Advanced Immobilized Platinum Catalyst Technology for Scalable Hydroxylamine Compound Manufacturing
Advanced Immobilized Platinum Catalyst Technology for Scalable Hydroxylamine Compound Manufacturing
Introduction to Patent CN100335460C and Technical Breakthroughs
The chemical manufacturing landscape is constantly evolving to meet the stringent demands of high-purity intermediate production, and patent CN100335460C represents a significant leap forward in the synthesis of hydroxylamine compounds. This intellectual property discloses a novel method for producing hydroxylamine compounds by utilizing a platinum catalyst that is firmly immobilized on an ion-exchange resin matrix, fundamentally altering the efficiency and safety profile of the reduction process. Unlike traditional heterogeneous catalysis where metal leaching and difficult separation are persistent challenges, this technology ensures that the platinum active sites are securely anchored within the porous structure of the resin, leading to exceptional stability and reusability. The innovation addresses critical pain points in the pharmaceutical and agrochemical sectors by offering a pathway to produce target hydroxylamines with minimal by-product formation, specifically suppressing the over-reduction to amines which often plagues conventional nitro compound reductions. By leveraging this advanced catalytic system, manufacturers can achieve industrial-scale production with enhanced safety protocols, as the process eliminates the need for hazardous reducing agents like sodium hydrosulfide that generate toxic odors and waste streams. The technical robustness of this method provides a solid foundation for supply chain partners seeking reliable sources of high-value intermediates that require strict impurity control and consistent quality assurance across large production batches.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of hydroxylamine compounds such as phenylhydroxylamine has relied on methods that are fraught with significant technical and operational inefficiencies that hinder large-scale commercial viability. One prevalent conventional approach involves the reduction of nitrobenzene using sodium hydrosulfide, a process that suffers from inherently low reaction selectivity resulting in suboptimal yields of the target hydroxylamine compound that fail to meet industrial economic thresholds. Furthermore, the use of sodium hydrosulfide introduces severe environmental and occupational health hazards due to the generation of malodorous and toxic hydrogen sulfide gas, necessitating complex and costly abatement systems to protect the working environment and surrounding communities. Another traditional method employs platinum carbon catalysts for hydrogenation, but this technique often struggles to achieve yields sufficient for industrial application without compromising selectivity, as increasing reaction temperatures to boost conversion rates frequently leads to a sharp decline in selectivity and increased formation of unwanted aniline by-products. Additionally, the physical properties of conventional platinum carbon catalysts, characterized by extremely fine particle sizes, make post-reaction recovery and reuse operations cumbersome and prone to material loss, thereby driving up operational costs and creating waste disposal challenges. The combustion properties of dry platinum carbon also mandate rigorous safety handling procedures, adding another layer of complexity and risk to the manufacturing process that modern facilities strive to eliminate through safer technological alternatives.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes an ion-exchange resin-immobilized platinum catalyst that fundamentally resolves the selectivity and recovery issues associated with traditional catalytic systems. By fixing the platinum metal onto a robust ion-exchange resin substrate, the catalyst maintains high activity and selectivity even under conditions that would typically degrade performance in conventional setups, allowing for the efficient and safe industrial production of target hydroxylamine compounds. The structural integrity of the resin matrix prevents the leaching of platinum into the reaction mixture, ensuring that the final product remains free from heavy metal contamination which is a critical quality parameter for pharmaceutical and electronic grade intermediates. Moreover, the significantly larger particle size of the resin-supported catalyst compared to fine platinum carbon powders facilitates straightforward filtration and separation processes, enabling easy recovery and repeated reuse of the expensive platinum material without significant loss of catalytic activity over multiple cycles. This technological advancement not only streamlines the production workflow by reducing downtime associated with catalyst changeovers but also enhances the overall economic feasibility of the process by maximizing the utility of the precious metal catalyst. The ability to operate with high selectivity and minimal by-product generation translates directly into simplified downstream purification steps, reducing solvent consumption and energy requirements while delivering a superior quality product that meets the exacting standards of global supply chains.

Mechanistic Insights into Ion Exchange Resin Immobilized Platinum Catalysis
The core of this technological breakthrough lies in the precise engineering of the catalyst structure, where platinum metal is immobilized on specific ion-exchange resin matrices to create a highly stable and selective catalytic environment. The resin backbone is typically derived from copolymers such as styrene-divinylbenzene or polyacrylate esters, which are functionalized with ion-exchange groups like tertiary amino or quaternary ammonium groups that serve as anchoring points for the platinum species. During the catalyst preparation phase, platinum-containing ions from aqueous solutions are adsorbed onto these functional groups through ion exchange mechanisms, followed by a reduction treatment that converts the ionic platinum into metallic platinum clusters firmly embedded within the resin structure. This immobilization strategy ensures that the active platinum sites are spatially distributed in a manner that favors the partial reduction of nitro groups to hydroxylamines while sterically or electronically hindering the further reduction to amines, thereby achieving the high selectivity observed in the patent examples. The chemical environment provided by the resin matrix also modulates the electronic state of the platinum, potentially enhancing its activity towards the specific hydrogenation of the nitro group using hydrogen sources like hydrazine without promoting side reactions.
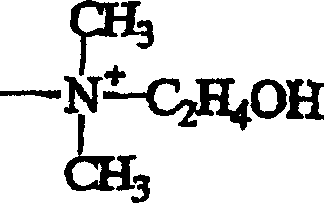
Impurity control is another critical aspect of the mechanism, as the immobilized catalyst system significantly suppresses the formation of aniline, which is the primary by-product in conventional nitrobenzene reduction processes. The patent data indicates that by optionally adding specific poisoning agents or modifiers to the reaction system, the selectivity towards the hydroxylamine compound can be further enhanced, suggesting a tunable catalytic surface that can be optimized for different substrate profiles. The stability of the catalyst over repeated use cycles, as evidenced by consistent yield and selectivity data in the patent examples, implies that the platinum clusters do not aggregate or leach significantly during the reaction conditions, maintaining their active surface area and geometric configuration. This mechanistic stability is crucial for commercial operations where batch-to-batch consistency is paramount, as it ensures that the impurity profile of the product remains within tight specifications without requiring extensive re-optimization of reaction parameters for each new production run. The combination of structural stability, high selectivity, and ease of separation makes this catalytic system a superior choice for the manufacture of sensitive intermediates where purity and process reliability are non-negotiable requirements for downstream application success.
How to Synthesize Hydroxylamine Compound Efficiently
The synthesis of hydroxylamine compounds using this advanced catalytic technology involves a streamlined sequence of steps that begins with the preparation of the specialized ion-exchange resin-immobilized platinum catalyst followed by the reduction reaction under controlled conditions. Operators must first ensure that the catalyst is properly activated and reduced before introducing the nitro compound substrate and the hydrogen source, typically hydrazine, into the reaction vessel to initiate the transformation. The process allows for flexibility in solvent selection, including water, alcohols, or mixtures thereof, and can be conducted under atmospheric or elevated pressure depending on the specific reactivity of the nitro substrate being processed. Detailed standardized synthesis steps see the guide below for precise operational parameters regarding temperature, pressure, and stoichiometry to ensure optimal yield and safety.
- Prepare the ion-exchange resin support by selecting a styrene-divinylbenzene copolymer with appropriate anion or cation exchange groups.
- Adsorb platinum ions from an aqueous platinum compound solution onto the resin matrix followed by thorough washing.
- Perform reduction treatment using hydrogen or hydrazine to fix platinum metal on the resin before contacting with nitro compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this immobilized catalyst technology translates into tangible strategic advantages that directly impact the bottom line and operational resilience of the manufacturing organization. The elimination of hazardous reagents like sodium hydrosulfide removes the need for specialized handling equipment and costly waste treatment protocols associated with toxic sulfur by-products, thereby simplifying the regulatory compliance landscape and reducing the overall environmental footprint of the production facility. Furthermore, the enhanced stability and reusability of the catalyst mean that the frequency of catalyst replacement is drastically reduced, leading to substantial cost savings on precious metal consumption and minimizing supply chain disruptions caused by catalyst procurement delays. The high selectivity of the process reduces the burden on downstream purification units, allowing for higher throughput and lower energy consumption per unit of product, which collectively contribute to a more competitive cost structure in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The immobilization of platinum on the resin matrix eliminates the need for expensive and complex heavy metal removal steps that are typically required when using homogeneous or fine particulate catalysts, resulting in significant operational cost optimizations. By preventing platinum leaching, the process ensures that the valuable metal remains within the catalyst bed for reuse, drastically reducing the raw material costs associated with precious metal consumption over the long term. Additionally, the high selectivity minimizes the formation of by-products that would otherwise require costly separation and disposal processes, further enhancing the economic efficiency of the manufacturing route. The simplified workflow also reduces labor and utility costs associated with extended reaction times and complex workup procedures, providing a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robust nature of the resin-immobilized catalyst ensures consistent performance over multiple batches, reducing the risk of production failures or off-spec material that could disrupt supply commitments to key customers. The ease of catalyst recovery and reuse means that facilities can maintain continuous operation with minimal downtime for catalyst changeovers, thereby improving overall equipment effectiveness and delivery reliability. Moreover, the use of safer reagents and conditions reduces the likelihood of safety incidents that could halt production, ensuring a more stable and predictable supply of high-purity intermediates to the market. This reliability is critical for pharmaceutical and agrochemical supply chains where continuity of supply is often contractually mandated and essential for patient and crop protection.
- Scalability and Environmental Compliance: The large particle size of the catalyst facilitates easy scale-up from laboratory to commercial production without the filtration and handling issues associated with fine powders, enabling rapid deployment of the technology to meet increasing market demand. The process generates significantly less hazardous waste compared to conventional methods, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. The absence of toxic sulfur emissions and the reduction in solvent usage due to higher selectivity contribute to a greener manufacturing profile that enhances the brand reputation of the supplier. This environmental compliance not only mitigates regulatory risk but also appeals to end-customers who are prioritizing sustainable sourcing in their supply chain strategies.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical specifications and experimental data provided in the patent documentation to address common technical and commercial inquiries regarding this catalytic technology. These insights are intended to clarify the operational benefits and mechanistic advantages that distinguish this method from traditional hydroxylamine synthesis routes currently in use. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing infrastructure or for procurement teams assessing the quality and reliability of the supply source.
Q: How does the immobilized catalyst improve selectivity compared to conventional methods?
A: The ion-exchange resin matrix stabilizes the platinum active sites, preventing over-reduction to amines and minimizing by-product formation like aniline, which is a common issue with traditional platinum carbon catalysts.
Q: Is the catalyst reusable for multiple production batches?
A: Yes, the large particle size of the resin-immobilized catalyst allows for easy filtration and recovery, and the patent data confirms that catalytic activity remains stable over repeated cycles without significant decline.
Q: What safety advantages does this method offer over sodium hydrosulfide reduction?
A: This method eliminates the use of sodium hydrosulfide, thereby removing the risk of toxic hydrogen sulfide gas generation and unpleasant odors, creating a safer industrial working environment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxylamine Compound Supplier
The technical potential of this immobilized platinum catalyst route represents a significant opportunity for optimizing the production of high-value hydroxylamine intermediates, and NINGBO INNO PHARMCHEM stands ready to leverage this expertise for your commercial needs. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing with rigorous adherence to quality standards. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the low impurity profiles achievable with this advanced catalytic method, guaranteeing that every batch meets the exacting requirements of the pharmaceutical and specialty chemical industries. We understand the critical nature of intermediate supply and are committed to delivering consistent quality and reliability that supports your long-term product lifecycle management.
We invite you to engage with our technical procurement team to discuss how this technology can be tailored to your specific production requirements and to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits for your operation. Please contact us to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this approach for your target molecules. Our team is prepared to provide the technical depth and commercial flexibility needed to secure your supply chain and enhance your competitive position in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
