Efficient Synthesis of 3-Spirocyclohexenone Substituted Chroman Scaffolds for Drug Discovery and Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to construct complex bioactive scaffolds, particularly those combining multiple pharmacophores into a single molecular architecture. Patent CN116003369A introduces a groundbreaking methodology for the synthesis of 3-spirocyclohexenone substituted chroman structures, a privileged scaffold found in numerous natural products and drug candidates. This innovation leverages a unique hydrogen migration strategy to achieve what was previously a challenging multi-step transformation in a single, highly efficient operation. By utilizing a Lewis acid catalyst, specifically Scandium Trifluoromethanesulfonate, the process enables the rapid assembly of these intricate spirocyclic systems under remarkably mild conditions. For R&D directors and procurement specialists, this represents a significant leap forward in accessing high-purity pharmaceutical intermediates with reduced process complexity and enhanced economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of spirocyclic frameworks containing both cyclohexenone and chroman moieties has been fraught with synthetic challenges that hinder commercial scalability. Conventional approaches often require the separate synthesis of the individual ring systems followed by difficult coupling reactions, which inevitably leads to lower overall yields and increased waste generation. These multi-step sequences frequently demand harsh reaction conditions, such as high temperatures or strong bases, which can compromise the integrity of sensitive functional groups present in advanced drug intermediates. Furthermore, the reliance on precious metal catalysts or stoichiometric oxidants in older methodologies introduces significant cost burdens and environmental concerns related to heavy metal removal and disposal. The cumulative effect of these inefficiencies results in prolonged lead times and inflated manufacturing costs, making it difficult for supply chain managers to secure reliable sources of these critical building blocks for drug development pipelines.
The Novel Approach
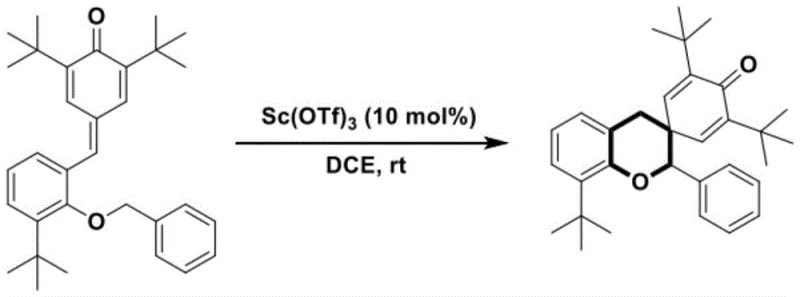
In stark contrast to these legacy methods, the technology disclosed in CN116003369A offers a streamlined, one-step solution that fundamentally reshapes the synthetic landscape for these compounds. The novel approach utilizes readily available p-methylene quinone derivatives as starting materials, which undergo an elegant cascade reaction to form the target spirochroman skeleton directly. This method operates effectively at room temperature or slightly elevated temperatures up to 100°C, significantly reducing energy consumption compared to traditional high-heat processes. The use of Scandium Trifluoromethanesulfonate as a catalyst ensures high conversion rates, with experimental data showing yields reaching up to 94% in optimized examples. This drastic simplification of the synthetic route not only accelerates the timeline from bench to kilogram scale but also minimizes the formation of by-products, thereby simplifying downstream purification and enhancing the overall purity profile of the final active pharmaceutical ingredient intermediates.
Mechanistic Insights into Sc(OTf)3-Catalyzed Hydrogen Migration Cyclization
The core innovation of this synthesis lies in its sophisticated mechanistic pathway, which harnesses the power of intramolecular hydrogen migration to drive the cyclization process. Upon activation by the Scandium Trifluoromethanesulfonate catalyst, the p-methylene quinone derivative undergoes a precise [1,5]-hydrogen shift. This migration generates a reactive zwitterionic intermediate, a transient species that possesses both nucleophilic and electrophilic character simultaneously. This unique electronic configuration facilitates an immediate intramolecular dearomatization-cyclization reaction, effectively stitching together the chroman and cyclohexenone rings in a concerted fashion. Understanding this mechanism is crucial for process chemists, as it highlights the atom-economical nature of the reaction where no atoms are wasted in leaving groups, and the stereochemical outcome is dictated by the rigid transition state of the spiro-center formation.
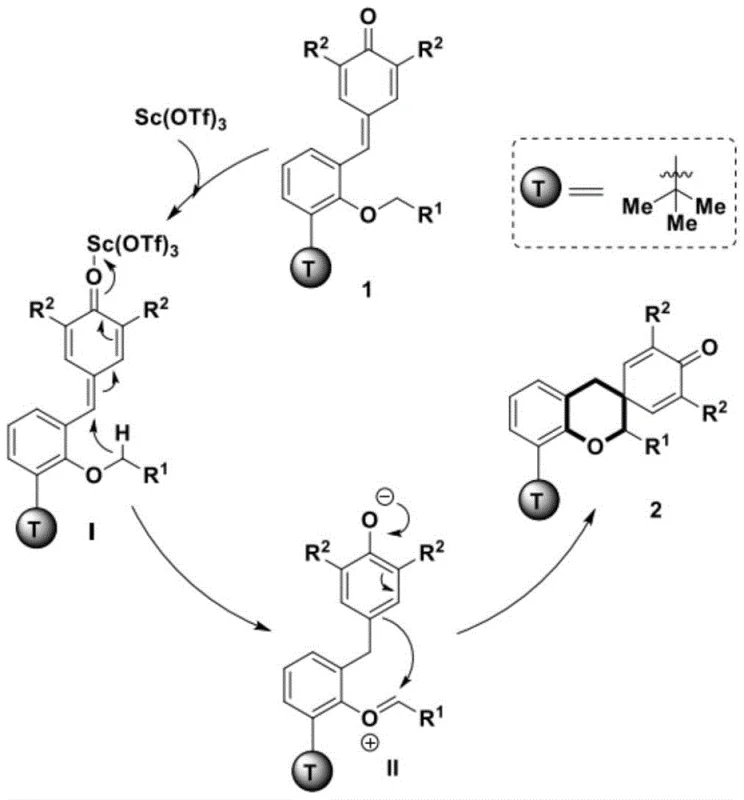
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or oxidative coupling methods. Because the reaction proceeds through a well-defined ionic pathway rather than a chaotic radical chain reaction, the generation of random polymeric by-products or regioisomers is significantly suppressed. The specificity of the [1,5]-hydrogen migration ensures that the reaction occurs at the intended position on the aromatic ring, leading to a cleaner crude reaction mixture. This inherent selectivity reduces the burden on purification teams, allowing for simpler chromatographic separations or even direct crystallization in some cases. For quality control laboratories, this translates to a more consistent impurity profile across different batches, which is a critical requirement for regulatory compliance when manufacturing intermediates intended for human therapeutic use.
How to Synthesize 3-Spirocyclohexenone Substituted Chroman Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to catalyst loading and solvent choice to maximize efficiency. The protocol dictates dissolving the p-methylene quinone derivative in a suitable solvent such as 1,2-dichloroethane, followed by the addition of the scandium catalyst. The reaction is then allowed to proceed with stirring, monitored closely via thin-layer chromatography to ensure complete consumption of the starting material without over-reaction. Once the reaction is deemed complete, standard workup procedures involving silica gel column chromatography are employed to isolate the pure product. The detailed standardized synthesis steps, including specific molar ratios and safety precautions for scaling this procedure, are outlined in the guide below.
- Mix p-methylene quinone derivative with 1,2-dichloroethane solvent and 10 mol% Scandium Trifluoromethanesulfonate catalyst.
- Stir the reaction mixture at room temperature until TLC indicates complete consumption of raw materials.
- Purify the crude product using silica gel column chromatography and rotary evaporation to obtain the target spirochroman compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology presents a compelling value proposition centered around cost stability and operational reliability. By eliminating the need for multiple synthetic steps and expensive reagents, the overall cost of goods sold (COGS) for these intermediates can be drastically reduced. The simplicity of the one-pot reaction means fewer unit operations are required, which lowers capital expenditure on equipment and reduces the manpower needed for production. Furthermore, the use of commercially available and relatively inexpensive starting materials mitigates the risk of supply disruptions often associated with exotic or custom-synthesized precursors. This robustness ensures a steady flow of materials to downstream API manufacturing sites, safeguarding production schedules against raw material shortages.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts like palladium or platinum, which are often subject to volatile market pricing, leads to substantial cost savings. Additionally, the high yield of the reaction minimizes raw material waste, ensuring that every gram of starting material contributes effectively to the final product output. The simplified purification process further reduces the consumption of silica gel and organic solvents, lowering both material costs and waste disposal fees associated with hazardous chemical treatment.
- Enhanced Supply Chain Reliability: Since the starting materials are structurally simple and widely available from bulk chemical suppliers, the risk of supply chain bottlenecks is significantly minimized. The mild reaction conditions allow for production in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. This flexibility enables manufacturers to utilize existing infrastructure, speeding up the technology transfer process and ensuring that production capacity can be ramped up quickly to meet surging demand from pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction's tolerance for ambient pressure and moderate temperatures makes it inherently safer and easier to scale from gram to ton quantities. The reduced generation of hazardous by-products aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes for new manufacturing lines. Moreover, the potential to recover and recycle the solvent and potentially the catalyst adds another layer of sustainability, appealing to eco-conscious stakeholders and reducing the long-term environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spirochroman synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing manufacturing portfolio.
Q: What are the optimal reaction conditions for this spirochroman synthesis?
A: According to patent CN116003369A, the optimal conditions involve using Scandium Trifluoromethanesulfonate (10 mol%) as the catalyst in 1,2-dichloroethane solvent at room temperature, achieving yields up to 94%.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent substrate universality. It tolerates both electron-withdrawing and electron-donating groups, and the position of substituents does not significantly impact the reaction yield.
Q: Is the purification process complex for large-scale production?
A: The purification is straightforward, typically involving standard silica gel column chromatography followed by rotary evaporation, which is highly scalable and avoids complex crystallization or distillation steps often required in multi-step syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Spirocyclohexenone Substituted Chroman Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel synthetic route for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from discovery to market. Our state-of-the-art facilities are equipped to handle the specific requirements of Lewis acid-catalyzed reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug discovery programs with reliable and consistent quality.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific pipeline needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient synthesis can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom targets, and let us help you accelerate your path to clinical success.
