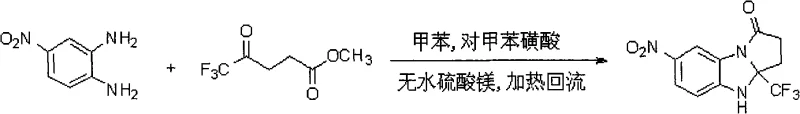
Scalable Synthesis of 7-Nitro-Trifluoromethyl Benzimidazole Pyrrolone for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic intermediates that balance high purity with manufacturing efficiency. Patent CN101514203A introduces a significant advancement in this domain by detailing the synthesis of 7-nitro-3a-(trifluoromethyl)-2,3,3a,4-tetrahydro-1H-benzo[d]pyrrole[1,2-a]imidazol-1-one. This specific fluorinated derivative represents a critical building block for potential antiepileptic and anticonvulsant agents, addressing the growing demand for metabolically stable drug candidates. The disclosed methodology leverages a streamlined one-pot approach that significantly reduces processing time and waste generation compared to traditional multi-step sequences. By integrating the trifluoromethyl moiety directly into the core scaffold during the cyclization event, the process ensures precise structural control while maintaining operational simplicity. For global procurement teams and R&D directors, understanding the nuances of this patented route is essential for securing a reliable supply of high-value pharmaceutical intermediates that meet stringent quality specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for benzimidazole pyrrolone derivatives often suffer from excessive step counts, requiring tedious isolation and purification between each transformation. These conventional methods frequently rely on harsh reaction conditions or expensive transition metal catalysts that introduce heavy metal impurities, necessitating costly downstream removal processes to meet regulatory standards. Furthermore, the introduction of fluorine atoms in legacy routes is often achieved through late-stage functionalization, which can lead to poor regioselectivity and reduced overall yields. The accumulation of solvent waste and the need for specialized equipment to handle hazardous reagents further exacerbate the environmental footprint and production costs. Such inefficiencies create bottlenecks in the supply chain, making it difficult for manufacturers to scale production rapidly in response to market demand without compromising on product consistency or purity profiles.
The Novel Approach
In contrast, the novel approach outlined in the patent utilizes a highly efficient one-pot condensation strategy that merges the formation of the heterocyclic core with the incorporation of the trifluoromethyl group. By employing p-toluenesulfonic acid as a reusable organic catalyst and toluene as a benign solvent, the process avoids the use of toxic heavy metals entirely. The reaction proceeds under standard reflux conditions, allowing for straightforward temperature control and energy management within existing manufacturing infrastructure. This method not only simplifies the operational workflow but also enhances the overall atom economy of the synthesis. The ability to achieve the target molecule in a single reactor vessel minimizes material transfer losses and reduces the risk of contamination, thereby ensuring a cleaner crude product that requires less intensive purification efforts to achieve the desired pharmaceutical grade quality.
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the acid-catalyzed condensation between methyl trifluoro γ-keto acid and p-nitro-o-phenylenediamine. The mechanism initiates with the activation of the carbonyl group by p-toluenesulfonic acid, facilitating nucleophilic attack by the amine functionality of the diamine. Anhydrous magnesium sulfate plays a crucial dual role as both a drying agent to drive the equilibrium forward by removing water and as a mild Lewis acid to stabilize intermediate species. The subsequent cyclization involves an intramolecular nucleophilic attack that closes the pyrrolone ring, locking the trifluoromethyl group at the 3a-position. This specific arrangement is thermodynamically favored under the reflux conditions provided, ensuring high regioselectivity. The stepwise addition of the catalyst allows for controlled reaction kinetics, preventing the formation of polymeric byproducts and ensuring that the reaction proceeds smoothly to the desired imidazolone structure without significant degradation of the sensitive nitro group.
Controlling impurity profiles in fluorinated heterocycles is paramount for pharmaceutical applications, and this mechanism offers inherent advantages in that regard. The use of a non-oxidative acid catalyst prevents unwanted side reactions that could compromise the integrity of the nitro substituent, which is often susceptible to reduction or displacement under harsher conditions. The choice of toluene as a solvent ensures that all reactants and intermediates remain in solution at elevated temperatures, promoting homogeneous reaction conditions that minimize localized hot spots where decomposition could occur. Furthermore, the final purification via silica gel chromatography using a petroleum ether and ethyl acetate system effectively separates the target yellow solid from any unreacted starting materials or minor isomeric byproducts. This rigorous control over the reaction environment results in a product with a defined melting point and consistent spectral data, providing confidence in the batch-to-batch reproducibility required for clinical supply chains.
How to Synthesize 7-Nitro-3a-(trifluoromethyl) Derivative Efficiently
Implementing this synthesis requires careful attention to the stoichiometry and timing of reagent addition to maximize yield and purity. The process begins with the preparation of the reaction mixture containing the keto ester and catalyst, followed by a controlled heating phase to activate the system before introducing the diamine. Maintaining the reflux temperature consistently throughout the extended reaction period is critical to ensure complete conversion of the starting materials. Following the reaction, the workup procedure involves simple filtration to remove the drying agent and concentration of the filtrate, which streamlines the isolation process significantly. Detailed standardized operating procedures regarding specific molar ratios, heating rates, and purification parameters are essential for transferring this technology from the laboratory bench to pilot plant operations successfully.
- Dissolve methyl trifluoro γ-keto acid and catalytic p-toluenesulfonic acid in toluene with anhydrous magnesium sulfate, then reflux for 20-60 minutes.
- Add p-nitro-o-phenylenediamine to the mixture and continue refluxing for 10-15 hours to initiate the cyclization process.
- Introduce a second portion of p-toluenesulfonic acid catalyst and reflux for an additional 10-15 hours before purification to isolate the yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical novelty. The simplification of the manufacturing process directly translates to reduced operational overheads, as fewer unit operations are required to produce the final intermediate. The elimination of transition metal catalysts removes the need for expensive scavenging resins and complex analytical testing for residual metals, which are significant cost drivers in pharmaceutical manufacturing. Additionally, the use of commodity chemicals like toluene and p-toluenesulfonic acid ensures that raw material sourcing is stable and不受 geopolitical fluctuations that often affect specialized reagents. This stability allows for more accurate long-term forecasting and inventory planning, reducing the risk of production stoppages due to material shortages.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis drastically reduces solvent consumption and energy usage compared to multi-step alternatives. By consolidating multiple reaction stages into a single vessel, manufacturers can lower labor costs and decrease the turnaround time for each batch. The absence of expensive noble metal catalysts further contributes to substantial cost savings, making the final intermediate more price-competitive in the global market. These efficiencies allow suppliers to offer more flexible pricing models while maintaining healthy margins, providing a distinct advantage in cost-sensitive therapeutic areas.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials ensures a robust supply chain that is less vulnerable to disruptions. Since the reagents are common industrial chemicals, sourcing can be diversified across multiple vendors to mitigate risk. The simplicity of the process also means that it can be easily replicated across different manufacturing sites, providing redundancy and ensuring continuity of supply even if one facility faces operational challenges. This resilience is crucial for maintaining the uninterrupted production of downstream active pharmaceutical ingredients that depend on this key intermediate.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-gram scales in the patent examples without loss of efficiency. The use of toluene, while requiring proper handling, is a well-understood solvent in the industry with established recovery and recycling protocols that support environmental compliance. The reduction in waste generation aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and corporate sustainability goals. This alignment facilitates smoother regulatory approvals and enhances the brand reputation of companies adopting this greener synthetic pathway.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated intermediate. Understanding these details helps stakeholders make informed decisions about integrating this material into their development pipelines. The answers are derived directly from the technical specifications and experimental data provided in the source patent documentation, ensuring accuracy and relevance for professional users.
Q: What are the key advantages of this one-pot synthesis method?
A: The patented method eliminates complex multi-step isolation procedures, utilizing a single solvent system (toluene) and readily available catalysts to streamline production and reduce operational complexity.
Q: Is this intermediate suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process is explicitly designed for scalability, utilizing standard reflux conditions and common reagents that facilitate easy transition from laboratory to commercial metric-ton production.
Q: How does the trifluoromethyl group impact the biological profile?
A: The introduction of the electron-withdrawing trifluoromethyl group at the 3a-position enhances metabolic stability and lipophilicity, potentially improving absorption and physiological activity compared to non-fluorinated analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Nitro-3a-(trifluoromethyl) Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 7-nitro-3a-(trifluoromethyl)-2,3,3a,4-tetrahydro-1H-benzo[d]pyrrole[1,2-a]imidazol-1-one meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex synthetic challenges efficiently, providing you with a secure and consistent supply of this valuable building block.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can support your commercial goals. By partnering with us, you gain access to a wealth of chemical expertise and a dedication to service that sets us apart as a leader in the fine chemical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
