Advanced Synthesis of 5-Methyl-2-Trifluoromethyl Benzoxazole for Commercial Pharmaceutical Applications
Advanced Synthesis of 5-Methyl-2-Trifluoromethyl Benzoxazole for Commercial Pharmaceutical Applications
The introduction of fluorine atoms into organic scaffolds represents a cornerstone strategy in modern medicinal chemistry, profoundly enhancing the metabolic stability and lipophilicity of drug candidates. Patent CN101012205A discloses a highly efficient and robust synthetic methodology for producing 5-methyl-2-trifluoromethyl benzoxazole, a critical fluorinated heterocyclic building block. This specific intermediate serves as a pivotal precursor in the development of advanced pharmaceutical agents and agrochemical formulations, where the trifluoromethyl group imparts superior biological activity and resistance to enzymatic degradation. The disclosed process leverages a modified Appel-type cyclization condition, utilizing a synergistic combination of triphenylphosphine, triethylamine, and trifluoroacetic acid in a carbon tetrachloride solvent system. By operating under mild reflux conditions, this technique achieves exceptional conversion rates while minimizing the formation of complex impurity profiles often associated with harsher dehydration protocols. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediates supplier, this technology offers a validated pathway to secure high-quality materials with consistent batch-to-batch reproducibility.
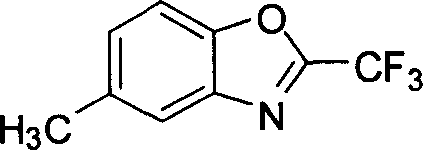
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of benzoxazole rings bearing electron-withdrawing groups has presented significant challenges in process chemistry, often necessitating the use of aggressive dehydrating agents such as thionyl chloride or polyphosphoric acid. These traditional methodologies frequently suffer from poor atom economy, generating substantial quantities of acidic waste streams that complicate downstream processing and environmental compliance. Furthermore, conventional thermal cyclization of o-aminophenols with carboxylic acids often requires excessively high temperatures, which can lead to the decomposition of sensitive functional groups and the formation of polymeric tars that are difficult to separate. The reliance on stoichiometric amounts of hazardous reagents not only escalates the raw material costs but also introduces severe safety risks during the scale-up phase, particularly when handling exothermic reactions on a multi-kilogram scale. Additionally, older synthetic routes often lack regioselectivity control, potentially yielding isomeric mixtures that require tedious and costly chromatographic separations, thereby reducing the overall throughput and economic viability of the manufacturing process.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN101012205A introduces a streamlined one-pot synthesis that dramatically simplifies the operational workflow while maximizing yield efficiency. By employing a specific molar ratio of triphenylphosphine to triethylamine and trifluoroacetic acid, the reaction proceeds through a mild activation mechanism that facilitates ring closure at relatively moderate temperatures. This approach effectively circumvents the need for extreme thermal conditions, preserving the integrity of the trifluoromethyl moiety and preventing side reactions that typically plague high-temperature syntheses. The use of carbon tetrachloride as a solvent, while requiring careful handling, provides an ideal medium for the solubility of the phosphine complexes and ensures homogeneous reaction kinetics throughout the reflux period. The result is a process that delivers yields ranging from 75% to 99%, representing a substantial improvement over conventional benchmarks and offering a compelling value proposition for cost reduction in fine chemical manufacturing. This methodological shift allows producers to achieve commercial scale-up of complex heterocyclic intermediates with greater confidence in process safety and environmental manageability.
Mechanistic Insights into Triphenylphosphine-Mediated Cyclization
The core of this synthetic innovation lies in the in situ generation of a highly reactive acylating species through the interaction of trifluoroacetic acid with the triphenylphosphine-carbon tetrachloride system. Mechanistically, the triphenylphosphine attacks the carbon tetrachloride to generate a dichlorotriphenylphosphorane intermediate, which subsequently activates the carboxylic acid group of the trifluoroacetic acid. This activation step converts the relatively unreactive hydroxyl group of the acid into a superior leaving group, effectively forming a mixed anhydride or an acyl chloride equivalent under mild conditions. The ortho-amino group of the 5-methyl-2-hydroxyaniline then acts as a nucleophile, attacking the activated carbonyl carbon to form an amide intermediate. Following this initial acylation, the proximal phenolic hydroxyl group undergoes an intramolecular nucleophilic attack on the imidoyl carbon, facilitated by the basic environment provided by triethylamine. This cyclization step eliminates a molecule of water (captured by the phosphine species) and aromatizes the oxazole ring, driving the reaction to completion with high thermodynamic favorability.
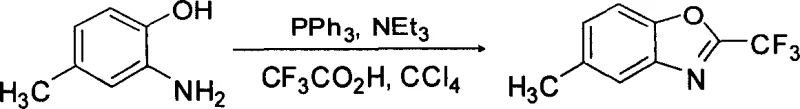
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of oligomeric byproducts. The rapid consumption of the activated acid species by the amino-phenol substrate prevents self-condensation of the acid or polymerization of the aniline derivative, which are common failure modes in less controlled systems. The presence of triethylamine serves a dual purpose: it scavenges the hydrochloric acid generated during the activation phase and maintains a neutral to slightly basic pH that protects the acid-sensitive benzoxazole ring from hydrolysis. Furthermore, the byproduct of the reaction, triphenylphosphine oxide, is a solid that can be easily separated from the liquid product during the workup phase, either through filtration or simple washing protocols. This clean reaction profile ensures that the final crude material possesses a high degree of chemical purity, reducing the burden on the purification team and allowing for more efficient column chromatography or crystallization steps to achieve the stringent specifications required for GMP-grade intermediates.
How to Synthesize 5-Methyl-2-Trifluoromethyl Benzoxazole Efficiently
The execution of this synthesis requires precise adherence to the molar ratios and temperature controls specified in the patent to ensure optimal performance and safety. The process begins with the preparation of the reaction vessel under an inert nitrogen atmosphere to prevent moisture ingress, which could deactivate the phosphine reagent. Reagents are added in a specific sequence to manage the exotherm associated with the formation of the phosphorane intermediate, ensuring that the reaction mixture remains stable before heating commences. Once the reflux temperature is reached, the system is maintained for a defined period to allow full conversion, monitored typically by TLC or HPLC to confirm the disappearance of the starting aminophenol. The detailed standardized synthesis steps, including exact quantities for pilot and commercial scales, are outlined in the technical guide below.
- Under nitrogen protection, dissolve triphenylphosphine, triethylamine, trifluoroacetic acid, and 5-methyl-2-hydroxyaniline in carbon tetrachloride at a molar ratio of 3: 3:1:1~1.2.
- Stir the mixture in an ice-water bath until fully dissolved, then heat to reflux for 3 to 5 hours to complete the cyclization reaction.
- Remove solvent under reduced pressure, purify the residue via neutral alumina column chromatography using petroleum ether and ethyl acetate (10: 1) to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into enhanced operational resilience and significant cost optimization opportunities. The reliance on commodity chemicals such as triphenylphosphine, triethylamine, and trifluoroacetic acid ensures that the raw material supply base is broad and competitive, mitigating the risk of single-source bottlenecks that often plague specialty reagent markets. The high yield profile of 75% to 99% drastically reduces the volume of waste generated per kilogram of product, lowering disposal costs and aligning with increasingly strict environmental regulations regarding chemical manufacturing effluents. Moreover, the simplicity of the workup procedure, which involves solvent evaporation and straightforward chromatographic purification, reduces the requirement for complex unit operations and specialized equipment, thereby lowering the capital expenditure barrier for production facilities.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous dehydrating agents like thionyl chloride removes the need for specialized corrosion-resistant reactors and extensive scrubbing systems, leading to substantial capital and operational savings. The high atom efficiency of the reaction ensures that a greater proportion of the input mass is converted into valuable product, directly improving the cost of goods sold (COGS) metrics for the final intermediate. Additionally, the ability to recover and recycle the solvent system further contributes to long-term economic sustainability, making this route highly attractive for high-volume production scenarios where margin compression is a constant concern.
- Enhanced Supply Chain Reliability: By utilizing reagents that are produced on a multi-ton global scale, manufacturers can secure long-term supply contracts with favorable pricing structures, insulating the production schedule from market volatility. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in utility parameters such as cooling water temperature or steam pressure, ensuring consistent output quality even in diverse manufacturing environments. This reliability is critical for maintaining just-in-time delivery schedules for downstream API manufacturers who depend on a steady flow of high-quality heterocyclic building blocks to meet their own production targets.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis minimizes the number of isolation and transfer steps, reducing the potential for material loss and operator exposure to hazardous substances. The process generates primarily solid phosphine oxide waste and aqueous amine salts, which are easier to treat and dispose of compared to the gaseous byproducts of traditional chlorination methods. This cleaner waste profile simplifies the permitting process for new production lines and supports corporate sustainability goals by reducing the overall carbon footprint and E-factor of the chemical synthesis, positioning the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated benzoxazole derivative. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this intermediate into their specific development pipelines.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method utilizes a mild one-pot cyclization strategy that avoids harsh dehydrating agents, resulting in significantly higher yields (75-99%) and simplified purification processes suitable for scale-up.
Q: Is this intermediate suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process uses readily available commodity reagents like triphenylphosphine and trifluoroacetic acid, ensuring supply chain stability and cost-effectiveness for metric-ton production.
Q: What is the expected purity profile of the final benzoxazole derivative?
A: The protocol employs neutral alumina column chromatography which effectively removes phosphine oxide byproducts, delivering high-purity material essential for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Methyl-2-Trifluoromethyl Benzoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends on the availability of high-quality, consistently manufactured intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 5-methyl-2-trifluoromethyl benzoxazole meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance extends beyond mere compliance; we actively collaborate with our clients to optimize process parameters for their specific needs, ensuring maximum yield and minimal impurity levels.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can enhance your supply chain efficiency and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this superior methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be your strategic partner in navigating the complexities of fine chemical sourcing and accelerating your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
