Advanced Silver-Catalyzed Synthesis of Beta-Amino Phosphoric Acid Derivatives for Commercial Scale
The chemical landscape for synthesizing nitrogen-containing organophosphorus compounds has evolved significantly with the disclosure of patent CN110950906B, which introduces a robust and versatile preparation method for beta-amino phosphoric acid derivatives. This technology leverages a silver-catalyzed radical reaction system that utilizes readily available olefins as starting materials, fundamentally shifting the paradigm from complex, hazardous traditional routes to a more streamlined and industrially viable process. The invention demonstrates exceptional adaptability, accommodating a wide variety of olefin substrates including styrenes, alkyl olefins, and heterocyclic variants, thereby expanding the accessible chemical space for drug discovery and agrochemical development. By operating under mild reaction conditions typically ranging from 10°C to 50°C, this method significantly reduces energy consumption and thermal stress on sensitive functional groups, ensuring high fidelity in the molecular architecture of the target compounds. Furthermore, the use of tert-butyl nitrite as a nitrogen source in conjunction with silver catalysts provides a controlled radical generation mechanism that enhances reaction selectivity and overall yield, making it a compelling choice for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-aminophosphonic acid derivatives has been plagued by significant operational challenges and safety concerns that hinder efficient commercial manufacturing. Traditional routes often rely on the use of highly corrosive reagents such as thionyl chloride, which necessitates specialized corrosion-resistant equipment and rigorous safety protocols to manage hazardous gas emissions and potential equipment failure. These conventional methods frequently involve multi-step sequences with harsh reaction conditions, including extreme temperatures and pressures, which can lead to the degradation of sensitive substrates and the formation of complex impurity profiles that are difficult to separate. The reliance on difficult-to-obtain raw materials in older synthetic strategies further exacerbates supply chain vulnerabilities, leading to inconsistent availability and fluctuating costs that disrupt production schedules. Additionally, the post-treatment processes associated with these legacy methods are often labor-intensive, requiring extensive neutralization and purification steps to remove toxic byproducts, thereby increasing the overall environmental footprint and operational expenditure of the manufacturing process.
The Novel Approach
In stark contrast to these legacy challenges, the novel approach disclosed in the patent utilizes a direct oxidative phosphonylation strategy that simplifies the synthetic route while enhancing safety and efficiency. By employing common olefins and phosphorus reagents in the presence of a silver catalyst and tert-butyl nitrite, the reaction proceeds smoothly under mild conditions, eliminating the need for corrosive chlorinating agents and reducing the risk of hazardous incidents. This methodology not only streamlines the operation by reducing the number of synthetic steps but also improves the atom economy of the process, ensuring that a higher proportion of raw materials are converted into the desired target product. The versatility of this system allows for the facile modification of the olefin substrate, enabling the rapid generation of diverse derivative libraries for structure-activity relationship studies without the need for extensive process re-optimization. Consequently, this approach offers a sustainable and economically attractive pathway for the production of complex organophosphorus compounds, aligning with modern green chemistry principles and industrial safety standards.

Mechanistic Insights into Silver-Catalyzed Oximino-Phosphonylation
The core of this technological advancement lies in the intricate radical mechanism facilitated by the silver catalyst, which orchestrates the addition of the phosphorus species across the olefin double bond with high regioselectivity. The silver salt, such as silver nitrate or silver carbonate, acts as a single-electron transfer mediator that activates the tert-butyl nitrite to generate nitrogen-centered radicals, which subsequently abstract hydrogen or initiate the addition sequence on the olefin substrate. This radical cascade is carefully balanced to prevent polymerization or over-oxidation of the olefin, ensuring that the phosphonyl group is installed precisely at the beta-position relative to the emerging nitrogen functionality. The mild thermal conditions employed in this reaction are critical for maintaining the stability of the radical intermediates, allowing the reaction to proceed with controlled kinetics that favor the formation of the beta-hydroxyimino phosphono derivative over competing side products. Understanding this mechanistic pathway is essential for process chemists to fine-tune reaction parameters such as catalyst loading and solvent polarity to maximize yield and minimize the formation of regioisomers or oligomeric byproducts.
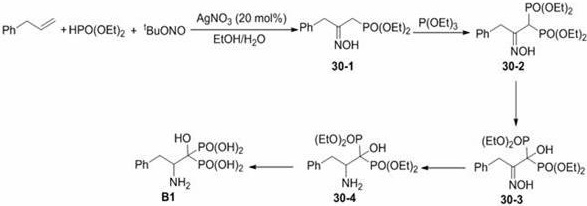
Beyond the initial phosphonylation, the patent outlines a sophisticated downstream transformation sequence that converts the hydroxyimino intermediates into valuable beta-amino phosphoric acid derivatives through reduction and hydrolysis steps. The impurity control mechanism is inherently built into the high selectivity of the initial silver-catalyzed step, which reduces the burden on downstream purification units and ensures a cleaner profile for the final active pharmaceutical ingredient. The subsequent reduction of the oxime group to the amine is typically achieved using nickel catalysis or borohydride reagents under alkaline conditions, which are well-established and scalable processes in the fine chemical industry. This modular approach allows manufacturers to isolate the stable hydroxyimino intermediate if needed or proceed directly to the amino acid analogue, providing flexibility in supply chain management and inventory control. The ability to control the stereochemistry and purity at each stage of this multi-step sequence is paramount for meeting the rigorous regulatory requirements of the pharmaceutical sector, ensuring that the final product is safe and effective for its intended therapeutic application.
How to Synthesize Beta-Amino Phosphoric Acid Derivatives Efficiently
Implementing this synthesis route in a production environment requires a clear understanding of the operational parameters and safety considerations associated with handling radical initiators and silver catalysts. The process begins with the precise weighing and dissolution of the olefin substrate and phosphorus reagent in a selected solvent, followed by the controlled addition of the tert-butyl nitrite and silver catalyst under an inert atmosphere to prevent unwanted oxidation. Reaction monitoring is typically conducted using thin-layer chromatography or high-performance liquid chromatography to determine the exact endpoint, ensuring that the reaction is quenched before any significant decomposition of the product occurs. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-yielding process with consistency and safety.
- Dissolve olefin, phosphorus reagent, tert-butyl nitrite, and silver catalyst in a suitable solvent such as methanol or acetonitrile.
- Maintain the reaction mixture at a mild temperature range between 10°C and 50°C while monitoring progress via TLC.
- Upon completion, isolate the crude product and purify using column chromatography to obtain high-purity beta-hydroxyiminophosphono derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial strategic advantages for procurement managers and supply chain directors looking to optimize their sourcing strategies for phosphorus-containing intermediates. The shift away from hazardous reagents like thionyl chloride not only reduces the cost associated with specialized waste disposal and safety compliance but also mitigates the risk of supply disruptions caused by regulatory restrictions on dangerous chemicals. By utilizing commodity olefins as starting materials, manufacturers can leverage existing global supply chains to secure raw materials at stable prices, insulating the production cost from the volatility often seen with specialized synthetic building blocks. The simplified post-treatment process further contributes to cost efficiency by reducing the consumption of solvents and purification media, leading to a lower overall cost of goods sold and improved margin potential for the final product. These factors combine to create a resilient supply chain model that is capable of scaling rapidly to meet market demand without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of expensive and corrosive reagents significantly lowers the direct material costs associated with the synthesis, while the mild reaction conditions reduce energy consumption for heating and cooling systems. The high yield of the target product minimizes raw material waste, ensuring that a greater proportion of the input cost is converted into saleable product value. Furthermore, the reduced need for complex purification steps lowers the operational expenditure related to chromatography media and solvent recovery, contributing to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: Sourcing readily available olefins and common phosphorus reagents ensures a stable and continuous supply of raw materials, reducing the risk of production stoppages due to material shortages. The robustness of the reaction conditions allows for manufacturing in a wider range of facilities without the need for specialized corrosion-resistant infrastructure, increasing the flexibility of the supply network. This reliability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical customers, fostering long-term partnerships and trust in the supplier's ability to meet critical project timelines.
- Scalability and Environmental Compliance: The mild nature of the reaction chemistry facilitates easy scale-up from laboratory to commercial production volumes without the need for extensive re-engineering of the process equipment. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the compliance burden and potential liability associated with chemical manufacturing. This environmental stewardship enhances the corporate reputation of the manufacturer and opens up opportunities in markets that prioritize sustainable and green chemical sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the technical specifications and experimental data disclosed in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these aspects is critical for assessing the feasibility of integrating this route into existing production portfolios and for communicating value to internal and external partners.
Q: What are the primary advantages of this silver-catalyzed method over traditional synthesis?
A: This method eliminates the need for corrosive reagents like thionyl chloride and operates under significantly milder conditions, reducing safety hazards and equipment corrosion while simplifying post-treatment processes.
Q: Is this process suitable for large-scale industrial production of pharmaceutical intermediates?
A: Yes, the use of readily available olefin starting materials, common solvents, and mild temperature requirements makes this pathway highly adaptable for commercial scale-up and continuous manufacturing environments.
Q: How does this synthesis route impact the impurity profile of the final product?
A: The high selectivity of the silver-catalyzed radical reaction minimizes side reactions, resulting in a cleaner crude product that requires less intensive purification to meet stringent pharmaceutical purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Phosphoric Acid Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical innovation, leveraging advanced technologies like the silver-catalyzed synthesis route to deliver high-quality pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of large-scale drug manufacturing projects with precision and reliability. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-amino phosphoric acid derivatives meets the highest industry standards for safety and efficacy. Our commitment to technical excellence allows us to navigate complex synthetic challenges, providing our partners with a secure and dependable source for critical building blocks in their drug development pipelines.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this efficient synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating the tangible economic benefits of switching to this greener and more economical route. Please contact us to request specific COA data and route feasibility assessments, and let us show you how our expertise can accelerate your project timelines and enhance your competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
