Revolutionizing Electron-Rich Heterocycle Production: A Green Acid-Catalyzed Strategy for Commercial Scale-Up
The pharmaceutical and agrochemical industries rely heavily on the availability of diverse, high-purity heterocyclic building blocks to construct complex active pharmaceutical ingredients (APIs). Patent CN112538068A introduces a transformative synthetic methodology for preparing multi-substituted electron-rich five-membered heterocyclic rings, specifically targeting thiophene and pyrrole scaffolds which are ubiquitous in medicinal chemistry. This innovation addresses critical bottlenecks in traditional organic synthesis by replacing harsh, metal-dependent protocols with a mild, non-metal catalytic system. By utilizing cheap Lewis or Bronsted acids to catalyze the [3+2] cycloaddition between electron-rich alkynes and alpha-mercaptoketone analogs, this technology enables the rapid assembly of complex molecular architectures with exceptional efficiency. The strategic shift towards metal-free catalysis not only enhances the environmental profile of the synthesis but also drastically simplifies the purification process, making it an ideal candidate for reliable pharma intermediate supplier operations seeking to optimize their manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted five-membered heterocycles has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical intermediates. Classical approaches such as the Gewald reaction or Paal-Knorr synthesis often suffer from severe substrate limitations, restricting the types of functional groups that can be tolerated at specific positions on the heterocyclic ring. Furthermore, modern transition metal-catalyzed methods, while versatile, typically require the use of expensive noble metals like palladium, gold, or rhodium, alongside harsh oxidants and elevated temperatures. These conditions not only inflate raw material costs but also introduce the persistent risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet stringent regulatory purity specifications. The generation of hazardous waste and the incompatibility with sensitive functional groups further limit the utility of these traditional routes in the synthesis of delicate drug candidates.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112538068A offers a streamlined, green alternative that leverages the power of cheap acid catalysis to drive the formation of these valuable scaffolds. This novel approach utilizes inexpensive catalysts such as acetic acid or boron trifluoride etherate to facilitate a tandem [3+2] cycloaddition reaction under remarkably mild conditions, often at room temperature. The reaction is highly atom-economical, producing water as the sole byproduct, which aligns perfectly with modern principles of green chemistry and waste reduction. By avoiding the use of transition metals and strong oxidants, this method inherently prevents heavy metal residue issues, thereby ensuring a cleaner product profile right from the crude reaction mixture. The versatility of this system allows for the introduction of diverse substituents at the 2, 3, 4, and 5-positions of the heterocycle, providing chemists with unprecedented control over molecular architecture without compromising on yield or operational simplicity.
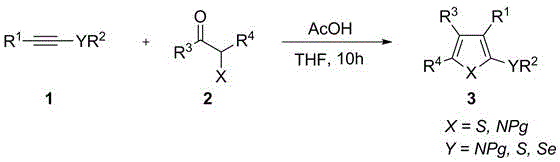
Mechanistic Insights into Acid-Catalyzed [3+2] Cycloaddition
The core of this technological breakthrough lies in the activation of the carbonyl component by a non-metal acid catalyst, which initiates a cascade of nucleophilic attacks and cyclization events. In the presence of a Lewis acid like BF3·Et2O or a Bronsted acid like AcOH, the electrophilicity of the alpha-mercaptoketone or its nitrogen analog is significantly enhanced. This activation facilitates the nucleophilic attack by the electron-rich alkyne species, leading to the formation of a key intermediate that subsequently undergoes intramolecular cyclization. The reaction proceeds through a concerted or stepwise mechanism depending on the specific substrates, ultimately resulting in the aromatization of the five-membered ring and the elimination of a water molecule. This mechanistic pathway is distinct from oxidative coupling reactions, as it does not require external oxidants to drive the formation of the heterocyclic bond, thereby preserving sensitive functional groups that might otherwise be degraded under oxidative stress.
From an impurity control perspective, the absence of transition metals is a decisive advantage for maintaining high product purity throughout the synthesis. Traditional metal-catalyzed routes often generate complex impurity profiles containing metal-ligand complexes or side products derived from metal-mediated decomposition, which are notoriously difficult to remove. In this acid-catalyzed system, the impurity profile is significantly simplified, primarily consisting of unreacted starting materials or minor regioisomers that are easily separated via standard flash column chromatography. The mild reaction conditions also minimize thermal degradation of the product, ensuring that the final isolated material retains its structural integrity. This robustness in impurity management is critical for R&D directors who require consistent, high-quality batches for biological screening and subsequent process development, as it reduces the analytical burden and accelerates the timeline for candidate selection.
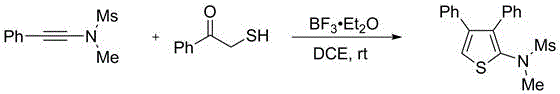
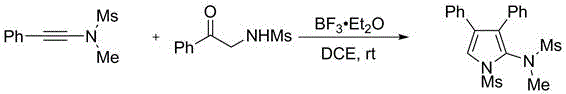
How to Synthesize Multi-Substituted Thiophene Efficiently
The practical implementation of this synthesis route is designed for operational ease, allowing laboratories to produce high-value heterocycles with minimal equipment requirements. The general procedure involves dissolving the electron-rich alkyne and the alpha-mercaptoketone analog in a common organic solvent such as tetrahydrofuran (THF) or dichloroethane (DCE). A catalytic amount of the chosen acid is then added to the mixture, which is stirred at room temperature for a period ranging from minutes to several hours, depending on the specific reactivity of the substrates. Upon completion, the reaction is quenched with a simple aqueous workup involving saturated sodium bicarbonate and brine, followed by extraction and concentration. The detailed standardized synthesis steps see the guide below for precise stoichiometric ratios and workup parameters optimized for maximum yield.
- Mix electron-rich alkyne and alpha-mercaptoketone (or analog) substrates in a suitable solvent like THF or DCE at room temperature.
- Add a catalytic amount of cheap Lewis acid (e.g., BF3·Et2O) or Bronsted acid (e.g., AcOH) to initiate the [3+2] cycloaddition.
- Stir the reaction mixture until completion, then quench with saturated salt solutions and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this acid-catalyzed technology represents a strategic opportunity to enhance supply security while driving down the total cost of ownership for key intermediates. By shifting away from precious metal catalysts, manufacturers can insulate their production costs from the volatile pricing of commodities like palladium and gold, leading to more stable long-term budgeting. The mild reaction conditions also translate to lower energy consumption, as there is no need for heating or specialized pressure vessels, further contributing to operational expenditure savings. Additionally, the simplified purification process reduces the consumption of silica gel and solvents during chromatography, offering substantial cost savings in consumables. These factors collectively create a more resilient and cost-effective supply chain capable of responding rapidly to market demands without the bottlenecks associated with complex metal removal processes.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and ligands fundamentally alters the cost structure of heterocycle production. Instead of relying on scarce resources that require complex recovery systems, this method utilizes abundant, low-cost acids that are readily available in bulk quantities. This substitution removes the need for specialized scavenger resins or extensive washing protocols designed to reduce metal levels to ppm ranges, thereby shortening the production cycle time. Consequently, the overall manufacturing cost is significantly reduced, allowing for more competitive pricing strategies in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized catalytic systems enhances the robustness of the supply chain. Sourcing acetic acid or boron trifluoride is far less risky than securing high-purity organometallic complexes, which can be subject to geopolitical supply disruptions. Furthermore, the ambient temperature operation reduces the safety risks associated with exothermic reactions or high-pressure systems, minimizing the potential for unplanned shutdowns due to safety incidents. This reliability ensures a continuous flow of materials, which is essential for maintaining just-in-time inventory levels and meeting tight delivery schedules for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of hazardous oxidants and the generation of benign byproducts. The fact that water is the only byproduct simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the costs associated with waste disposal and environmental compliance monitoring. As global regulations on chemical manufacturing become increasingly stringent, adopting such environmentally friendly processes provides a distinct competitive advantage and future-proofs the production capability against evolving legal standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this chemistry into their existing production workflows.
Q: What are the primary advantages of this acid-catalyzed method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic precious metals like palladium, gold, or rhodium. It operates under mild room temperature conditions, produces water as the only byproduct, and avoids heavy metal residue issues, significantly simplifying downstream purification and environmental compliance.
Q: Can this synthesis method be applied to both thiophene and pyrrole scaffolds?
A: Yes, the patent demonstrates high universality. By varying the heteroatom source in the starting materials (using alpha-mercaptoketones for thiophenes or nitrogen-containing analogs for pyrroles), the method efficiently constructs both sulfur and nitrogen-containing five-membered heterocyclic rings with high yields.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The use of cheap, commercially available acid catalysts, combined with room temperature operation and simple aqueous workup procedures, makes this process highly scalable. It removes the safety hazards associated with strong oxidants and high temperatures found in conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multi-Substituted Thiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this acid-catalyzed methodology for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this green chemistry approach, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity OLED material and pharmaceutical intermediates that meet the exacting standards of the global market, leveraging our expertise to optimize yield and minimize waste at every stage of the process.
We invite you to collaborate with us to unlock the full commercial potential of this technology for your specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating how this route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these critical building blocks.
