Scalable Aqueous Synthesis of Phosphorylated Geminal Difluorodienes for Advanced Pharma Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to incorporate fluorine-containing motifs into complex molecular architectures, driven by the unique metabolic stability and lipophilicity these groups impart. Patent CN115010753A discloses a groundbreaking approach for the preparation of phosphorylated geminal difluorodiene compounds directly in an aqueous phase, representing a significant leap forward in sustainable organic synthesis. This technology addresses the critical need for efficient access to gem-difluorodiene functional groups, which serve as versatile bioisosteres for carbonyls and precursors to various fluorinated scaffolds. By leveraging a metal-free defluorinative phosphorylation strategy, this invention offers a pathway that is not only chemically elegant but also commercially viable for the production of high-purity pharmaceutical intermediates.
Traditionally, the construction of geminal difluorodiene frameworks has been plagued by significant synthetic hurdles that hinder large-scale adoption. Conventional methods often rely heavily on expensive transition metal catalysts, such as palladium or copper complexes, which introduce substantial cost burdens and complicate downstream purification due to strict limits on residual metal content in active pharmaceutical ingredients. Furthermore, these legacy processes frequently require harsh reaction conditions, including high temperatures or the use of sensitive, moisture-intolerant additives that demand rigorous anhydrous environments. Such constraints severely limit the functional group tolerance of the reaction, making it unsuitable for late-stage functionalization of complex drug candidates where diverse chemical moieties are present.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on transition metals in prior art methodologies creates a bottleneck for supply chain efficiency, as the removal of trace metals requires additional processing steps like scavenging or recrystallization, thereby extending lead times and increasing waste generation. Additionally, the use of organic solvents in traditional protocols poses environmental and safety challenges, necessitating complex solvent recovery systems and increasing the carbon footprint of the manufacturing process. The sensitivity of many conventional catalysts to air and moisture further complicates operational procedures, requiring specialized equipment and inert atmosphere handling that drives up capital expenditure. Consequently, there has been a persistent demand for a more robust, cost-effective, and environmentally benign alternative that can deliver these valuable fluorinated building blocks without compromising on yield or selectivity.
The Novel Approach
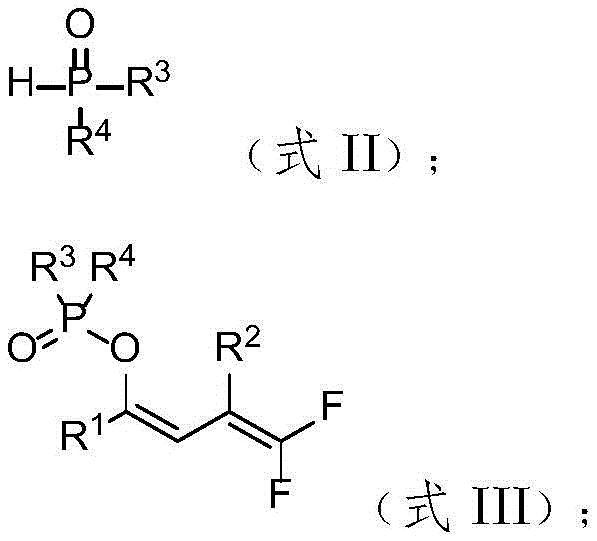
In stark contrast to these limitations, the methodology described in CN115010753A utilizes a simple yet powerful system comprising a trifluoromethyl olefin ketone compound and a phosphine oxide compound reacting in water under basic conditions. This metal-free protocol operates under mild thermal conditions, typically around 70°C, and demonstrates exceptional functional group tolerance, accommodating substrates with halogens, ethers, and heterocycles without degradation. The use of water as the sole reaction medium not only drastically reduces solvent costs but also simplifies workup procedures, aligning perfectly with green chemistry principles. This novel approach effectively bypasses the need for precious metal catalysts, offering a streamlined route to phosphorylated gem-difluorodienes that is ideally suited for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Metal-Free Defluorinative Phosphorylation
The core of this innovation lies in the base-mediated defluorinative phosphorylation mechanism, which proceeds through a concerted elimination-addition pathway facilitated by the unique reactivity of the trifluoromethyl group. In the presence of a strong inorganic base such as cesium carbonate, the phosphine oxide acts as a nucleophile, attacking the electron-deficient olefinic system while simultaneously triggering the elimination of a fluoride ion. This process is highly regioselective, favoring the formation of the thermodynamically stable (Z)-isomer of the phosphorylated geminal difluorodiene product. The aqueous environment plays a crucial role in stabilizing the ionic intermediates and facilitating proton transfer steps, which enhances the overall reaction efficiency without the need for phase-transfer catalysts.
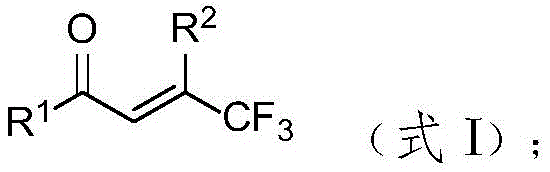
Furthermore, the reaction exhibits remarkable chemoselectivity, tolerating a wide array of substituents on both the ketone and phosphine oxide components, including electron-withdrawing and electron-donating groups on aromatic rings. The mechanism avoids the formation of radical species that often lead to side reactions in metal-catalyzed processes, thereby ensuring a cleaner impurity profile and higher crude purity. This level of control is essential for R&D teams aiming to synthesize complex analogs for structure-activity relationship studies, as it minimizes the burden on purification teams. The ability to generate diverse phosphorylated scaffolds from readily available trifluoromethyl precursors underscores the versatility of this chemistry in constructing high-purity fluorinated building blocks for drug discovery.
How to Synthesize Phosphorylated Geminal Difluorodiene Efficiently
To implement this synthesis effectively, operators must adhere to specific stoichiometric ratios and environmental controls to maximize yield and reproducibility. The patent data indicates that a molar ratio of 1:1.5 between the trifluoromethyl olefin ketone and the phosphine oxide compound is optimal, ensuring complete conversion while minimizing excess reagent waste. The reaction is best conducted under a nitrogen atmosphere to prevent potential oxidation of sensitive intermediates, although the aqueous nature of the system provides inherent stability. Detailed standard operating procedures regarding temperature ramping and quenching protocols are essential for maintaining safety and consistency during scale-up operations.
- Combine trifluoromethyl olefin ketone compound and phosphine oxide compound in water with cesium carbonate base.
- Stir the reaction mixture under nitrogen atmosphere at 70°C for 12 hours to facilitate defluorinative phosphorylation.
- Quench with saturated NH4Cl, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous, metal-free synthesis route offers transformative benefits that extend far beyond simple yield improvements. By eliminating the dependency on volatile precious metal markets and complex catalyst supply chains, manufacturers can achieve significant cost stabilization and predictability in their raw material budgeting. The simplified downstream processing, characterized by straightforward extraction and crystallization from water, reduces the consumption of organic solvents and energy-intensive distillation steps. This translates directly into substantial cost savings in manufacturing overheads and a reduced environmental compliance burden, making the supply of these critical intermediates more resilient and sustainable.
- Cost Reduction in Manufacturing: The complete absence of expensive transition metal catalysts removes a major cost driver from the bill of materials, while the use of inexpensive inorganic bases like cesium carbonate further lowers reagent costs. The elimination of metal scavenging steps and the reduction in solvent usage significantly decrease the operational expenditure associated with waste treatment and solvent recovery. This economic efficiency allows for more competitive pricing strategies when sourcing high-purity fluorinated building blocks, enhancing the overall margin profile of the final pharmaceutical product.
- Enhanced Supply Chain Reliability: Utilizing water as the primary reaction solvent mitigates risks associated with the supply and storage of hazardous organic solvents, which are often subject to strict regulatory controls and transportation restrictions. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or out-of-specification results. This reliability is crucial for maintaining continuous supply lines for critical drug substances, ensuring that downstream formulation activities are not disrupted by upstream synthesis variability.
- Scalability and Environmental Compliance: The mild reaction temperature of 70°C and the use of non-flammable aqueous media make this process inherently safer for large-scale commercial production compared to exothermic metal-catalyzed reactions. The reduced generation of hazardous waste streams simplifies environmental permitting and disposal logistics, aligning with increasingly stringent global sustainability mandates. This scalability ensures that the commercial scale-up of complex organophosphorus compounds can be achieved rapidly to meet market demand without compromising on safety or regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing pipelines. The answers provided are derived directly from the experimental data and beneficial effects outlined in the patent documentation.
Q: What are the advantages of this water-phase synthesis over traditional methods?
A: This method eliminates the need for expensive transition metal catalysts and sensitive additives, utilizing mild conditions (70°C) in water, which significantly reduces cost and environmental waste.
Q: What is the regioselectivity of the phosphorylated gem-difluorodiene products?
A: The reaction exhibits excellent regioselectivity, predominantly yielding the (Z)-isomer of the phosphorylated geminal difluorodiene compounds with high purity.
Q: Can this process be scaled for commercial production?
A: Yes, the use of water as a solvent and inexpensive inorganic bases like cesium carbonate makes the process highly scalable and suitable for industrial manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorylated Geminal Difluorodiene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this metal-free aqueous synthesis technology in advancing the development of next-generation fluorinated therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of phosphorylated geminal difluorodiene intermediate meets the highest industry standards for potency and impurity control.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can optimize your supply chain and reduce overall project costs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific molecule, along with specific COA data and route feasibility assessments. Our experts are ready to provide the technical support and commercial flexibility needed to secure your position as a leader in the competitive landscape of fluorinated pharmaceutical intermediates.
