Optimizing Alpha-Arylalkanoic Acid Production via Novel Thiocarbamate Rearrangement for Commercial Scale
Optimizing Alpha-Arylalkanoic Acid Production via Novel Thiocarbamate Rearrangement for Commercial Scale
The pharmaceutical and fine chemical industries continuously seek robust synthetic pathways for alpha-arylalkanoic acids, which serve as critical scaffolds for non-steroidal anti-inflammatory drugs (NSAIDs) and various bioactive agents. Patent CN1217913C introduces a transformative methodology for preparing meta or para-substituted alpha-arylalkanoic acids of formula (I), addressing long-standing challenges in efficiency and cost. This patent details a sophisticated sequence involving the conversion of alpha-hydroxy derivatives into thiocarbamate intermediates, followed by a thermal rearrangement and subsequent catalytic hydrogenation. The significance of this intellectual property lies in its ability to bypass expensive noble metal catalysts and complex purification protocols that have historically burdened the supply chain. By leveraging a Newman-Karnes type rearrangement, the process achieves high chemoselectivity, preserving sensitive functional groups such as ketones while effectively removing the phenolic hydroxyl moiety. For global procurement and R&D leaders, understanding this technology is essential for evaluating next-generation suppliers capable of delivering high-purity pharmaceutical intermediates with enhanced economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the removal of phenolic hydroxyl groups from arylalkanoic acid derivatives has relied on derivatization strategies followed by reduction, often involving significant operational drawbacks. Traditional approaches frequently utilize expensive reagents or lack the necessary selectivity to preserve other functional groups on the molecule, leading to complex impurity profiles that are difficult to manage during scale-up. For instance, methods disclosed in earlier literature often require palladium on carbon (Pd/C) catalysts under hydrogenation conditions, which not only inflate raw material costs but also introduce risks of heavy metal contamination that require stringent removal steps. Other strategies involving perfluoroalkyl sulfonic acid esters or complex acid chloride mixtures add layers of procedural complexity and safety concerns, particularly when handling hazardous reagents on a multi-ton scale. These conventional pathways often necessitate the isolation and purification of unstable intermediates, which drastically reduces overall yield and extends manufacturing lead times. Consequently, the cumulative effect of these inefficiencies results in higher production costs and reduced supply chain reliability, making it challenging for manufacturers to remain competitive in the global market for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the process outlined in CN1217913C offers a streamlined and economically superior alternative by utilizing a thiocarbamate-mediated deoxygenation strategy. This novel approach begins with the conversion of the alpha-hydroxy derivative into an O-dialkyl aryl thiocarbamate, a transformation that utilizes readily available and cost-effective reagents such as thiophosgene and simple amines. The core innovation lies in the subsequent thermal rearrangement, which converts the O-thiocarbamate into an S-dialkyl aryl thiocarbamate without the need for additional reagents, thereby simplifying the reaction matrix. Following this rearrangement, the sulfur moiety is efficiently removed via catalytic hydrogenation using Raney nickel, a catalyst that is significantly more affordable and easier to handle than palladium-based systems. Crucially, this method allows for the direct progression of intermediates without rigorous purification between steps, as the reaction conditions are sufficiently selective to minimize byproduct formation. This telescoping of steps not only accelerates the production timeline but also reduces solvent consumption and waste generation, aligning with modern green chemistry principles while delivering substantial cost savings in the manufacturing of complex alpha-arylalkanoic acids.
Mechanistic Insights into Thiocarbamate Rearrangement and Desulfurization
The chemical elegance of this process is rooted in the precise control of the Newman-Karnes rearrangement, which facilitates the migration of the thiocarbamoyl group from the oxygen to the sulfur atom on the aromatic ring. This thermal rearrangement step is critical as it sets the stage for the final reductive cleavage, ensuring that the carbon-oxygen bond is effectively replaced by a carbon-hydrogen bond without disturbing the stereochemistry or integrity of the alpha-alkyl carboxylic acid side chain. The mechanism proceeds through a concerted cyclic transition state during the heating phase, which explains the high regioselectivity observed in the formation of the S-thiocarbamate intermediate. By carefully controlling the temperature and reaction time, the process avoids the decomposition of sensitive ester or ketone functionalities that might be present on the aryl ring, a common failure point in less sophisticated reduction methods. This mechanistic robustness ensures that the final product retains the necessary structural features required for downstream pharmaceutical synthesis, thereby reducing the risk of batch rejection due to structural impurities.
Furthermore, the impurity control mechanism is inherently built into the choice of the Raney nickel catalytic hydrogenation step for desulfurization. Unlike harsh chemical reduction methods that might attack carbonyl groups or double bonds, the Raney nickel system under the specified conditions exhibits remarkable chemoselectivity towards the carbon-sulfur bond. This selectivity is paramount for maintaining the purity of the final alpha-arylalkanoic acid, as it prevents the formation of over-reduced byproducts or side-reactions that could compromise the quality of the active pharmaceutical ingredient (API) precursor. The process also mitigates the risk of racemization at the chiral center, which is a critical quality attribute for many NSAID intermediates. By avoiding strong acidic or basic conditions during the cleavage step, the integrity of the chiral alpha-carbon is preserved, ensuring that the optical purity remains within the stringent specifications demanded by regulatory bodies. This level of control over the reaction pathway underscores the technical superiority of this patent and highlights its value for manufacturers seeking to produce high-purity pharmaceutical intermediates with consistent quality.
How to Synthesize Alpha-Arylalkanoic Acids Efficiently
The implementation of this synthesis route requires a disciplined approach to reaction engineering, focusing on the seamless transition between the thiocarbamate formation, rearrangement, and hydrogenation stages. The patent provides a clear framework for executing these transformations, emphasizing the importance of reagent stoichiometry and thermal management to maximize yield and minimize waste. Operators must ensure that the initial conversion to the thiocarbamate is complete before initiating the thermal rearrangement, as residual starting material can lead to complex impurity profiles that are difficult to separate later. The subsequent hydrogenation step demands careful monitoring of hydrogen uptake and catalyst activity to ensure complete desulfurization without over-processing the batch. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety protocols essential for scaling this chemistry.
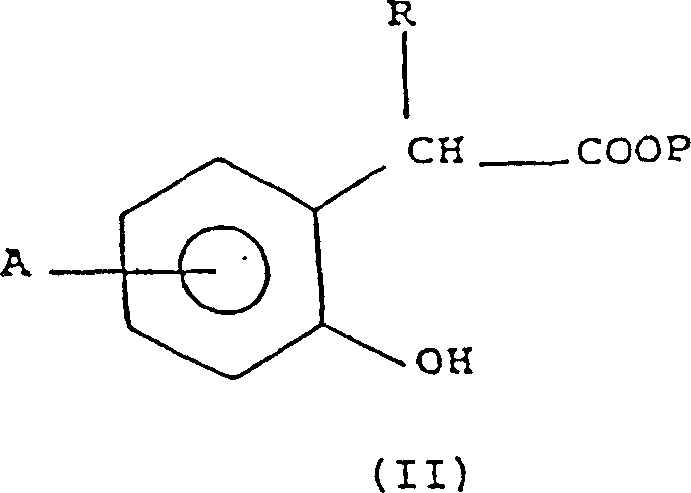
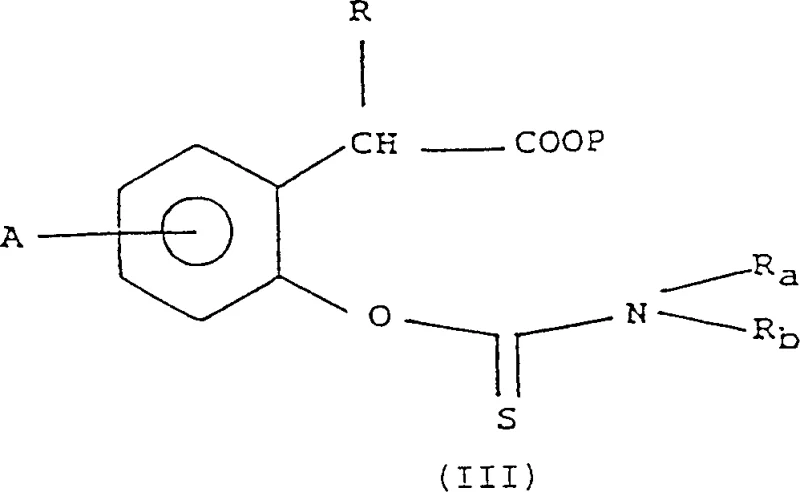
- Convert the alpha-hydroxy arylalkanoic acid derivative (Formula II) into an O-dialkyl aryl thiocarbamate (Formula III) using thiophosgene and an amine base.
- Perform a thermal rearrangement (Newman-Karnes rearrangement) on the O-thiocarbamate to generate the S-dialkyl aryl thiocarbamate intermediate (Formula IIIb).
- Execute catalytic hydrogenation using Raney nickel to remove the sulfur moiety, followed by hydrolysis to yield the final alpha-arylalkanoic acid (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the technology described in CN1217913C offers profound advantages for procurement managers and supply chain directors looking to optimize their sourcing strategies for pharmaceutical intermediates. The primary value driver is the significant reduction in manufacturing costs achieved through the substitution of expensive noble metal catalysts with commodity-grade reagents and Raney nickel. This shift not only lowers the direct material cost but also reduces the dependency on volatile precious metal markets, thereby stabilizing the long-term pricing structure for these critical intermediates. Additionally, the elimination of intermediate purification steps translates into a drastically simplified production workflow, which reduces labor costs, solvent consumption, and energy usage per kilogram of product. These efficiencies compound to offer a highly competitive cost position, enabling suppliers to pass on substantial cost savings to their clients while maintaining healthy margins.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the strategic replacement of high-cost reagents with affordable alternatives like thiophosgene and amines, coupled with the use of Raney nickel instead of palladium. By removing the need for intermediate isolation and purification, the process significantly reduces the consumption of solvents and filtration media, which are often hidden cost drivers in fine chemical manufacturing. Furthermore, the high yield and selectivity of the reaction minimize the loss of valuable starting materials, ensuring that the overall material efficiency is maximized. This comprehensive approach to cost optimization results in a lower cost of goods sold (COGS), making the final alpha-arylalkanoic acids more affordable for downstream drug manufacturers without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals rather than specialized or scarce catalysts enhances the resilience of the supply chain against disruptions. Raney nickel and thiophosgene are produced by multiple global suppliers, reducing the risk of single-source bottlenecks that can delay production schedules. The robustness of the chemical process also means that batches are less likely to fail quality control due to impurity issues, ensuring a consistent and reliable flow of product to customers. This reliability is crucial for pharmaceutical companies that operate on tight production schedules and cannot afford delays in the delivery of key intermediates, thereby strengthening the partnership between the supplier and the end-user.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that can be safely translated from laboratory glassware to large-scale industrial reactors. The reduction in solvent usage and the avoidance of heavy metal contaminants like palladium simplify the waste treatment process, making it easier to comply with increasingly stringent environmental regulations. The ability to run the process without intermediate purification also reduces the volume of waste generated per unit of product, contributing to a smaller environmental footprint. These factors make the technology not only economically attractive but also sustainable, aligning with the corporate social responsibility goals of modern chemical enterprises and ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiocarbamate rearrangement technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this process into their existing manufacturing portfolios or sourcing strategies. The responses highlight the balance between technical innovation and practical application, ensuring that both R&D and commercial teams have a clear understanding of the value proposition.
Q: What are the primary advantages of the thiocarbamate rearrangement method over traditional hydrogenation?
A: The thiocarbamate rearrangement method described in CN1217913C avoids the use of expensive palladium catalysts often required in traditional methods. It utilizes cost-effective reagents like thiophosgene and Raney nickel, and crucially, it does not require the purification of intermediate products, which significantly streamlines the manufacturing process and reduces overall production costs.
Q: How does this process ensure high purity for pharmaceutical intermediates?
A: The process demonstrates high chemoselectivity, particularly protecting carbonyl functions such as benzoyl groups during the reduction step. The use of specific thermal rearrangement conditions followed by controlled catalytic hydrogenation minimizes the formation of side products, ensuring that the final alpha-arylalkanoic acid meets stringent purity specifications required for pharmaceutical applications without complex downstream purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is highly scalable. The reagents used are commodity chemicals available in bulk, and the elimination of intermediate purification steps reduces processing time and waste generation. The use of Raney nickel, a robust and widely available catalyst, further supports the feasibility of scaling this process from laboratory to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Arylalkanoic Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes like the one described in CN1217913C for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory chemistry is successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that monitor every stage of the synthesis, from raw material intake to final product release. We understand that the consistency of alpha-arylalkanoic acids is vital for the efficacy and safety of the final drug product, and our state-of-the-art facilities are designed to meet the highest global regulatory standards.
We invite procurement leaders and technical directors to collaborate with us to leverage this advanced technology for their supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific synthetic route can optimize your current procurement strategy. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Our team is ready to discuss how we can support your development timelines and commercial goals with reliable, high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
