Advanced Chiral Phosphonic Amide Esters: Enabling High-Selectivity Asymmetric Catalysis for Global Pharma
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and fine chemical sectors. Patent CN102070671A introduces a significant advancement in this domain through the development of novel chiral phosphonic amide ester compounds. These compounds, specifically characterized by their 4H-1,3,2-benzoxazaphosphole oxide core structure, represent a breakthrough in ligand design for transition metal catalysis. The innovation lies not merely in the molecular architecture but in the practical application of these molecules as highly efficient catalysts for asymmetric cyanosilylation reactions. For R&D directors and process chemists, this patent offers a tangible pathway to achieving superior enantiomeric excess (ee) values, which is critical for the production of active pharmaceutical ingredients (APIs). The technology addresses the perennial challenge of balancing catalytic activity with stereocontrol, providing a robust platform for synthesizing complex chiral building blocks. By leveraging the unique electronic and steric properties of the phosphonic amide ester moiety combined with the chiral oxazoline ring, this invention sets a new benchmark for performance in asymmetric transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral cyanohydrins and related intermediates has relied heavily on traditional Lewis acid catalysts or early-generation chiral ligands that often suffer from significant drawbacks. Many conventional systems require cryogenic temperatures to maintain stereocontrol, leading to excessive energy consumption and operational complexity in a manufacturing setting. Furthermore, traditional catalysts frequently exhibit modest turnover numbers and limited substrate scope, necessitating high catalyst loadings that drive up production costs and complicate downstream purification processes. The presence of residual metals in the final product is another persistent issue, often requiring additional scavenging steps that reduce overall yield and extend lead times. In the context of large-scale pharmaceutical manufacturing, these inefficiencies translate into substantial economic burdens and supply chain vulnerabilities. The reliance on sensitive reagents that degrade rapidly upon exposure to moisture or air further exacerbates these challenges, demanding stringent handling protocols that are difficult to maintain consistently across different production batches. Consequently, there is a pressing need for more robust, efficient, and scalable catalytic systems that can overcome these inherent limitations.
The Novel Approach
The methodology outlined in CN102070671A presents a transformative solution by utilizing a distinct class of chiral phosphonic amide esters that operate effectively under milder conditions. Unlike traditional systems that may struggle with stability, these novel catalysts demonstrate remarkable resilience and activity at temperatures ranging from 20°C to 30°C, significantly reducing energy requirements. The structural integration of the oxazoline ring with the phosphorus center creates a rigid chiral environment that effectively directs the stereochemical outcome of the reaction, as evidenced by the high enantiomeric excess values reported in the patent data. This approach eliminates the need for extreme cooling, thereby simplifying reactor operations and enhancing safety profiles. Moreover, the synthetic route to the catalyst itself is streamlined, involving straightforward condensation and phosphorylation steps that utilize widely available starting materials. This accessibility ensures that the catalyst can be produced reliably at scale, mitigating supply risks associated with exotic or proprietary reagents. For procurement managers, this translates to a more predictable cost structure and reduced dependency on single-source suppliers for critical catalytic components.
Mechanistic Insights into Phosphonic Amide Ester Catalysis
The exceptional performance of these chiral phosphonic amide esters can be attributed to the sophisticated interplay between the phosphorus atom and the chiral oxazoline scaffold. The phosphorus center acts as a potent Lewis acid, activating the electrophilic substrate through coordination, while the adjacent nitrogen atom in the oxazoline ring provides additional stabilization and stereochemical guidance. This bidentate coordination mode creates a well-defined chiral pocket that restricts the approach of the nucleophile, ensuring high facial selectivity during the bond-forming event. The substituents on the oxazoline ring, derived from various L-amino alcohols such as L-leucinol or L-phenylalaninol, play a crucial role in tuning the steric bulk around the active site. For instance, the benzyl group in the 2c variant provides significant steric hindrance that effectively blocks one face of the substrate, leading to the observed 98% ee in specific transformations. Understanding this mechanism allows process chemists to rationally select or modify the R-group to optimize performance for different substrates, offering a level of tunability that is often absent in rigid, non-modular catalyst systems.
Controlling impurity profiles is paramount in the synthesis of pharmaceutical intermediates, and the mechanism of this catalytic system inherently supports high purity outcomes. The high specificity of the catalyst minimizes the formation of side products and regioisomers, which are common contaminants in less selective reactions. The reaction proceeds through a concerted transition state that favors the desired enantiomer, thereby reducing the burden on downstream purification units such as crystallization or chromatography. Additionally, the stability of the catalyst under reaction conditions prevents decomposition pathways that could generate difficult-to-remove byproducts. This intrinsic selectivity not only improves the quality of the final product but also enhances the overall mass balance of the process. For quality assurance teams, this means fewer out-of-specification batches and a more consistent product profile, which is essential for regulatory compliance. The ability to achieve such high levels of control without resorting to complex additive packages further simplifies the formulation and reduces the risk of introducing new impurities into the system.
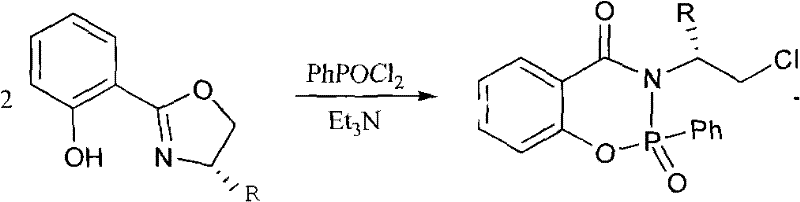
How to Synthesize Chiral Phosphonic Amide Ester Efficiently
The preparation of these high-value catalysts follows a logical two-step sequence that is amenable to standard organic synthesis infrastructure. The process begins with the formation of the chiral oxazoline intermediate, followed by phosphorylation to install the active phosphonic amide motif. This modular approach allows for the independent optimization of each step, ensuring maximum yield and purity before proceeding to the final stage. The use of common solvents like chlorobenzene and toluene facilitates easy solvent recovery and recycling, aligning with green chemistry principles. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are critical for reproducibility. The following guide outlines the generalized workflow for producing these catalysts, serving as a foundational reference for process development teams aiming to implement this technology.
- Prepare the oxazoline intermediate by refluxing 2-cyanophenol with L-amino alcohol in chlorobenzene using ZnCl2 catalyst under anhydrous conditions for 24 hours.
- Isolate the intermediate via extraction and column chromatography to ensure high purity before the phosphorylation step.
- React the purified intermediate with phenyl phosphonyl chloride in toluene and triethylamine under reflux for 24 hours to yield the target chiral catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers compelling advantages for procurement and supply chain stakeholders. The primary benefit lies in the simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By enabling reactions to proceed at near-ambient temperatures, the technology eliminates the need for expensive cryogenic cooling systems, resulting in significant energy savings over the lifecycle of the production campaign. Furthermore, the high selectivity of the catalyst reduces the volume of waste generated, lowering disposal costs and minimizing the environmental footprint of the manufacturing site. These efficiency gains contribute to a more sustainable and cost-effective production model, enhancing the overall competitiveness of the supply chain. For supply chain heads, the robustness of the process ensures consistent output, reducing the risk of delays caused by batch failures or extended processing times.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven by the elimination of complex purification steps and the reduction in catalyst loading requirements. Because the catalyst exhibits high turnover and selectivity, less material is needed to achieve the same conversion, directly lowering the raw material cost per kilogram of product. Additionally, the avoidance of heavy metal scavengers or extensive chromatographic purification reduces the consumption of auxiliary chemicals and consumables. This streamlined workflow translates to substantial cost savings in both direct material costs and labor hours associated with processing. The use of readily available starting materials further stabilizes pricing, protecting the project from volatility associated with specialty reagents. Overall, the process economics favor a leaner, more efficient manufacturing operation that maximizes margin potential.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any manufacturing operation, and this technology supports reliability through the use of commoditized feedstocks. The key precursors, such as 2-cyanophenol and L-amino alcohols, are produced by multiple global suppliers, mitigating the risk of single-source bottlenecks. The synthetic route is robust and tolerant to minor variations in input quality, ensuring that production schedules can be maintained even when facing supply fluctuations. This resilience is particularly valuable in the current geopolitical climate, where supply chain disruptions are a constant concern. By diversifying the source of raw materials and simplifying the synthesis, companies can build a more agile and responsive supply network. This reliability extends to the catalyst itself, which can be stockpiled effectively due to its stability, providing a buffer against unexpected demand spikes.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces unforeseen challenges, but this catalytic system is designed with scalability in mind. The reaction conditions are compatible with standard stainless steel reactors, avoiding the need for specialized glass-lined or Hastelloy equipment. The exotherm is manageable, and the solvent system allows for efficient heat transfer, ensuring safe operation at larger volumes. From an environmental standpoint, the process generates less hazardous waste compared to traditional methods, facilitating easier compliance with increasingly stringent environmental regulations. The reduction in solvent usage and waste generation aligns with corporate sustainability goals, enhancing the brand reputation of the manufacturer. This combination of scalability and environmental stewardship makes the technology an attractive option for long-term strategic planning and capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral phosphonic amide ester technology. These answers are derived directly from the experimental data and specifications provided in the underlying patent documentation. They serve to clarify the operational parameters and potential benefits for stakeholders evaluating this technology for adoption. Understanding these details is essential for making informed decisions regarding process integration and resource allocation. The responses cover aspects ranging from catalytic performance to raw material sourcing, providing a comprehensive overview of the technology's capabilities.
Q: What is the enantioselectivity performance of the 2c catalyst variant?
A: According to patent data CN102070671A, the 2c variant (derived from L-phenylalaninol) demonstrates exceptional enantioselectivity, achieving up to 98% ee in the asymmetric cyanosilylation of 2-tolyl aldehyde.
Q: Are the starting materials for this catalyst commercially available?
A: Yes, the synthesis relies on readily available bulk chemicals such as 2-cyanophenol, L-leucinol, L-valinol, and phenyl phosphonyl chloride, ensuring a robust and continuous supply chain for manufacturing.
Q: Does this process require exotic or hazardous reaction conditions?
A: The process operates under standard anhydrous and oxygen-free reflux conditions using common solvents like chlorobenzene and toluene, making it highly adaptable for existing industrial reactor setups without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphonic Amide Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in driving innovation within the pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be successfully translated into viable manufacturing processes. We are committed to delivering high-purity chiral phosphonic amide esters that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in process optimization allows us to tailor the synthesis to meet specific client requirements, whether it be maximizing yield, minimizing impurities, or adapting to existing infrastructure. By partnering with us, you gain access to a wealth of technical knowledge and a reliable supply chain that can support your most demanding projects.
We invite you to explore how our solutions can enhance your current manufacturing capabilities and drive down costs without compromising on quality. Our technical procurement team is ready to assist you in evaluating the feasibility of integrating this catalytic system into your workflow. We encourage you to request a Customized Cost-Saving Analysis to understand the specific economic benefits for your operation. Furthermore, we can provide specific COA data and route feasibility assessments to support your internal validation processes. Let us help you navigate the complexities of chiral synthesis and secure a competitive advantage in the global market. Contact us today to discuss your specific needs and discover how NINGBO INNO PHARMCHEM can be your trusted partner in chemical innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
