Advanced Iridium-Catalyzed Synthesis of Chiral Beta-Hydroxy Alpha-Amino Acid Derivatives for Pharmaceutical Applications
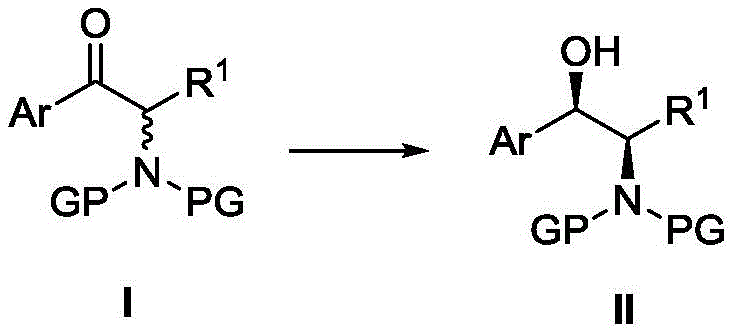
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral scaffolds, particularly those found in critical antibiotics and bioactive molecules. Patent CN115160162A introduces a groundbreaking asymmetric hydrogenation method for alpha-amino beta-ketoesters, addressing long-standing challenges in stereoselective synthesis. This technology leverages a novel iridium-f-amphamidol catalyst system to convert prochiral ketones into high-value chiral amino alcohol intermediates with exceptional precision. The significance of this innovation lies in its ability to produce key precursors for drugs such as Thiamphenicol, Florfenicol, and Droxidopa with superior optical purity compared to traditional transfer hydrogenation techniques. By shifting from atom-inefficient hydrogen donors to direct molecular hydrogen activation, this process represents a paradigm shift towards greener and more economical chemical manufacturing.
For research and development directors evaluating new synthetic routes, the mechanistic elegance of this iridium-catalyzed system offers a compelling alternative to legacy methods. Conventional approaches often rely on asymmetric hydrogen transfer using formic acid or amine mixtures, which suffer from poor atom economy and generate substantial waste streams that complicate downstream purification. Furthermore, existing catalytic systems frequently struggle to maintain high diastereoselectivity across a broad range of aryl substituents, often requiring tedious recrystallization steps to achieve pharmaceutical-grade purity. In contrast, the novel approach described in the patent utilizes a specialized ligand architecture that enforces strict stereocontrol through non-covalent interactions during the hydride transfer step. This results in a streamlined workflow where the crude reaction mixture already possesses the requisite optical purity, drastically reducing the need for extensive chromatographic separation and solvent consumption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-hydroxy alpha-amino acid derivatives has been plagued by inefficiencies inherent to transfer hydrogenation protocols. These methods typically necessitate the use of stoichiometric amounts of hydrogen donors, leading to low atom utilization rates that are economically and environmentally unsustainable for large-scale production. Additionally, the reaction conditions often require elevated temperatures or prolonged reaction times to drive conversion, which can inadvertently promote racemization or decomposition of sensitive functional groups present in complex drug intermediates. The reliance on specific protecting groups to induce stereoselectivity in older catalytic systems further limits substrate scope, forcing chemists to adopt multi-step protection and deprotection sequences that inflate overall production costs. Consequently, manufacturers face significant bottlenecks in scaling these processes while maintaining the rigorous quality standards demanded by global regulatory agencies.
The Novel Approach
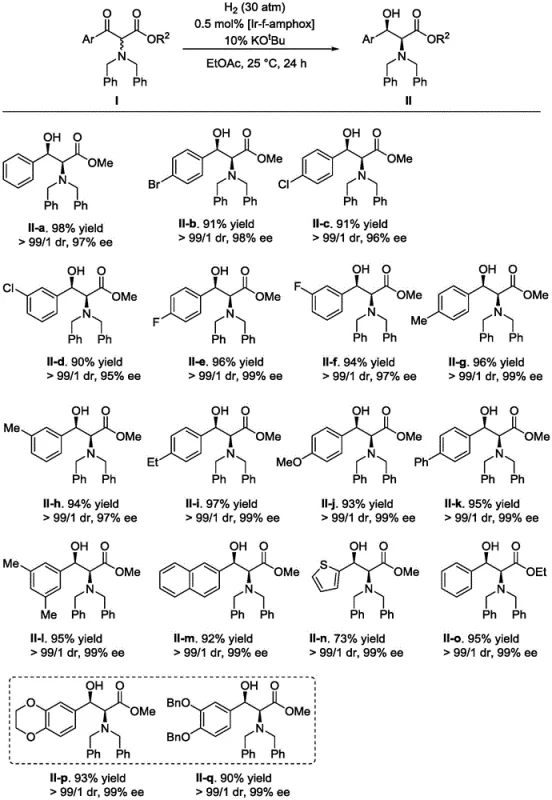
The methodology disclosed in CN115160162A overcomes these hurdles by employing a highly active iridium complex capable of activating molecular hydrogen under mild conditions. This direct hydrogenation strategy eliminates the need for external hydrogen donors, thereby simplifying the reaction mixture and improving the overall mass balance of the process. The catalyst system demonstrates remarkable tolerance to diverse electronic and steric environments on the aromatic ring, enabling the synthesis of a wide array of derivatives without compromising yield or selectivity. Operational simplicity is another key advantage, as the reaction proceeds efficiently at room temperature and moderate hydrogen pressures, reducing energy consumption and safety risks associated with high-pressure operations. This robust performance profile makes the technology an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their manufacturing portfolios.
Mechanistic Insights into Ir-f-amphamidol Catalyzed Asymmetric Hydrogenation
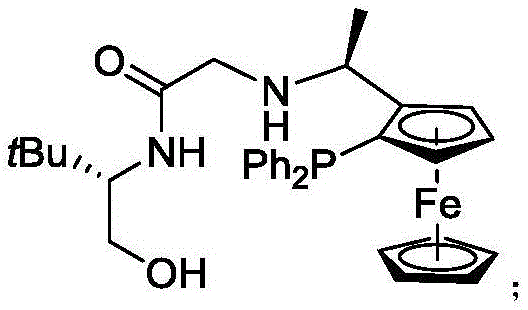
At the heart of this technological advancement is the unique structure of the f-amphamidol ligand, which coordinates with the iridium center to create a chiral pocket essential for enantiofacial discrimination. The ligand features a ferrocene backbone coupled with specific amine and phosphine functionalities that rigidify the catalyst geometry, ensuring that hydride delivery occurs exclusively from one face of the ketone substrate. This precise spatial arrangement is critical for achieving the observed syn-selectivity, where the newly formed hydroxyl and amino groups adopt a specific relative configuration dictated by the nitrogen protecting group. Understanding this mechanism allows process chemists to predict outcomes for novel substrates and tailor reaction parameters to maximize efficiency. The interplay between the metal center and the chiral ligand creates a dynamic environment that facilitates rapid turnover while suppressing competing non-selective pathways.
Impurity control is inherently built into the catalytic cycle due to the high specificity of the iridium-f-amphamidol complex. Unlike less selective catalysts that may reduce other functional groups or promote side reactions such as dehydration, this system targets the ketone moiety with surgical precision. The use of mild bases like potassium tert-butoxide further stabilizes the active catalytic species without inducing epimerization at the alpha-carbon. This level of control ensures that the final product stream contains minimal levels of diastereomeric impurities, which is a crucial metric for regulatory approval of active pharmaceutical ingredients. By minimizing the formation of hard-to-remove byproducts, the process significantly lowers the burden on purification units, resulting in a cleaner and more sustainable manufacturing operation that aligns with modern green chemistry principles.
How to Synthesize Chiral Beta-Hydroxy Alpha-Amino Acid Derivatives Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction conditions to fully realize its potential benefits. The process begins with the in situ generation of the active iridium species by mixing the metal precursor with the chiral ligand in an alcoholic solvent, followed by the addition of the substrate and base. Maintaining an inert atmosphere during the setup prevents catalyst deactivation, while the subsequent introduction of hydrogen gas drives the reduction to completion. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during scale-up operations.
- Prepare the chiral catalyst by mixing the metal iridium precursor [Ir(COD)Cl]2 with 1.2 equivalents of the f-amphamidol ligand in isopropanol and stirring for 10 hours.
- In a hydrogenation vessel under inert atmosphere, combine the alpha-amino beta-ketoester substrate, the prepared iridium catalyst (0.00005-0.01 molar equivalent), and a base such as potassium tert-butoxide in ethyl acetate.
- Pressurize the reactor with hydrogen gas to 30-50 atm and maintain the reaction at 25°C for 24 hours, followed by solvent removal and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial opportunities for cost reduction in API manufacturing by streamlining the supply chain for critical chiral building blocks. The elimination of expensive stoichiometric reductants and the ability to use ultra-low catalyst loadings directly translate to lower raw material costs per kilogram of finished product. Furthermore, the high selectivity of the reaction reduces the volume of solvents and adsorbents required for purification, leading to significant savings in waste disposal and environmental compliance fees. These efficiencies make the process economically attractive for producing high-volume commodities where margin compression is a constant challenge for suppliers.
- Cost Reduction in Manufacturing: The shift to direct hydrogenation removes the recurring expense of purchasing and handling bulk quantities of formic acid or amine donors, which are both costly and hazardous. Additionally, the catalyst's high turnover number means that precious metal consumption is minimized, preserving capital and reducing exposure to volatile metal markets. The simplified workup procedure also decreases labor hours and utility usage, contributing to a leaner overall cost structure that enhances competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials and standard hydrogenation equipment, manufacturers can mitigate risks associated with specialized reagent shortages. The robustness of the catalyst system ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or out-of-specification results. This reliability is essential for maintaining continuous supply lines to downstream drug manufacturers who depend on just-in-time delivery models to manage their inventory levels effectively.
- Scalability and Environmental Compliance: The mild reaction conditions facilitate easy scale-up from laboratory to commercial production without the need for exotic high-pressure reactors or cryogenic cooling systems. The reduced generation of chemical waste aligns with increasingly stringent environmental regulations, lowering the risk of fines and permitting issues. This sustainability profile not only improves corporate social responsibility metrics but also future-proofs the manufacturing process against evolving regulatory landscapes regarding chemical emissions and resource utilization.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and embodiments described in the patent documentation to provide accurate guidance for potential adopters.
Q: What is the stereoselectivity outcome of this hydrogenation method?
A: The method utilizing the Ir-f-amphamidol complex consistently yields products with syn-structure stereochemistry, achieving diastereomeric ratios greater than 99:1 dr and enantiomeric excesses exceeding 99% ee for various substrates.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes direct hydrogen gas rather than stoichiometric hydrogen donors, operates at mild temperatures (20-50°C), and uses extremely low catalyst loadings, making it highly suitable for large-scale commercial manufacturing.
Q: What types of protecting groups are compatible with this reaction?
A: The reaction tolerates a wide range of nitrogen protecting groups including benzyl (Bn), tert-butoxycarbonyl, allyloxycarbonyl, and alkoxycarbonyl groups, allowing for flexible downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha Amino Beta Ketoester Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced catalytic technologies to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral intermediate meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence allows us to navigate complex synthetic challenges with confidence and precision.
We invite you to engage with our technical procurement team to discuss how this innovative hydrogenation method can be integrated into your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your strategic sourcing decisions and drive your projects forward with certainty.
