Advanced Grignard-Based Synthesis of Resveratrol for Commercial Scale-Up
Introduction to Patent CN100509729C
The pharmaceutical and nutraceutical industries constantly seek robust synthetic routes for high-value antioxidants like resveratrol, a compound renowned for its cardioprotective and anticancer properties. Patent CN100509729C introduces a transformative methodology that addresses the longstanding inefficiencies of traditional synthesis pathways. This innovation leverages a strategic Grignard reaction sequence followed by acid-catalyzed dehydration and demethylation to achieve superior yields and purity profiles. For R&D directors and procurement specialists, this patent represents a pivotal shift away from transition-metal dependency towards a more economically viable and operationally simple framework. By optimizing the construction of the stilbene backbone through nucleophilic addition rather than olefination or cross-coupling, the technology ensures a consistent supply of high-purity intermediates essential for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of resveratrol has been plagued by significant technical bottlenecks inherent to older methodologies such as the Perkin, Wittig, and Heck reactions. The Perkin reaction, for instance, often suffers from poor stereoselectivity, yielding a mixture of cis and trans isomers that necessitates energy-intensive separation processes to isolate the bioactive trans-form. Similarly, while the Wittig reaction offers mild conditions, it generates stoichiometric amounts of phosphine oxide waste, creating substantial environmental disposal costs and complicating the purification of the final product. 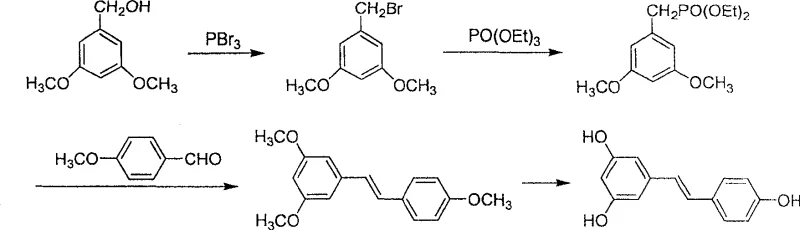
Furthermore, the Heck coupling strategy, although effective for forming carbon-carbon double bonds, relies heavily on expensive palladium catalysts which pose severe contamination risks in pharmaceutical grades. The requirement for strictly anhydrous and oxygen-free environments in Heck chemistry further escalates operational expenditures and limits the feasibility of large-scale batch processing. These cumulative drawbacks result in higher production costs, extended lead times, and inconsistent quality, presenting a major hurdle for supply chain managers aiming to secure reliable resveratrol suppliers.
The Novel Approach
In stark contrast, the methodology disclosed in CN100509729C circumvents these issues by employing a Grignard-based strategy that is both chemically elegant and commercially pragmatic. This novel approach constructs the central carbon skeleton through the nucleophilic addition of a 3,5-dimethoxybenzyl magnesium bromide species to anisaldehyde, effectively bypassing the need for precious metal catalysts or phosphorous reagents. The subsequent dehydration step utilizes inexpensive p-toluenesulfonic acid to drive the formation of the stilbene double bond with high thermodynamic preference for the trans-configuration. This route not only simplifies the reaction workflow but also drastically reduces the impurity profile associated with metal residues, thereby lowering the burden on downstream purification units and enhancing overall process efficiency.
Mechanistic Insights into Grignard-Mediated Stilbene Formation
The core of this synthetic breakthrough lies in the precise generation and utilization of the Grignard reagent, which serves as the nucleophilic engine for carbon-carbon bond formation. As illustrated in the reaction scheme, 3,5-dimethoxybenzyl bromide reacts with activated magnesium metal in anhydrous tetrahydrofuran or diethyl ether to form the corresponding organomagnesium species. 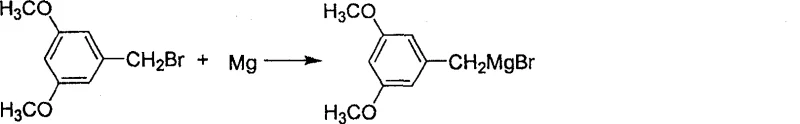
This step is critical, as the presence of electron-donating methoxy groups on the aromatic ring enhances the stability of the intermediate while maintaining sufficient nucleophilicity for the subsequent attack. The patent specifies that ultrasonic irradiation can be employed to facilitate the initiation of the Grignard formation, ensuring complete consumption of the magnesium and minimizing the formation of Wurtz-coupling byproducts. This attention to initiation mechanics ensures a high concentration of the active reagent, which is paramount for driving the subsequent addition reaction to completion with minimal side reactions.
Following the formation of the Grignard reagent, the process proceeds to a controlled nucleophilic addition with anisaldehyde. The reaction is conducted at low temperatures (10-15°C) to manage the exotherm and prevent competing side reactions, followed by stirring at room temperature to ensure full conversion to the secondary alcohol intermediate, 3,5,4'-trimethoxydiphenyl ethanol. The subsequent dehydration step is equally mechanistically significant; by employing a catalytic amount of p-toluenesulfonic acid in a refluxing aromatic solvent like toluene, the reaction leverages azeotropic distillation to remove water. This drives the equilibrium towards the elimination product, 3,5,4'-trimethoxystilbene, with high stereoselectivity for the trans-isomer due to steric minimization in the transition state. 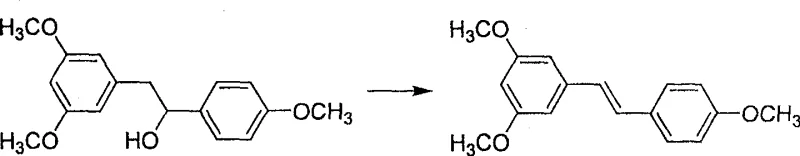
Finally, the demethylation step utilizes boron tribromide to cleave the methyl ethers, revealing the phenolic hydroxyl groups characteristic of resveratrol. This Lewis acid-mediated cleavage is highly specific and proceeds under mild conditions, preserving the integrity of the sensitive stilbene double bond while ensuring quantitative conversion to the final target molecule. The combination of these mechanistic steps results in a process that is not only chemically robust but also inherently safer and more scalable than transition-metal catalyzed alternatives.
How to Synthesize Resveratrol Efficiently
The synthesis of resveratrol via this patented Grignard route involves a logical sequence of four distinct chemical transformations that can be seamlessly integrated into existing multipurpose reactor setups. The process begins with the activation of magnesium and formation of the organometallic species, followed by the carbonyl addition, acid-catalyzed dehydration, and final deprotection. Each step has been optimized in the patent examples to maximize yield and minimize waste, providing a clear roadmap for process chemists aiming to implement this technology. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- React 3,5-dimethoxybenzyl bromide with magnesium metal in anhydrous THF or ether to form the Grignard reagent.
- Perform nucleophilic addition of the Grignard reagent to anisaldehyde to generate 3,5,4'-trimethoxydiphenyl ethanol.
- Dehydrate the resulting alcohol using p-toluenesulfonic acid in toluene to form 3,5,4'-trimethoxystilbene.
- Demethylate the trimethoxystilbene using boron tribromide in dichloromethane to obtain the final resveratrol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Grignard-based synthesis offers profound advantages for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediate manufacturing. The elimination of palladium catalysts, which are subject to volatile market pricing and stringent regulatory limits on residual metals in APIs, translates directly into significant raw material savings and simplified quality control protocols. Furthermore, the avoidance of phosphine reagents removes the logistical and environmental costs associated with the disposal of phosphine oxide waste, a common burden in Wittig-based processes. This streamlined chemical profile allows for a more predictable cost structure and reduces the risk of supply chain disruptions caused by the scarcity of specialized catalysts.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with commodity chemicals like magnesium and p-toluenesulfonic acid fundamentally alters the cost basis of production. By removing the need for costly metal scavenging resins or complex chromatography steps required to meet heavy metal specifications, the overall cost of goods sold is substantially decreased. Additionally, the use of common solvents such as THF, toluene, and dichloromethane ensures that solvent recovery and recycling can be managed efficiently within standard distillation infrastructure, further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically 3,5-dimethoxybenzyl bromide and anisaldehyde, are widely available bulk chemicals with stable supply chains, unlike specialized vinyl halides or phosphonium salts. This accessibility ensures that production schedules are not held hostage by the lead times of niche reagents. The robustness of the reaction conditions, which tolerate standard industrial handling better than air-sensitive palladium cycles, also means that batch failure rates are minimized, guaranteeing a consistent and reliable flow of high-purity resveratrol to downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as reflux, extraction, and crystallization that are standard in multi-tonne manufacturing facilities. The absence of heavy metal contaminants simplifies the environmental compliance landscape, reducing the load on wastewater treatment systems and lowering the cost of regulatory auditing. This green chemistry profile aligns perfectly with the increasing demand for sustainable manufacturing practices, making the facility more attractive to eco-conscious partners and reducing the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational requirements. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the advantages of this Grignard route over the Heck reaction?
A: Unlike the Heck reaction which requires expensive palladium catalysts and strict anhydrous/oxygen-free conditions leading to potential metal contamination, this Grignard route utilizes readily available magnesium and avoids heavy metal residues, significantly simplifying purification and reducing raw material costs.
Q: How is stereochemistry controlled in this synthesis?
A: The dehydration step (Step 3) utilizing p-toluenesulfonic acid in refluxing toluene favors the thermodynamic stability of the trans-isomer, ensuring high stereoselectivity for the biologically active trans-resveratrol without the need for complex isomerization steps found in Perkin reactions.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process employs common solvents like THF, toluene, and dichloromethane and avoids exotic catalysts. The workup procedures involve standard extractions and recrystallizations, making it highly scalable for industrial production compared to methods requiring chromatography or specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Resveratrol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic routes like the one described in CN100509729C for securing a competitive edge in the global nutraceutical and pharmaceutical markets. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Grignard methodology are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of resveratrol meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can reduce your lead time for high-purity resveratrol and optimize your overall procurement strategy.
