Advanced Organocatalytic Synthesis for Scalable Pharmaceutical Intermediate Production
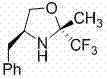
The pharmaceutical and fine chemical industries are currently undergoing a significant paradigm shift towards greener, more sustainable synthetic methodologies, driven by stringent regulatory requirements and the need for cost-effective manufacturing. Patent CN102093303B introduces a pivotal advancement in this domain with the disclosure of (2R,4S)-4-phenyl-2-methyl-2-trifluoromethyl oxazoline, a novel chiral organocatalyst designed to facilitate highly stereoselective aldol condensation reactions. This specific molecular architecture represents a breakthrough in asymmetric organic synthesis, offering a metal-free alternative to traditional transition metal complexes that have long dominated the field. By leveraging the unique electronic properties of the trifluoromethyl group combined with a rigid oxazoline backbone, this catalyst enables the construction of complex chiral building blocks with exceptional precision. For R&D directors and procurement strategists, understanding the implications of this technology is crucial, as it directly addresses critical pain points regarding impurity profiles, regulatory compliance, and overall process efficiency in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alpha,beta-dihydroxy ketone compounds, which are vital precursors for numerous active pharmaceutical ingredients, has relied heavily on transition metal catalysis or enzymatic processes. While effective, transition metal catalysts often introduce significant downstream challenges, primarily concerning the removal of toxic metal residues to meet strict ppm limits set by health authorities. The purification steps required to eliminate traces of metals like copper, zinc, or palladium are not only technically demanding but also substantially increase the cost of goods sold and extend the overall production lead time. Furthermore, many conventional organometallic reagents are sensitive to moisture and air, necessitating specialized equipment and inert atmosphere conditions that complicate scale-up efforts. Enzymatic approaches, although green, can suffer from limited substrate scope and stability issues under industrial reaction conditions, often requiring expensive cofactors or complex immobilization techniques. These inherent limitations create bottlenecks in the supply chain, reducing the agility of manufacturers to respond to market demands for complex chiral intermediates.
The Novel Approach
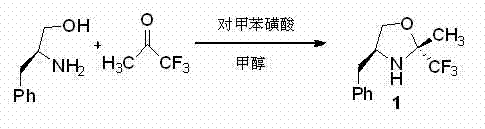
The methodology described in patent CN102093303B offers a transformative solution by utilizing a small organic molecule catalyst that circumvents the drawbacks associated with metal-based systems. This novel approach employs a condensation reaction between L-phenylalaninol and trifluoroacetone, catalyzed by p-toluenesulfonic acid in methanol, to generate the active oxazoline species. The resulting catalyst operates through a distinct hydrogen-bonding activation mechanism, which activates the carbonyl substrate without the need for toxic metals. This metal-free nature inherently simplifies the work-up procedure, as there is no need for specialized scavenging resins or extensive washing protocols to remove metal contaminants. The operational simplicity is further enhanced by the use of common solvents like methanol and standard reflux conditions, making the process robust and easily transferable from laboratory to pilot plant scales. By shifting to this organocatalytic platform, manufacturers can achieve a cleaner reaction profile, reduced environmental footprint, and a more streamlined path to regulatory approval for final drug substances.
Mechanistic Insights into Organocatalytic Asymmetric Aldol Reaction
The efficacy of the (2R,4S)-4-phenyl-2-methyl-2-trifluoromethyl oxazoline catalyst lies in its sophisticated ability to control stereochemistry through a well-defined transition state. The trifluoromethyl group at the 2-position of the oxazoline ring exerts a strong electron-withdrawing effect, which modulates the acidity of the nearby protons and enhances the electrophilicity of the catalyst-substrate complex. During the aldol reaction, the catalyst likely forms a transient enamine or iminium intermediate with the ketone donor, organizing the spatial arrangement of the reacting species through a network of hydrogen bonds and steric interactions. The bulky phenyl group at the 4-position provides a steric shield that directs the approach of the aldehyde acceptor, ensuring that the nucleophilic attack occurs from a specific face. This precise spatial control is what allows the system to achieve high enantiomeric excess, particularly with electron-deficient aromatic aldehydes, where electronic complementarity further stabilizes the transition state. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction parameters for maximum yield and selectivity in commercial settings.
Impurity control is another critical aspect where this catalytic system excels, directly impacting the quality of the final pharmaceutical intermediate. In traditional metal-catalyzed reactions, side reactions such as over-reduction or metal-mediated decomposition can lead to complex impurity profiles that are difficult to separate. In contrast, the organocatalytic pathway described here proceeds through a cleaner mechanism with fewer competing side reactions. The moderate reaction temperatures and the specific activation mode minimize the formation of by-products, resulting in a crude reaction mixture that is easier to purify. The patent data indicates that the catalyst maintains good stability over the reaction course, preventing degradation products from accumulating. For quality assurance teams, this translates to a more consistent product quality and a reduced risk of batch failure due to out-of-specification impurities. The ability to consistently produce high-purity material is a key determinant in securing long-term supply contracts with top-tier pharmaceutical companies.
How to Synthesize (2R,4S)-4-phenyl-2-methyl-2-trifluoromethyl oxazoline Efficiently
The preparation of this high-value chiral catalyst is designed for operational simplicity and robustness, making it an ideal candidate for technology transfer. The synthesis begins with the dissolution of L-phenylalaninol and a catalytic amount of p-toluenesulfonic acid in anhydrous methanol, creating a homogeneous reaction mixture. Under controlled ice-water bath conditions, trifluoroacetone is added slowly to manage the exotherm, ensuring safety and preventing thermal degradation of the sensitive intermediates. The mixture is then allowed to warm to reflux and stirred for a period ranging from 24 to 36 hours, allowing the condensation to reach completion. Following the reaction, the solvent is removed under reduced pressure, and the crude product is purified using standard silica gel column chromatography. The detailed standardized synthesis steps see the guide below.
- Dissolve L-phenylalaninol and p-toluenesulfonic acid catalyst in anhydrous methanol under ice-water bath conditions.
- Slowly add trifluoroacetone to the mixture while maintaining low temperature to control exothermic reaction.
- Warm to reflux and stir for 24 to 36 hours, then purify via silica gel column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology presents a compelling value proposition centered around cost efficiency and supply reliability. The elimination of transition metals from the synthetic route removes a significant cost center associated with metal scavengers, specialized waste disposal, and extended purification times. This simplification of the process flow directly contributes to a reduction in the overall cost of manufacturing, allowing for more competitive pricing strategies in the global market. Furthermore, the raw materials required for this synthesis, such as L-phenylalaninol and trifluoroacetone, are commercially available in bulk quantities from established chemical suppliers, mitigating the risk of raw material shortages. The robustness of the reaction conditions, which do not require inert atmospheres or cryogenic temperatures, reduces the capital expenditure needed for specialized reactor infrastructure. These factors combined create a resilient supply chain capable of sustaining high-volume production without the fragility often associated with complex biocatalytic or organometallic processes.
- Cost Reduction in Manufacturing: The transition to a metal-free organocatalytic process fundamentally alters the cost structure of producing chiral intermediates. By removing the necessity for expensive transition metal salts and the subsequent purification steps required to meet residual metal limits, manufacturers can achieve substantial cost savings. The simplified work-up procedure reduces solvent consumption and labor hours, while the use of common solvents like methanol avoids the premium pricing of specialized anhydrous or degassed solvents. Additionally, the moderate reaction conditions lower energy consumption compared to processes requiring deep cooling or high-pressure equipment. These cumulative efficiencies result in a significantly lower cost of goods sold, enhancing the margin potential for downstream API production.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized reagents with long lead times or single-source suppliers. This technology leverages commodity chemicals that are produced on a multi-ton scale globally, ensuring a stable and diverse supply base. The operational simplicity of the synthesis reduces the dependency on highly specialized technical staff, making it easier to replicate the process across multiple manufacturing sites if needed. This flexibility is crucial for mitigating risks associated with geopolitical disruptions or facility-specific issues. By adopting a process with broad raw material availability and standard equipment requirements, companies can build a more agile and responsive supply network capable of meeting fluctuating market demands.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces unforeseen challenges, particularly regarding heat transfer and mixing efficiency. The exothermic nature of this condensation reaction is manageable under standard reflux conditions, making it inherently scalable from kilogram to multi-ton batches. From an environmental perspective, the absence of heavy metals simplifies waste treatment and disposal, aligning with increasingly strict environmental regulations and corporate sustainability goals. The reduced toxicity profile of the reagents and products enhances workplace safety and lowers the regulatory burden associated with hazardous material handling. This alignment with green chemistry principles not only improves the environmental footprint but also strengthens the brand reputation of the manufacturer as a responsible and sustainable partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organocatalytic technology. These insights are derived directly from the patent data and practical considerations for industrial application, providing clarity on performance metrics and operational requirements. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this catalyst into their existing manufacturing workflows.
Q: What are the primary advantages of this organocatalyst over transition metal catalysts?
A: This organocatalyst eliminates the risk of heavy metal contamination, which is critical for pharmaceutical compliance. It offers simpler operation, lower toxicity, and avoids expensive metal removal steps, significantly streamlining the purification process.
Q: What is the enantioselectivity performance in aldol reactions?
A: The catalyst demonstrates high stereoselectivity, achieving up to 99% ee in specific aldol condensations with electron-withdrawing aromatic aldehydes, ensuring high optical purity for downstream API synthesis.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the method utilizes readily available raw materials and standard reflux conditions in methanol. The absence of sensitive transition metals and the robustness of the reaction conditions make it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2R,4S)-4-phenyl-2-methyl-2-trifluoromethyl oxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in the development of next-generation pharmaceuticals. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and regulatory compliance, enabling you to accelerate your drug development timelines with confidence.
We invite you to engage with our technical procurement team to discuss how this organocatalytic route can optimize your specific synthesis requirements. We offer a Customized Cost-Saving Analysis to evaluate the potential economic benefits of switching to this metal-free methodology for your projects. Please contact us to request specific COA data and route feasibility assessments tailored to your target molecules. Our goal is to provide not just a chemical product, but a comprehensive strategic solution that enhances your competitive advantage in the global marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
