Revolutionizing Vitamin E Production: A Deep Dive into Non-Corrosive Catalytic Technology
Revolutionizing Vitamin E Production: A Deep Dive into Non-Corrosive Catalytic Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to optimize the synthesis of critical nutrients like Vitamin E. Patent CN1190431C introduces a groundbreaking approach to the manufacture of (all-rac)-alpha-tocopherol, utilizing a novel catalyst system that addresses long-standing inefficiencies in traditional condensation reactions. This technology replaces corrosive Lewis acids with hydrogen tris(oxalato)phosphate, offering a pathway to higher purity and reduced environmental impact. For R&D directors and procurement specialists, understanding this shift is vital for securing a reliable alpha-tocopherol intermediate supplier capable of meeting stringent global standards. The patent details a process where trimethylhydroquinone reacts with isophytol or phytol in the presence of this unique phosphate catalyst, achieving yields that rival or exceed conventional methods while drastically simplifying downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of d,l-alpha-tocopherol has relied heavily on catalyst systems involving zinc chloride, boron trifluoride, or strong mineral acids like hydrochloric acid. These traditional methods present severe operational challenges, primarily due to the highly corrosive nature of the reagents which accelerates equipment degradation and increases maintenance overheads. Furthermore, processes utilizing zinc or iron salts generate significant quantities of heavy metal-contaminated wastewater, creating substantial environmental liabilities and complicating waste disposal protocols. The formation of undesirable by-products, such as phytyl toluene and chlorophytal, is also common in these acidic environments, necessitating complex and costly purification steps to achieve the required pharmaceutical grade purity. Additionally, the difficulty in separating the catalyst from the final product often leads to residual metal impurities, which is a critical failure point for high-value API intermediates intended for human consumption.
The Novel Approach
In stark contrast, the methodology disclosed in CN1190431C utilizes hydrogen tris(oxalato)phosphate, a protic acid catalyst that circumvents the pitfalls of heavy metal usage. This innovative approach operates effectively in organic solvents, including polar aprotic solvents like gamma-butyrolactone or biphasic systems involving alkylene carbonates and aliphatic hydrocarbons. The novel catalyst demonstrates exceptional selectivity, remarkably suppressing the dehydration of allylic alcohols into phytadienes, a common side reaction that plagues acid-catalyzed condensations. By eliminating the need for corrosive Lewis acids, this process not only extends the lifespan of reactor vessels but also ensures that the final product is free from toxic metal residues. The ability to recover and reuse the catalyst further enhances the economic viability of this route, positioning it as a superior alternative for cost reduction in vitamin E manufacturing compared to legacy technologies.
Mechanistic Insights into Hydrogen Tris(oxalato)phosphate Catalysis
The core of this technological advancement lies in the unique chemical structure and behavior of the hydrogen tris(oxalato)phosphate catalyst. Unlike simple mineral acids, this complex phosphate species offers a controlled acidic environment that facilitates the Friedel-Crafts type alkylation of trimethylhydroquinone without promoting excessive decomposition of the sensitive isophytol substrate. The catalyst functions by protonating the hydroxyl group of the isophytol, generating a reactive carbocation intermediate that subsequently attacks the electron-rich aromatic ring of the hydroquinone. Crucially, the steric and electronic properties of the tris(oxalato) ligand sphere appear to modulate the acidity, preventing the harsh conditions that typically lead to polymerization or elimination reactions. This precise control over the reaction pathway is essential for maintaining high atom economy and ensuring that the stereochemical integrity of the product mixture remains within acceptable limits for industrial applications.
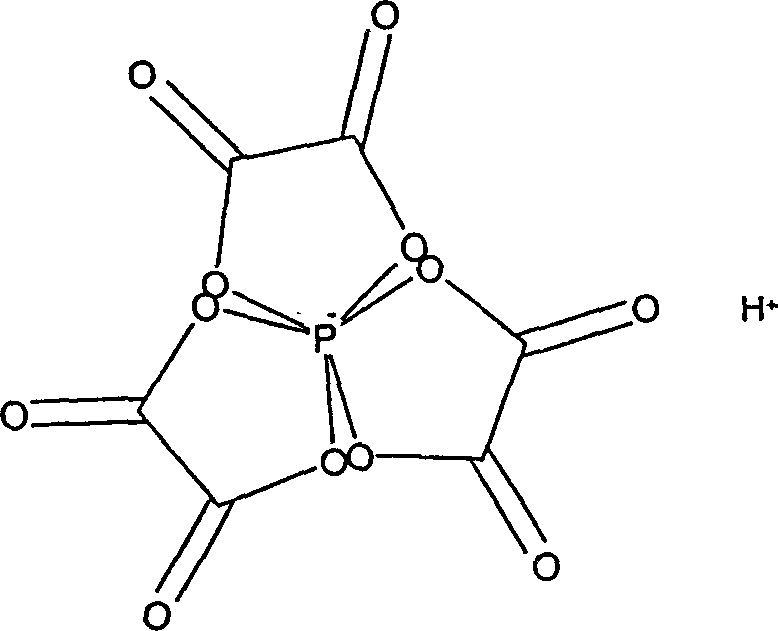
Furthermore, the mechanism allows for operation in a variety of solvent systems, providing flexibility for process optimization. The patent highlights that the reaction can proceed efficiently in single-phase polar solvents or biphasic mixtures, where the non-polar phase aids in the extraction of the product while the polar phase retains the catalyst. This phase behavior is instrumental in the ease of separation, as the catalyst remains largely in the polar layer or can be precipitated out, avoiding the emulsions often encountered with surfactant-like by-products in older methods. The suppression of furan derivatives and other cyclic by-products is another mechanistic advantage, attributed to the specific interaction between the phosphate center and the reactants. For technical teams, this implies a cleaner reaction profile that reduces the burden on distillation columns and crystallization units, directly translating to energy savings and higher throughput capabilities in a commercial plant setting.
How to Synthesize (all-rac)-alpha-tocopherol Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst and the control of reaction parameters to maximize yield and purity. The process begins with the in-situ generation or separate preparation of the hydrogen tris(oxalato)phosphate, typically by reacting phosphorus pentachloride with anhydrous oxalic acid in an aprotic solvent like diethyl ether. Once the catalyst is prepared, it is introduced to a suspension of trimethylhydroquinone in a suitable solvent system, such as propylene carbonate or a mixture of ethylene carbonate and heptane. The reaction is then initiated by the controlled addition of isophytol at elevated temperatures, generally ranging from 90°C to 125°C, under an inert atmosphere to prevent oxidation. Detailed standardized synthesis steps see the guide below.
- Preparation of the catalyst by reacting phosphorus pentachloride with anhydrous oxalic acid in an aprotic solvent like diethyl ether.
- Suspension of trimethylhydroquinone in a polar aprotic solvent such as gamma-butyrolactone or a biphasic system.
- Addition of isophytol and the catalyst at temperatures between 90°C and 125°C under an inert atmosphere.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology represents a strategic opportunity to optimize the total cost of ownership for Vitamin E intermediates. The elimination of heavy metal catalysts like zinc chloride removes the need for expensive chelating agents and complex wastewater treatment facilities, resulting in significant operational expenditure reductions. Moreover, the non-corrosive nature of the new catalyst system reduces the frequency of reactor maintenance and replacement, ensuring higher asset utilization rates and uninterrupted production schedules. The high selectivity of the process minimizes the formation of hard-to-remove impurities, which streamlines the purification workflow and shortens the overall batch cycle time. These factors combined contribute to a more resilient supply chain capable of delivering high-purity all-rac-alpha-tocopherol with greater consistency and reliability.
- Cost Reduction in Manufacturing: The transition to a metal-free catalyst system fundamentally alters the cost structure of production by removing the expenses associated with hazardous waste disposal and metal scavenging. Since the catalyst can be recovered and reused multiple times without significant loss of activity, the raw material cost per kilogram of product is drastically lowered. Additionally, the simplified work-up procedure reduces solvent consumption and energy usage during distillation, further enhancing the margin profile. This economic efficiency makes the process highly competitive for large-scale commercial scale-up of complex vitamin intermediates, allowing suppliers to offer more aggressive pricing without compromising quality standards.
- Enhanced Supply Chain Reliability: Reliance on corrosive and toxic reagents often introduces supply risks due to strict regulatory controls on transportation and storage. By utilizing a safer, organic phosphate-based catalyst, the logistical complexity is reduced, ensuring a smoother flow of materials into the manufacturing site. The robustness of the reaction conditions, which tolerate a range of solvents and moderate temperatures, means that production is less susceptible to minor fluctuations in utility supplies or raw material grades. This stability is crucial for maintaining continuous supply to downstream formulators who depend on just-in-time delivery of critical nutritional ingredients for their own production lines.
- Scalability and Environmental Compliance: As global environmental regulations tighten, processes that generate heavy metal sludge are becoming increasingly untenable. This patented method aligns perfectly with green chemistry principles by avoiding toxic metals and reducing the E-factor of the synthesis. The scalability is proven by the ability to run the reaction in continuous or batch modes with simple addition protocols, making it suitable for expansion from pilot plants to multi-tonne annual capacities. Companies adopting this technology future-proof their operations against impending environmental legislation, securing their license to operate and enhancing their corporate sustainability profile in the eyes of stakeholders and customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic process for alpha-tocopherol synthesis. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into existing manufacturing frameworks or sourcing strategies. The answers reflect the balance between chemical performance and operational practicality required for industrial success.
Q: What are the primary advantages of hydrogen tris(oxalato)phosphate over traditional zinc chloride catalysts?
A: Unlike traditional zinc chloride or boron trifluoride systems, this novel catalyst eliminates heavy metal contamination in wastewater and significantly reduces equipment corrosion, leading to lower maintenance costs and easier regulatory compliance.
Q: How does this process control the formation of phytadiene by-products?
A: Despite being a protic acid, the specific structure of the tris(oxalato)phosphate catalyst demonstrates surprisingly high selectivity, minimizing the dehydration of allylic alcohols like isophytol into unwanted phytadienes.
Q: Is the catalyst reusable in this synthesis method?
A: Yes, the patent data indicates that the catalyst can be easily separated from the reaction mixture and reused multiple times without significant loss of activity, enhancing overall process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Tocopherol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN1190431C for the Vitamin E market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are successfully translated into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-tocopherol intermediate meets the highest international pharmacopeial standards. Our commitment to technical excellence allows us to navigate the complexities of novel synthesis routes, delivering products that support the health and nutrition sectors globally.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this cleaner, more efficient production method. We encourage potential partners to contact us for specific COA data and route feasibility assessments, enabling you to make informed decisions that enhance your supply chain resilience. Let us collaborate to drive innovation and efficiency in the production of essential life-science ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
