Scalable Synthesis of Fluorinated Imidoyl Chlorides for High-Value Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for synthesizing fluorinated intermediates, which serve as critical building blocks for next-generation bioactive compounds. Patent CN1927818A introduces a highly efficient preparation method for N-(2-methyl-4-nitrophenyl)-2,2,2-trifluoroacetimidoyl chloride, a specialized fluorine-containing imine acyl chloride. This compound is characterized by its active carbon-chlorine bond and the presence of a strong electron-withdrawing nitro group on the phenyl ring, making it an exceptionally versatile precursor for constructing heterocyclic systems. The disclosed technology leverages a one-pot synthetic strategy that significantly streamlines the production process compared to traditional multi-step approaches. By utilizing a specific combination of triphenylphosphine, triethylamine, and trifluoroacetic acid under controlled thermal conditions, the method achieves remarkable yields ranging from 65% to 94%. For R&D directors and procurement specialists, this represents a pivotal advancement in accessing high-purity fluorinated scaffolds that are essential for modifying host molecules to enhance physiological activity and metabolic stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex fluorinated imidoyl chlorides has been plagued by inefficient multi-step protocols that require rigorous isolation and purification between each stage. Conventional routes often involve harsh reaction conditions, expensive catalysts, or unstable intermediates that degrade rapidly upon exposure to moisture or air, leading to inconsistent batch quality and reduced overall throughput. The reliance on multiple solvent exchanges and extensive work-up procedures not only increases the operational complexity but also escalates the environmental footprint through higher waste generation. Furthermore, traditional methods frequently struggle to control the regioselectivity of substitution on the phenyl ring, resulting in impurity profiles that are difficult to manage during downstream processing. These inefficiencies translate directly into higher manufacturing costs and extended lead times, creating significant bottlenecks for supply chain managers who require reliable volumes of specialty chemicals for continuous production lines.
The Novel Approach
In stark contrast, the methodology outlined in CN1927818A offers a transformative one-pot solution that consolidates multiple reaction steps into a single, cohesive process. By employing carbon tetrachloride as a solvent and orchestrating the reaction between triphenylphosphine, triethylamine, trifluoroacetic acid, and 2-methyl-4-nitroaniline, the process eliminates the need for intermediate isolation. The reaction is initiated at low temperatures (0°C) to control the initial mixing and then allowed to proceed through a controlled exothermic phase, ensuring high conversion rates without the need for exotic reagents. This approach drastically simplifies the operational workflow, reducing the manpower and equipment time required for production. The ability to achieve yields as high as 94% with minimal byproduct formation demonstrates a superior level of process control, offering a compelling value proposition for manufacturers aiming to optimize their cost structures while maintaining stringent quality standards for high-value pharmaceutical intermediates.
Mechanistic Insights into Triphenylphosphine-Mediated Fluorination
The core of this innovative synthesis lies in the intricate interplay between triphenylphosphine and trifluoroacetic acid, which facilitates the activation of the carboxylic acid towards nucleophilic attack by the amine. Under nitrogen protection, the triphenylphosphine acts as a potent dehydrating agent, reacting with trifluoroacetic acid to generate a highly reactive mixed anhydride or acyl phosphonium intermediate in situ. This activation step is crucial as it lowers the energy barrier for the subsequent formation of the imidoyl chloride bond. The addition of triethylamine serves to scavenge the generated protons, driving the equilibrium forward and preventing the protonation of the amine nucleophile, which would otherwise render it unreactive. The specific molar ratios, ranging from 2.5 to 3.5 equivalents of triphenylphosphine relative to the acid, ensure that the activation is complete and that the reaction proceeds to full conversion without stalling at the amide stage.
Following the initial activation, the introduction of 2-methyl-4-nitroaniline triggers the cyclization and chlorination sequence that defines the final product structure. The nitro group on the aniline ring plays a dual role; it acts as a strong electron-withdrawing group that modulates the nucleophilicity of the amino group, preventing over-reaction, while also enhancing the physiological potential of the final molecule. The reaction mixture is heated to induce a vigorous exothermic response, typically around 60°C, which provides the necessary thermal energy to overcome the activation barrier for the elimination of water and the formation of the carbon-chlorine double bond character. 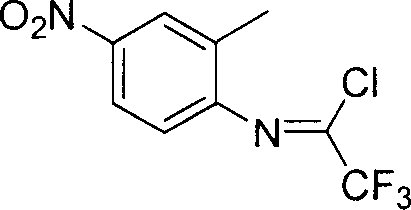 This structural integrity is preserved throughout the work-up, where the use of a petroleum ether and ethyl acetate mixture allows for the selective precipitation of triphenylphosphine oxide byproducts, leaving the desired yellow liquid product in solution for final distillation.
This structural integrity is preserved throughout the work-up, where the use of a petroleum ether and ethyl acetate mixture allows for the selective precipitation of triphenylphosphine oxide byproducts, leaving the desired yellow liquid product in solution for final distillation.
How to Synthesize N-(2-Methyl-4-Nitrophenyl)-2,2,2-Trifluoroacetimidoyl Chloride Efficiently
Implementing this synthesis requires precise adherence to the stoichiometric ratios and thermal profiles defined in the patent to ensure reproducibility and safety. The process begins with the careful preparation of the reaction vessel under an inert atmosphere to prevent moisture ingress, which could hydrolyze the sensitive acyl chloride functionality. Operators must monitor the exothermic profile closely during the heating phase to maintain optimal reaction kinetics without risking thermal runaway.
- Prepare the reaction vessel under nitrogen protection with carbon tetrachloride solvent, adding triphenylphosphine and triethylamine while cooling to 0°C.
- Introduce trifluoroacetic acid followed by 2-methyl-4-nitroaniline, maintaining specific molar ratios to ensure complete conversion.
- Heat to initiate exothermic reaction, maintain reflux for 3 to 5 hours, then isolate the product via reduced pressure distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere technical feasibility. The reliance on commercially abundant raw materials such as triphenylphosphine, triethylamine, and trifluoroacetic acid ensures a stable supply base that is not subject to the volatility often associated with specialized organometallic catalysts. This accessibility translates directly into enhanced supply chain reliability, as sourcing risks are minimized and lead times for raw material acquisition are significantly reduced. Furthermore, the one-pot nature of the reaction drastically cuts down on solvent consumption and waste disposal costs, aligning with modern environmental compliance standards and reducing the overall cost of goods sold. The high yield profile means that less raw material is wasted per unit of output, providing a natural hedge against raw material price fluctuations and improving margin stability for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the use of standard solvents like carbon tetrachloride and petroleum ether significantly lower the operational expenditure associated with production. By avoiding the need for expensive transition metal catalysts or cryogenic conditions, the process reduces energy consumption and equipment wear, leading to substantial cost savings over the lifecycle of the product. The high atom economy and yield efficiency mean that the effective cost per kilogram of the active intermediate is optimized, allowing for more competitive pricing strategies in the global market without compromising on quality or purity specifications.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup allows for rapid scale-up from laboratory benchtop to industrial reactor sizes without the need for specialized engineering modifications. This scalability ensures that supply can be ramped up quickly to meet surges in demand, providing a buffer against market volatility. Additionally, the robustness of the chemistry reduces the likelihood of batch failures, ensuring a consistent flow of material to downstream customers and minimizing the risk of production stoppages due to quality deviations or supply shortages of critical reagents.
- Scalability and Environmental Compliance: The process generates solid byproducts like triphenylphosphine oxide that can be easily filtered and potentially recycled, reducing the burden on waste treatment facilities. The use of reduced pressure distillation for final purification is a standard industrial unit operation that is easily integrated into existing infrastructure, facilitating seamless technology transfer. This alignment with standard industrial practices ensures that the manufacturing process remains compliant with increasingly stringent environmental regulations regarding solvent emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated intermediate. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What are the primary advantages of this one-pot synthesis method?
A: The method eliminates complex multi-step isolation procedures, utilizes readily available raw materials, and achieves yields between 65% and 94%, significantly simplifying the manufacturing workflow.
Q: How does the fluorine incorporation affect the biological activity?
A: Incorporating fluorine atoms enhances metabolic stability and lipophilicity, which improves absorption and transmission speed in vivo, making the compound highly valuable for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol uses common solvents like carbon tetrachloride and standard reagents, and the exothermic nature is manageable, making it highly adaptable for commercial scale-up from kilograms to tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2-Methyl-4-Nitrophenyl)-2,2,2-Trifluoroacetimidoyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of advanced pharmaceuticals and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence means that we do not just supply chemicals; we provide validated solutions that integrate smoothly into your existing manufacturing workflows, minimizing risk and maximizing output.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to specific COA data and route feasibility assessments that empower you to make informed decisions about your raw material strategy. Contact us today to discuss how we can support your R&D and production goals with reliable, high-performance chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
