Scaling High-Performance Blue Electroluminescent Materials for Next-Gen Displays
Scaling High-Performance Blue Electroluminescent Materials for Next-Gen Displays
The rapid evolution of organic light-emitting diode (OLED) technology has placed immense pressure on material scientists to develop stable, efficient blue emitters that can match the longevity of their red and green counterparts. Patent CN102464978B introduces a groundbreaking class of conductive cavity-type blue electroluminescent materials based on a unique triptycene-anthracene hybrid architecture. This innovation addresses the critical industry pain point of blue material instability by leveraging the rigid, three-dimensional structure of triptycene to inhibit molecular aggregation and recrystallization. For R&D directors and procurement specialists in the electronic chemical sector, understanding the synthetic accessibility and performance metrics of these compounds is vital for securing a competitive edge in the display manufacturing supply chain.
These materials, defined by structural formulas I, II, and III, incorporate electron-donating groups and alkyl chains that not only enhance hole transport properties but also drastically improve solubility for solution-processable fabrication methods. The strategic integration of thiophene linkers further optimizes the conjugation system, facilitating efficient charge injection while maintaining the high energy gap required for pure blue emission. As the demand for high-resolution, energy-efficient displays grows, the ability to source such high-purity OLED material becomes a strategic imperative for downstream device manufacturers seeking to optimize yield and performance consistency.
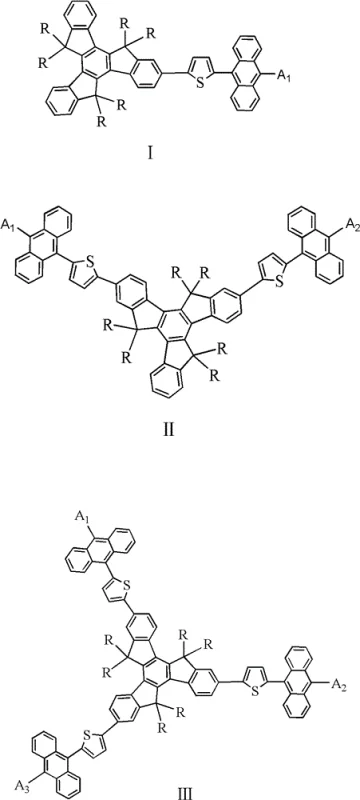
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of blue organic electroluminescent materials has been plagued by inherent stability issues associated with planar aromatic systems like anthracene single crystals. While anthracene offers excellent luminescent properties, its tendency to crystallize easily leads to morphological instability in thin films, causing device degradation and shortened operational lifetimes. Traditional approaches often rely on bulky substituents to sterically hinder packing, but these modifications frequently come at the cost of reduced charge mobility or complex, low-yield synthetic routes that are difficult to scale. Furthermore, many existing blue emitters suffer from poor solubility, limiting their application to vacuum deposition techniques which are more expensive and less versatile than solution processing methods like inkjet printing.
The Novel Approach
The methodology outlined in the patent data presents a sophisticated solution by utilizing a triptycene core as a central scaffold. This three-dimensional propeller-shaped structure acts as a molecular spacer, effectively preventing the close packing of the emissive anthracene units and thus suppressing excimer formation and crystallization. The synthetic strategy employs a modular Suzuki-Miyaura coupling approach, allowing for the precise assembly of the final molecule from readily available precursors. This convergent synthesis not only simplifies purification processes but also enables the facile tuning of electronic properties by varying the substituents on the anthracene periphery. By combining the stability of triptycene with the high fluorescence efficiency of anthracene-thiophene derivatives, this novel approach delivers a material that balances high quantum efficiency with exceptional thermal and morphological stability.
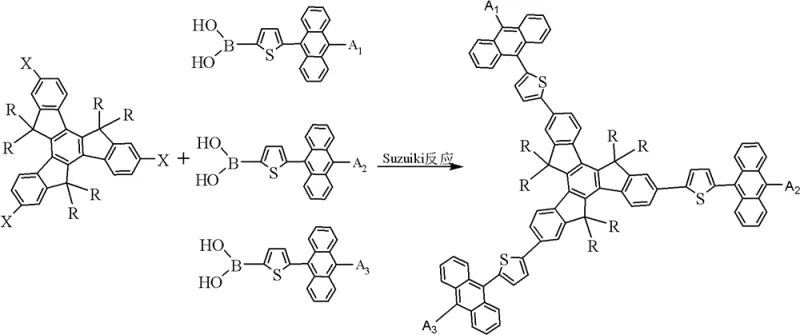
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling
The core of this synthesis relies on the palladium-catalyzed Suzuki-Miyaura cross-coupling reaction, a robust method for forming carbon-carbon bonds between aryl halides and organoboron compounds. In this specific application, the reaction involves the coupling of halogenated triptycene derivatives (Compounds A, B, or C) with thiophene-anthracene boronic acids (Compound D). The mechanism proceeds through a catalytic cycle involving oxidative addition of the aryl halide to the Pd(0) species, transmetallation with the boronate complex activated by base, and reductive elimination to form the biaryl bond. The use of organophosphorus ligands enhances the stability of the palladium catalyst and facilitates the reaction under relatively mild conditions, typically between 60°C and 130°C. This mechanistic pathway is highly tolerant of functional groups, ensuring that the delicate electron-donating substituents on the anthracene ring remain intact throughout the synthesis.
Impurity control is paramount in electronic grade materials, and this synthetic route offers distinct advantages in minimizing side products. The regioselectivity of the initial halogenation steps on the triptycene core ensures that coupling occurs only at the desired positions (2, 7, 12-positions), preventing the formation of structural isomers that could act as quenching sites in the final device. Furthermore, the byproducts of the Suzuki reaction, primarily inorganic boron salts and palladium residues, can be efficiently removed through standard aqueous workups and silica gel chromatography. The high crystallinity of the intermediate halogenated triptycenes also allows for recrystallization steps that significantly upgrade purity before the final coupling, ensuring that the final electroluminescent material meets the stringent purity specifications required for commercial OLED production.
How to Synthesize Conductive Cavity-Type Blue Electroluminescent Material Efficiently
The synthesis of these advanced materials follows a logical, step-wise progression that begins with the construction of the triptycene backbone from 1-indone. This foundational step establishes the rigid core necessary for morphological stability. Subsequent functionalization involves lithiation and alkylation to introduce solubilizing chains, followed by electrophilic aromatic substitution to install the reactive halide handles. The parallel synthesis of the anthracene-thiophene boronic acid coupling partners allows for modular variation of the electronic properties. The final convergence of these fragments via palladium catalysis yields the target compounds with high fidelity. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Synthesize the triptycene core from 1-indone via acid-catalyzed cyclization, followed by alkylation with alkyl halides using n-Butyl Lithium at -78°C.
- Perform regioselective halogenation on the alkylated triptycene using bromine or NBS to generate the electrophilic coupling partner.
- Execute the final Suzuki-Miyaura coupling reaction between the halogenated triptycene and anthracene-thiophene boronic acid derivatives under inert atmosphere.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described in patent CN102464978B offers significant advantages in terms of raw material availability and process robustness. The starting materials, such as 1-indone and anthracene derivatives, are commodity chemicals available from multiple global suppliers, reducing the risk of supply chain bottlenecks. The reliance on well-established reactions like Friedel-Crafts cyclization, lithiation, and Suzuki coupling means that the process can be transferred to standard multipurpose reactors without requiring specialized high-pressure or cryogenic equipment beyond standard low-temperature capabilities. This compatibility with existing infrastructure translates directly into lower capital expenditure for contract manufacturing organizations (CMOs) and faster time-to-market for new material grades.
- Cost Reduction in Manufacturing: The convergent nature of the synthesis allows for the optimization of individual fragments before final assembly, which minimizes the loss of high-value intermediates. By avoiding exotic reagents and utilizing catalytic amounts of palladium, the overall cost of goods sold (COGS) is significantly optimized compared to stoichiometric coupling methods. Additionally, the high solubility of the intermediates facilitates easier handling and purification, reducing solvent consumption and processing time. These factors collectively contribute to substantial cost savings in electronic chemical manufacturing, making high-performance blue emitters more economically viable for mass-market display applications.
- Enhanced Supply Chain Reliability: The modularity of the synthetic design means that different variants of the material (e.g., varying alkyl chain lengths or electron-donating groups) can be produced using the same core process flow. This flexibility allows suppliers to rapidly adapt to specific customer requirements without requalifying entirely new synthetic routes. Furthermore, the stability of the intermediates ensures that they can be stockpiled if necessary, providing a buffer against demand fluctuations. This resilience is crucial for maintaining continuous supply to panel manufacturers who operate on tight just-in-time production schedules.
- Scalability and Environmental Compliance: The reaction conditions are generally mild, operating at atmospheric pressure and moderate temperatures, which simplifies safety management and waste treatment protocols. The primary waste streams consist of aqueous salt solutions and organic solvents that can be recovered and recycled using standard distillation technologies. The absence of heavy metal stoichiometric reagents reduces the burden of hazardous waste disposal. Consequently, scaling this process from pilot plant to commercial tonnage is straightforward, aligning with modern green chemistry principles and regulatory standards for sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triptycene-based blue emitters. The answers are derived from the specific technical disclosures and experimental data found within the patent literature, providing a reliable foundation for decision-making.
Q: What are the key stability advantages of this triptycene-anthracene hybrid structure?
A: The rigid triptycene core prevents recrystallization and aggregation, which are common failure modes in traditional anthracene-based blue emitters, thereby significantly enhancing thermal stability and device lifetime.
Q: How does the Suzuki coupling method impact production scalability?
A: The Suzuki reaction utilizes mild conditions and widely available palladium catalysts, allowing for robust scale-up from gram to kilogram quantities without requiring extreme temperatures or pressures.
Q: Can the emission wavelength be tuned for specific display applications?
A: Yes, by modifying the electron-donating groups (A1, A2, A3) on the anthracene unit, the HOMO energy levels can be adjusted to fine-tune the emission spectrum within the blue light range (460 ± 20nm).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Blue Electroluminescent Material Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity organic semiconductors play in the advancement of next-generation display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like the triptycene-anthracene hybrids described in CN102464978B can be manufactured with consistent quality and reliability. We understand that the transition from lab-scale synthesis to industrial manufacturing requires rigorous process optimization, and our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards of the optoelectronics industry.
We invite you to collaborate with us to optimize your supply chain for advanced OLED materials. Whether you require custom synthesis of specific derivatives or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering expertise can accelerate your product development timeline while reducing overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
