of this synthesis method over traditional zinc reduction?
A: This method eliminates the need for expensive zinc powder and reduces the formation of isomers, leading to higher purity and simplified waste treatment compared to conventional chlorosulfonation and reduction routes.
Q: What is the expected yield for the commercial production of 4,4'-thiobisbenzenethiol?
A: According to patent data, the total molar yield for this specific pathway exceeds 60%, which is considered robust for multi-step sulfur-containing intermediate synthesis.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes mild reaction conditions (10-15°C for etherification) and readily available reagents like acetic anhydride and sulfur dichloride, making it highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Thiobisbenzenethiol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products, and we are committed to delivering 4,4'-thiobisbenzenethiol that meets the highest standards of purity and consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage development through to full-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to verify every batch, guaranteeing that the material you receive is free from the isomers and impurities that can compromise your downstream synthesis. Our dedication to technical excellence and customer satisfaction makes us the ideal partner for companies seeking a reliable 4,4'-thiobisbenzenethiol supplier who can deliver both quality and quantity.
We invite you to collaborate with us to optimize your supply chain and achieve your production goals through our advanced manufacturing capabilities and technical expertise. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and to obtain specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a wealth of chemical knowledge and production capacity that can drive your business forward, ensuring that you have a secure and efficient source of this vital intermediate for your pharmaceutical or agrochemical applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
