Scalable Silver-Catalyzed Synthesis of Beta-Amino Phosphonic Acid Derivatives for Pharma
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies for constructing nitrogen-containing organophosphorus scaffolds, which serve as critical motifs in enzyme inhibitors and antiviral agents. Patent CN110950906A introduces a groundbreaking preparation method for beta-amino phosphoric acid derivatives that fundamentally shifts the paradigm from complex, multi-step sequences to a direct, catalytic functionalization of olefins. This technology leverages a silver-catalyzed radical cascade involving tert-butyl nitrite and phosphorus reagents to generate beta-hydroxyiminophosphono intermediates with remarkable efficiency. For R&D directors and process chemists, this represents a significant opportunity to access diverse chemical space using commodity feedstocks rather than specialized, expensive precursors. The invention not only streamlines the synthesis of mono-phosphonates but also provides a versatile platform for generating bis-phosphonates and free radical compounds through subsequent transformations, thereby addressing the growing demand for high-purity pharmaceutical intermediates in a cost-effective manner.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-amino phosphate derivatives has been plagued by significant operational hazards and synthetic inefficiencies that hinder large-scale adoption. Traditional routes, such as those disclosed by D.M. Mizrahi and A. Obojska, often rely on the use of thionyl chloride, a highly corrosive reagent that necessitates specialized equipment and rigorous safety protocols to manage toxic gas evolution. Furthermore, these legacy processes typically involve multiple reaction steps, requiring the isolation of unstable intermediates and leading to cumulative yield losses that drastically increase the cost of goods sold. The harsh reaction conditions associated with these methods, often involving extreme temperatures or strong acids, can also compromise the integrity of sensitive functional groups on the substrate, limiting the scope of applicable starting materials. Consequently, procurement managers face challenges in securing reliable supply chains for these intermediates due to the limited number of manufacturers capable of safely executing these hazardous transformations.
The Novel Approach
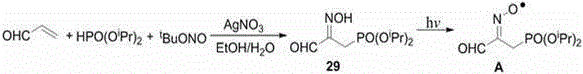
In stark contrast, the methodology described in CN110950906A utilizes a mild, silver-catalyzed radical addition strategy that operates effectively at temperatures ranging from 10°C to 50°C. By employing readily available olefins as initiators and tert-butyl nitrite as a nitrosating agent, this novel approach eliminates the need for corrosive chlorinating agents entirely. The reaction proceeds through a streamlined one-pot sequence to generate beta-hydroxyiminophosphono derivatives, which can then be further elaborated into the target amino phosphonic acids. This shift not only simplifies the post-treatment process, often requiring only standard column chromatography, but also significantly reduces the environmental footprint of the manufacturing process. The ability to utilize a wide variety of olefins, including styrenes, vinyl pyridines, and acrylates, underscores the versatility of this platform for cost reduction in pharmaceutical intermediate manufacturing.

Mechanistic Insights into Silver-Catalyzed Radical Functionalization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the silver catalyst, which promotes the homolytic cleavage of tert-butyl nitrite to generate reactive nitrogen-centered radicals. These radicals interact with the phosphorus reagent, likely forming a phosphonyl radical species that adds regioselectively to the electron-deficient or activated double bond of the olefin substrate. This radical addition step is crucial for establishing the carbon-phosphorus bond with high fidelity, avoiding the rearrangement issues common in ionic pathways. Following the radical addition, the trapping of the resulting carbon-centered radical by the nitroso species leads to the formation of the oxime functionality, yielding the beta-hydroxyiminophosphono scaffold. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as catalyst loading and solvent choice, to maximize conversion and minimize the formation of non-phosphorylated byproducts.
Furthermore, the patent details the subsequent transformation of these oxime intermediates into valuable bis-phosphonates and amino derivatives through controlled reduction and hydrolysis steps. For instance, the conversion of the initial oxime product into a beta-amino hydroxyl phosphono derivative involves nickel-catalyzed reduction under alkaline conditions, followed by acid hydrolysis to reveal the free amine. This multi-stage capability demonstrates the robustness of the initial radical adduct, which serves as a stable linchpin for diverse downstream modifications. The impurity profile is inherently managed by the mildness of the initial radical step, which avoids the aggressive conditions that typically generate complex degradation products. This level of control is essential for producing high-purity OLED material precursors or active pharmaceutical ingredients where trace impurities can have detrimental effects on biological activity or device performance.
How to Synthesize Beta-Amino Phosphonic Acid Derivatives Efficiently
The practical implementation of this synthesis route offers a straightforward protocol that can be adapted for both laboratory discovery and pilot-scale production. The process begins with the dissolution of the chosen olefin, phosphorus reagent, and silver catalyst in a polar solvent system, followed by the controlled addition of tert-butyl nitrite. Reaction monitoring is efficiently achieved using Thin Layer Chromatography (TLC), allowing for precise determination of endpoint to prevent over-reaction. The detailed standardized synthesis steps below outline the specific molar ratios and workup procedures required to achieve the high yields reported in the patent examples, ensuring reproducibility for technical teams evaluating this technology for commercial adoption.
- Dissolve the selected olefin substrate, phosphorus reagent (such as diethyl phosphite), tert-butyl nitrite, and a silver catalyst (e.g., AgNO3) in a suitable solvent like ethanol or acetonitrile.
- Maintain the reaction mixture at a mild temperature range between 10°C and 50°C, monitoring progress via Thin Layer Chromatography (TLC) until the starting materials are fully consumed.
- Upon completion, isolate the crude beta-hydroxyiminophosphono derivative through standard workup procedures, followed by purification via column chromatography to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this silver-catalyzed olefin functionalization strategy offers profound advantages in terms of raw material security and operational expenditure. The reliance on commodity olefins and standard phosphites means that procurement teams are not dependent on single-source suppliers for exotic starting materials, thereby mitigating the risk of supply disruptions. Additionally, the elimination of hazardous reagents like thionyl chloride removes the need for expensive scrubbing systems and specialized corrosion-resistant reactors, leading to substantial cost savings in capital expenditure and maintenance. The mild reaction temperatures further contribute to energy efficiency, reducing the heating and cooling loads required for large-scale batch processing compared to traditional high-temperature methods.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis directly translates to lower manufacturing costs by reducing the number of unit operations and eliminating the need for expensive hazard mitigation infrastructure. By avoiding corrosive reagents, the process extends the lifespan of standard stainless steel equipment and reduces the frequency of maintenance shutdowns. Furthermore, the high yields observed across a broad substrate scope mean that less raw material is wasted, optimizing the atom economy of the process and lowering the effective cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The versatility of the olefin substrate scope ensures that supply chains remain resilient even if specific feedstocks face temporary shortages. Since the chemistry tolerates a wide range of functional groups, including esters, halides, and heterocycles, manufacturers can switch between different olefin precursors without revalidating the entire process. This flexibility allows for rapid response to market demands and ensures continuous production of critical intermediates for the pharmaceutical and agrochemical sectors without lengthy lead times.
- Scalability and Environmental Compliance: The benign nature of the reagents and the aqueous-compatible workup procedures make this process highly scalable and environmentally compliant. The reduction in toxic waste generation simplifies effluent treatment and aligns with increasingly stringent global environmental regulations. This ease of scale-up facilitates the transition from gram-scale laboratory synthesis to multi-ton commercial production, ensuring that the supply of these valuable phosphonic acid derivatives can meet the growing demands of the life sciences industry without compromising on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction conditions and substrate compatibility. Understanding these nuances is critical for process development teams aiming to integrate this technology into their existing manufacturing portfolios.
Q: What is the primary advantage of this silver-catalyzed method over traditional synthesis?
A: Unlike conventional methods that require corrosive reagents like thionyl chloride and harsh conditions, this novel approach utilizes readily available olefins and operates under mild temperatures (10-50°C), significantly simplifying safety protocols and waste management.
Q: What types of olefin substrates are compatible with this reaction system?
A: The process demonstrates exceptional substrate scope, successfully accommodating aryl olefins such as styrenes, heteroaryl olefins like vinylpyridines, and alkyl olefins including acrylates and allyl benzene, making it versatile for diverse intermediate synthesis.
Q: How does this method impact the purity profile of the final amino phosphonic acid?
A: The mild reaction conditions minimize thermal degradation and side reactions, while the specific radical mechanism ensures high regioselectivity, resulting in a cleaner crude product that requires less intensive purification to meet stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Phosphonic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this silver-catalyzed methodology for the production of high-value nitrogen-phosphorus scaffolds. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for clinical and commercial applications. We are committed to delivering consistent quality and reliability for your most critical projects.
We invite you to engage with our technical procurement team to discuss how this innovative route can optimize your supply chain and reduce overall production costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your target molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your long-term strategic goals in the development of next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
