Advanced Chiral Tridentate Nitrogen Phosphine Ligands for Industrial Asymmetric Hydrogenation
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access chiral building blocks, particularly chiral secondary alcohols which serve as critical intermediates for blockbuster drugs like Duloxetine and Mirabegron. Patent CN114644663A introduces a groundbreaking class of chiral tridentate nitrogen phosphine ligands that address the longstanding challenges of cost, stability, and selectivity in asymmetric catalysis. These novel ligands, characterized by a robust ferrocene backbone and tunable steric properties, enable the highly enantioselective hydrogenation of diverse ketone substrates under mild conditions. By leveraging cheap and readily available amino acid-derived chiral modules, this technology offers a sustainable and economically viable solution for the commercial scale-up of complex pharmaceutical intermediates. As a leading entity in the sector, understanding the nuances of this innovation is crucial for maintaining a competitive edge in high-purity API intermediate production.
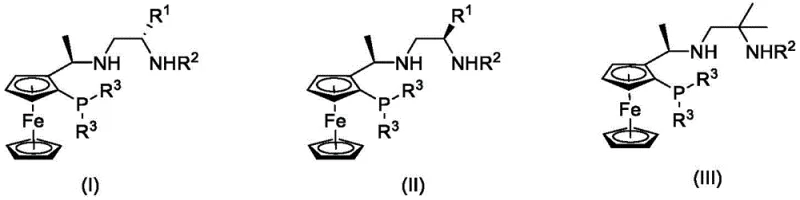
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of ketones has relied heavily on sophisticated ligand systems such as the spiro-chiral ligands reported by Zhou Qilin's group, which, while highly active, often involve complex multi-step syntheses that drive up manufacturing costs and limit scalability. Earlier generations of ferrocene-based PNN ligands, such as those designed by Zhang Shengyong's group, demonstrated good catalytic effects but were frequently hampered by moderate enantioselectivity, with ee values capping at around 86% for simple aromatic ketones. Furthermore, many traditional catalysts require harsh reaction conditions or expensive transition metals in high loadings, creating significant bottlenecks in cost reduction in fine chemical manufacturing. The instability of certain ligand frameworks under prolonged reaction times or elevated temperatures also poses risks for supply chain continuity, necessitating frequent catalyst replenishment and complicating process validation for regulatory compliance.
The Novel Approach
The technology disclosed in CN114644663A represents a paradigm shift by introducing a series of tridentate nitrogen-nitrogen-phosphorus (N-N-P) ligands built on a stable ferrocene scaffold. Unlike previous iterations, these ligands feature a modular design where the chiral information is derived from inexpensive amino acids, allowing for precise tuning of steric hindrance and electronic properties to match specific substrate requirements. This structural flexibility enables the catalyst to achieve unprecedented levels of activity and selectivity across a wide range of ketone classes, from simple acetophenones to complex heteroaromatic and functionalized ketones. The synthetic route for these ligands is notably streamlined, avoiding the cumbersome steps associated with spiro-systems, thereby facilitating easier access for reliable pharmaceutical intermediate suppliers looking to optimize their raw material sourcing and production workflows.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the formation of a highly active iridium complex when the tridentate ligand coordinates with precursors like [Ir(COD)Cl]2. The tridentate nature of the ligand ensures a rigid and well-defined coordination geometry around the metal center, which is essential for effective chirality transfer during the hydrogenation cycle. The ferrocene unit not only provides structural stability but also influences the electronic environment of the phosphorus atom, enhancing the catalyst's ability to activate molecular hydrogen and facilitate hydride transfer to the ketone substrate. This robust coordination prevents ligand dissociation under reaction conditions, a common failure mode in bidentate systems, thus ensuring consistent performance over extended reaction times and supporting the reducing lead time for high-purity pharmaceutical intermediates by minimizing batch-to-batch variability.
Furthermore, the mechanism allows for exceptional tolerance towards various functional groups, which is critical for late-stage functionalization in drug synthesis. The patent data demonstrates that the catalyst system operates effectively with substrates containing halogens, esters, amines, and hydroxyl groups without compromising enantioselectivity. This broad compatibility is achieved through the careful balance of steric bulk provided by the R1 and R3 groups on the ligand framework, which shields the active site from non-productive interactions while allowing the substrate to approach in the optimal orientation. Such mechanistic precision translates directly to higher yields and purity profiles, reducing the burden on downstream purification processes and aligning with the stringent quality standards required for high-purity OLED material and pharmaceutical applications.
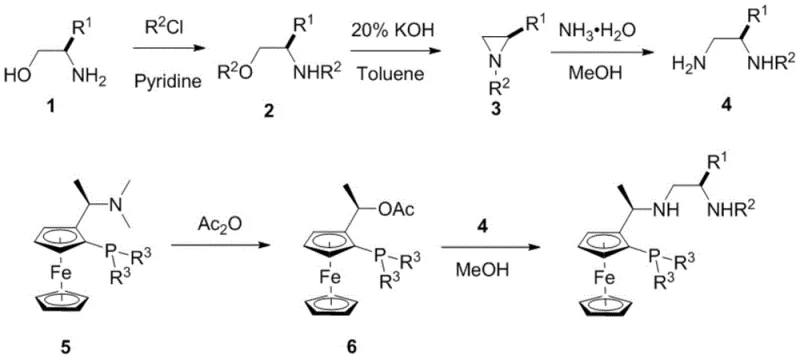
How to Synthesize Chiral Tridentate Nitrogen Phosphine Ligands Efficiently
The synthesis of these high-performance ligands follows a logical and scalable pathway that begins with readily available chiral amino alcohols. The process involves initial protection and activation steps to generate reactive aziridine intermediates, which are subsequently opened to form diamine precursors. These precursors are then coupled with ferrocene-phosphine derivatives that have been pre-functionalized via lithiation and acetylation. This convergent strategy allows for the independent optimization of the chiral amine component and the ferrocene-phosphine component, providing significant flexibility in library generation. For detailed operational parameters and specific stoichiometric ratios required to replicate these results in a GMP environment, please refer to the standardized synthesis protocol outlined below.
- React amino alcohol derivatives with TsCl and pyridine to form tosylated intermediates, followed by cyclization with KOH to generate aziridine structures.
- Perform ring-opening of the aziridine with ammonia water to obtain diamine precursors suitable for ligand assembly.
- Couple the diamine precursors with acetylated ferrocene-phosphine intermediates in methanol under reflux to yield the final tridentate ligands.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ligand technology offers tangible strategic benefits beyond mere technical performance. The reliance on cheap and easily available amino acids as chiral sources fundamentally alters the cost structure of catalyst production, removing dependence on scarce or exotic natural products. This accessibility ensures a stable supply of raw materials, mitigating the risk of shortages that can disrupt production schedules. Additionally, the mild reaction conditions specified in the patent, ranging from room temperature to 100°C and moderate hydrogen pressures, reduce the energy consumption and safety hazards associated with high-pressure hydrogenation processes, contributing to a safer and more sustainable manufacturing footprint.
- Cost Reduction in Manufacturing: The most significant economic driver is the remarkably low catalyst loading enabled by this system. The patent explicitly states that catalyst consumption can be reduced to 1/10,000 of the substrate molar ratio, which drastically lowers the cost per kilogram of the final chiral alcohol product. This high turnover number means that less expensive iridium metal is required per batch, and the simplified post-reaction workup reduces solvent usage and waste disposal costs. By eliminating the need for expensive metal scavengers often required with less stable catalysts, manufacturers can achieve substantial cost savings while maintaining high product purity standards essential for regulatory approval.
- Enhanced Supply Chain Reliability: The structural stability of the ferrocene-based ligands ensures a longer shelf life and consistent performance upon delivery, reducing the risk of degraded reagents causing batch failures. Since the synthesis does not rely on complex, multi-step sequences typical of spiro-ligands, the lead time for producing the ligand itself is significantly shortened, allowing suppliers to respond more rapidly to fluctuating market demands. This reliability is crucial for maintaining continuous production lines for critical medicines, ensuring that downstream customers receive their pharmaceutical intermediates on schedule without unexpected delays caused by catalyst supply bottlenecks.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing common solvents like isopropanol and methanol which are easier to recover and recycle compared to chlorinated solvents. The high selectivity of the reaction minimizes the formation of unwanted diastereomers and byproducts, simplifying the purification process and reducing the volume of chemical waste generated. This efficiency supports easier scale-up from laboratory to commercial production volumes, as the reaction kinetics remain favorable even at larger scales, facilitating the commercial scale-up of complex polymer additives and fine chemicals with minimal environmental impact.

Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing processes, we have compiled answers to common inquiries regarding the operational parameters and scope of the ligand system. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development scientists. Understanding these details is key to making informed decisions about catalyst selection and process optimization strategies.
Q: What is the catalyst loading efficiency for these ligands?
A: According to patent CN114644663A, the catalyst consumption can be reduced to as low as 1/10,000 molar ratio relative to the substrate, demonstrating exceptionally high turnover numbers.
Q: Are these ligands stable under industrial conditions?
A: Yes, the ferrocene-based backbone provides excellent thermal and chemical stability, allowing for robust performance during scale-up and storage compared to more fragile spiro-ligand systems.
Q: What types of ketones can be hydrogenated using this technology?
A: The technology supports a broad substrate scope including aromatic ketones, heteroaromatic ketones, hydroxyketones, halogenated ketones, ketoesters, enones, and aminoketones with high enantioselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tridentate Nitrogen Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral tridentate nitrogen phosphine ligands described in CN114644663A for the next generation of asymmetric synthesis. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or chiral alcohol intermediate meets the highest industry standards for enantiomeric excess and chemical purity.
We invite you to collaborate with our technical procurement team to explore how this advanced catalytic technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to move forward with confidence in developing cost-effective and sustainable manufacturing processes for your valuable chiral compounds.
