Advanced C-H Borylation for High-Purity Alkyl Borane Intermediates and Commercial Scale-Up
Introduction to Next-Generation Alkyl Borane Synthesis
The strategic incorporation of boron into organic frameworks has revolutionized modern medicinal chemistry, serving as a cornerstone for the development of potent proteasome inhibitors such as Bortezomib and Ixazomib, which exhibit remarkable anti-cancer efficacy. As depicted in the structural comparison below, these molecules rely heavily on precise alkyl borane motifs to achieve their biological activity, highlighting the critical demand for reliable synthetic methodologies in the pharmaceutical sector.
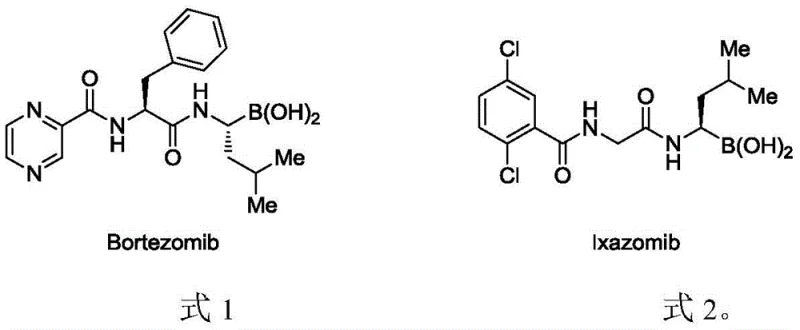
Addressing this critical need, the recent technological disclosure in patent CN113861228A introduces a groundbreaking approach for synthesizing alkyl borane derivatives through direct C-H bond activation. This innovation represents a significant paradigm shift from conventional multi-step protocols, offering a streamlined, one-step synthesis that utilizes aliphatic carboxylic acid derivatives and bis(pinacolato)diborane as starting materials. By leveraging a sophisticated palladium-catalyzed system, this method not only enhances regioselectivity but also broadens the scope of functional group tolerance, positioning it as a vital tool for the efficient production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkyl boranes has been fraught with significant operational challenges and safety hazards that impede efficient commercial manufacturing. Traditional pathways predominantly rely on the single-electron transfer of halogenated alkanes in the presence of metals and strong alkalis, necessitating harsh reaction conditions that are difficult to control on a large scale. Furthermore, these legacy methods invariably require the substrate to be pre-functionalized, which drastically limits the diversity of accessible substrates and inflates the overall cost of goods by adding unnecessary synthetic steps prior to the borylation event.
Alternative strategies, such as those disclosed in earlier literature involving transesterification, often employ highly flammable and moisture-sensitive reagents like trimethylboroxine or toxic gases such as carbon monoxide. These factors introduce severe safety risks and complicate waste management protocols, while the inherent complexity of multi-step procedures leads to relatively low overall reaction efficiency and substantial material loss, making them less attractive for cost-sensitive supply chains.
The Novel Approach
In stark contrast, the methodology outlined in the present patent data circumvents these historical bottlenecks by employing a direct C-H activation strategy that transforms simple aliphatic carboxylic acid derivatives directly into valuable alkyl borane scaffolds. This novel approach eliminates the need for hazardous pre-functionalization, allowing for the direct utilization of abundant and inexpensive feedstock chemicals. The reaction proceeds under relatively mild conditions using a palladium salt catalyst and a silver salt oxidant, facilitating a clean and efficient transformation that preserves the integrity of sensitive functional groups.
The versatility of this new synthetic route is underscored by its exceptional regioselectivity and functional group diversity, enabling the construction of complex molecular architectures that were previously difficult to access. By integrating bases and specific additives into the reaction matrix, the process achieves high yields of polysubstituted alkyl borane derivatives in a single operational step, thereby drastically reducing the time and resources required for production while enhancing the overall sustainability of the manufacturing process.
Mechanistic Insights into Pd-Catalyzed C-H Borylation
The core of this technological advancement lies in the intricate catalytic cycle driven by palladium species, which facilitates the cleavage of inert C-H bonds and their subsequent replacement with boron functionalities. As illustrated in the general reaction scheme, the process initiates with the coordination of the palladium catalyst to the substrate, followed by a concerted metalation-deprotonation (CMD) step that activates the specific C-H bond targeted for functionalization. The presence of bis(pinacolato)diborane serves as the boron source, which undergoes transmetallation with the organopalladium intermediate to install the pinacol boronate ester group.
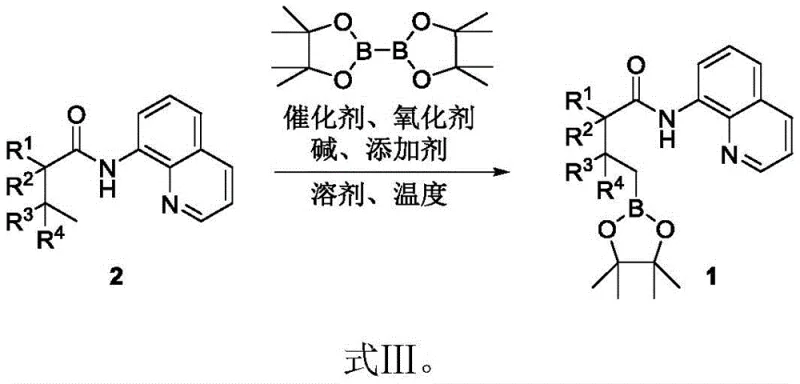
Crucially, the selection of the oxidant, specifically silver carbonate, plays a pivotal role in regenerating the active palladium species and driving the catalytic cycle forward, while additives like 2-chloroquinoline act as ligands to stabilize the transition state and enhance selectivity. This mechanistic precision ensures that side reactions are minimized, leading to a cleaner impurity profile which is paramount for pharmaceutical applications. The ability to tune the reaction outcome by varying the R groups on the aliphatic carboxylic acid derivative allows for the generation of a wide array of structurally diverse intermediates, providing R&D teams with a powerful platform for lead optimization.
How to Synthesize Alkyl Borane Derivatives Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to a precise protocol that balances reagent stoichiometry with thermal parameters to maximize yield and purity. The following guide outlines the standardized procedure derived from the patent examples, ensuring reproducibility and safety during the execution of this C-H activation transformation.
- Combine aliphatic carboxylic acid derivative, bis-pinacolyl diborane, Pd(OPiv)2 catalyst, silver carbonate oxidant, sodium bicarbonate base, and 2-chloroquinoline additive in acetonitrile solvent.
- Stir the reaction mixture at elevated temperatures (preferably 130 °C) for approximately 24 hours under an air atmosphere to facilitate C-H bond activation.
- Purify the crude reaction mixture via silica gel column chromatography using a petroleum ether and ethyl acetate eluent system to isolate the target alkyl borane derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic benefits that extend beyond mere technical feasibility. By shifting away from complex, multi-step sequences involving hazardous reagents, manufacturers can achieve a significant reduction in operational expenditures and mitigate the risks associated with handling volatile or toxic substances. The reliance on commercially available aliphatic carboxylic acid derivatives as starting materials ensures a stable and resilient supply chain, reducing dependency on specialized, hard-to-source precursors that often cause production delays.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps and the use of a one-pot reaction protocol inherently lower the cost of goods sold by reducing solvent consumption, labor hours, and purification burdens. Since the process avoids expensive halogenated substrates and toxic gases, the overhead costs related to safety compliance and waste disposal are substantially diminished, leading to a more economically viable production model for high-value intermediates.
- Enhanced Supply Chain Reliability: Utilizing robust and readily available raw materials such as bis(pinacolato)diborane and simple carboxylic acids minimizes the risk of supply disruptions caused by the scarcity of exotic reagents. The reaction's tolerance to air atmospheres and its operation in common solvents like acetonitrile further simplify logistics, allowing for flexible manufacturing schedules and faster turnaround times for custom synthesis projects.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this C-H activation method make it exceptionally well-suited for scale-up from kilogram to multi-ton quantities without compromising safety or quality. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, positioning companies that adopt this technology as leaders in sustainable chemical manufacturing and green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method, providing clarity for technical teams evaluating its integration into existing workflows. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation.
Q: What are the primary advantages of this C-H activation method over traditional halogenation routes?
A: Traditional methods often require pre-functionalized halogenated alkanes and harsh conditions involving strong bases or toxic gases like carbon monoxide. This novel method utilizes readily available aliphatic carboxylic acid derivatives, operates under milder conditions, and eliminates the need for substrate pre-functionalization, thereby simplifying the workflow and enhancing safety.
Q: Can the alkyl borane products be further functionalized for drug synthesis?
A: Yes, the resulting alkyl borane derivatives possess high synthetic utility. The amide group within the skeleton can be hydrolyzed to yield corresponding carboxylic acids, and the alkyl boron moiety serves as a versatile handle for further functionalization, making them ideal precursors for complex bioactive molecules and API intermediates.
Q: Is this synthesis method suitable for large-scale industrial production?
A: The method demonstrates excellent potential for scale-up due to its use of commercially available raw materials, a simple one-step reaction protocol, and robust reaction conditions that tolerate air atmospheres. The high regioselectivity and yield reported in the patent data suggest it is well-suited for commercial manufacturing of complex intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Borane Derivative Supplier
As the global demand for boron-containing therapeutics continues to surge, partnering with an experienced CDMO capable of executing complex C-H activation chemistries is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of alkyl borane intermediate meets the highest industry standards for downstream API synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative Pd-catalyzed borylation technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined route, along with access to specific COA data and comprehensive route feasibility assessments that will accelerate your development timeline.
