Advanced Metal-Free Synthesis of Indoline-5-ylmethylamine Dihydrochloride for Commercial Agrochemical Production
Advanced Metal-Free Synthesis of Indoline-5-ylmethylamine Dihydrochloride for Commercial Agrochemical Production
The global demand for high-performance plant growth regulators continues to surge, driving the need for efficient synthetic routes to key precursors like Abscisic Acid (ABA) analogs. Patent CN113501777B introduces a groundbreaking four-step synthesis method for indoline-5-ylmethylamine dihydrochloride, a critical intermediate in this sector. This technology addresses long-standing inefficiencies in organic chemical synthesis by utilizing indole as a low-cost starting material and employing a metal-free catalytic system. The innovation lies in its ability to combine amidation and double-bond reduction in a single pot, drastically shortening the process route while maintaining high reaction activity. For industry leaders seeking a reliable agrochemical intermediate supplier, this patent represents a significant leap forward in process chemistry, offering a pathway to high-purity products without the burden of expensive transition metals or hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indoline-5-ylmethylamine derivatives has been plagued by significant technical and economic hurdles that hinder large-scale production. Prior art methods predominantly rely on 5-(bromomethyl)indoline as the primary raw material, a compound that is not only industrially scarce but also commands a prohibitively high price due to complex upstream synthesis requirements. Furthermore, the reduction of nitrile groups in these traditional routes typically necessitates the use of lithium aluminum hydride, a reagent known for its violent exothermic reaction upon contact with water. This inherent instability creates severe safety risks in a manufacturing environment, requiring specialized equipment and rigorous safety protocols that inflate operational costs. Additionally, the purification processes associated with these older methods often involve cumbersome chromatographic column separation, which consumes vast quantities of solvents and generates substantial chemical waste, contradicting modern green chemistry principles and increasing the environmental footprint of production.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data leverages a strategic redesign of the synthetic pathway to overcome these bottlenecks effectively. By initiating the synthesis with indole, a widely available and inexpensive commodity chemical, the method immediately lowers the barrier to entry for raw material procurement. The core innovation involves a one-pot reaction strategy where amidation and double-bond reduction occur simultaneously under acid reflux conditions, eliminating the need for intermediate isolation and reducing energy consumption. This streamlined process avoids the use of dangerous reducing agents entirely, replacing them with safer, more manageable reagents like hydrazine hydrate and formic acid. The result is a synthesis route that is not only shorter and more cost-effective but also inherently safer and easier to control, providing a robust foundation for cost reduction in agrochemical intermediate manufacturing while ensuring consistent product quality.
Mechanistic Insights into Metal-Free Friedel-Crafts Alkylation
The chemical elegance of this synthesis lies in its meticulous orchestration of reaction conditions to maximize yield without metal catalysts. The process begins with the protection of indole via formylation, followed by a critical Friedel-Crafts reaction where the protected indoline reacts with an amination reagent such as N-hydroxymethylphthalimide. This step is conducted in concentrated sulfuric acid at controlled low temperatures between -5°C and -10°C to manage the exothermic nature of the reaction and prevent side products. The subsequent deprotection using hydrazine hydrate in a mixed solvent system of dichloromethane and methanol ensures the selective removal of the phthalimide group while preserving the integrity of the indoline core. This precise control over reaction parameters allows for the formation of the target amine with high atom economy, demonstrating a sophisticated understanding of organic reactivity that is essential for producing high-purity agrochemical intermediates.
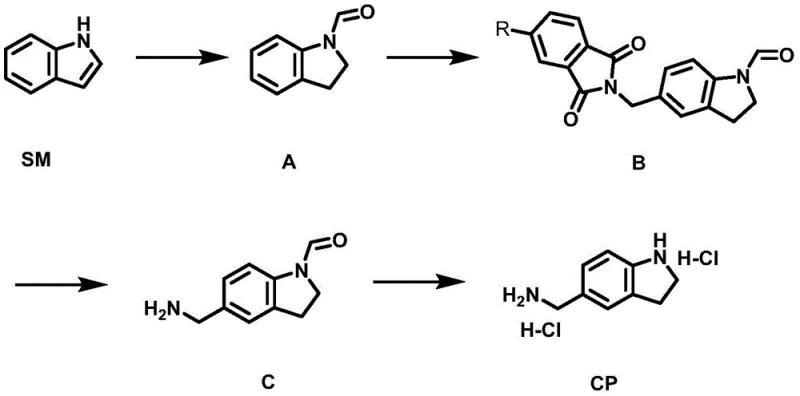
Impurity control is another cornerstone of this mechanistic design, ensuring that the final product meets stringent quality specifications required for biological applications. The patent highlights that the reaction products can be induced to precipitate directly during the post-treatment phase, which significantly simplifies purification. By avoiding chromatographic methods and relying on recrystallization techniques using solvents like toluene or dichloromethane, the process minimizes the retention of organic impurities. The use of specific molar ratios, such as 1:5 to 1:8 for intermediate B to hydrazine hydrate, is optimized to drive the reaction to completion while suppressing the formation of by-products. This focus on impurity management ensures that the commercial scale-up of complex agrochemical intermediates can proceed without the risk of batch failure due to contamination, thereby guaranteeing supply continuity for downstream pharmaceutical and agricultural clients.
How to Synthesize Indoline-5-ylmethylamine Dihydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the optimized conditions outlined in the patent to achieve the reported high yields and purity levels. The process is divided into four distinct stages, beginning with the reflux of indole in a high-boiling solvent like 1,3-dimethyl-2-imidazolidinone, followed by the low-temperature alkylation and subsequent deprotection steps. Each stage has been fine-tuned to balance reaction kinetics with safety, ensuring that the transformation from raw material to final dihydrochloride salt is both efficient and reproducible. For research and development teams looking to adopt this technology, the detailed standardized synthesis steps provided in the patent serve as a critical blueprint for laboratory validation and pilot plant trials. The following guide outlines the procedural framework necessary to replicate this success in a controlled environment.
- React indole with formic acid in a high-boiling solvent like 1,3-dimethyl-2-imidazolidinone at 226°C to achieve one-pot amidation and reduction, yielding formyl-protected indoline.
- Perform a Friedel-Crafts reaction using the intermediate and N-hydroxymethylphthalimide in concentrated sulfuric acid at -5°C to -10°C to introduce the aminomethyl group.
- Deprotect the phthalimide group using hydrazine hydrate in a dichloromethane and methanol mixed solvent system at room temperature to obtain the free amine intermediate.
- Finalize the synthesis by reacting the amine intermediate with HCl in isopropanol under reflux conditions to crystallize the target indoline-5-ylmethylamine dihydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis method offers transformative benefits for procurement managers and supply chain directors focused on efficiency and cost stability. The elimination of expensive transition metal catalysts and scarce starting materials directly translates to a more resilient supply chain that is less vulnerable to market fluctuations in raw material pricing. By simplifying the purification process and removing the need for column chromatography, the method significantly reduces solvent consumption and waste disposal costs, contributing to a leaner manufacturing operation. These structural improvements in the synthesis route provide a competitive edge, allowing manufacturers to offer high-quality intermediates at more attractive price points while maintaining healthy margins. The robustness of the process also means that production schedules are more predictable, reducing lead time for high-purity agrochemical intermediates and ensuring that customer demands are met consistently.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the substitution of costly reagents with commodity chemicals and the removal of metal catalysts. Traditional methods often incur hidden costs associated with the removal of trace heavy metals to meet regulatory standards, a step that is entirely unnecessary here. Furthermore, the one-pot nature of the initial reaction step reduces energy consumption by combining multiple transformation stages into a single heating cycle. This consolidation of steps not only saves on utility costs but also reduces the labor hours required for monitoring and handling, leading to substantial cost savings in agrochemical intermediate manufacturing without compromising on the quality of the final output.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the use of indole as the starting material, which is produced on a massive global scale and is readily available from multiple vendors. Unlike the scarce 5-(bromomethyl)indoline used in older methods, indole does not suffer from supply bottlenecks, ensuring that production can continue uninterrupted even during market shortages. Additionally, the avoidance of hazardous reagents like lithium aluminum hydride simplifies logistics and storage requirements, as there is no need for specialized containment or transport protocols for dangerous goods. This ease of sourcing and handling enhances the overall reliability of the supply chain, making it easier for partners to plan long-term production schedules with confidence.
- Scalability and Environmental Compliance: The design of this synthesis route is inherently scalable, utilizing standard industrial unit operations such as reflux, crystallization, and filtration that are easily adapted from laboratory to plant scale. The process avoids high-pressure reactions and extreme temperatures beyond standard reflux conditions, reducing the engineering complexity required for scale-up. From an environmental standpoint, the reduction in solvent usage and the elimination of heavy metal waste align with increasingly strict global environmental regulations. This compliance reduces the risk of regulatory penalties and facilitates smoother audits, ensuring that the commercial scale-up of complex agrochemical intermediates can proceed without environmental hindrances while promoting a greener manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of indoline-5-ylmethylamine dihydrochloride, based on the detailed data provided in the patent documentation. These answers are designed to clarify the operational benefits and technical feasibility of the method for potential partners and stakeholders. Understanding these nuances is crucial for making informed decisions about integrating this technology into existing production lines or sourcing strategies. The insights provided here reflect the core innovations of the patent, focusing on safety, cost, and scalability.
Q: Why is this synthesis method safer than conventional routes for indoline derivatives?
A: Conventional methods often rely on lithium aluminum hydride, which is explosively reactive with water, and expensive bromomethyl starting materials. This patented route eliminates dangerous reducing agents and uses stable, commercially available indole, significantly enhancing operational safety and reducing hazard management costs.
Q: How does the metal-free catalyst system impact production costs?
A: By avoiding expensive transition metal catalysts, the process eliminates the need for complex and costly heavy metal removal steps during purification. This simplification directly reduces raw material expenses and waste treatment costs, leading to substantial overall manufacturing savings.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It utilizes common industrial solvents like dichloromethane and methanol, avoids high-pressure equipment, and features robust crystallization steps that facilitate easy product isolation, making it highly viable for commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoline-5-ylmethylamine Dihydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation agrochemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one described in Patent CN113501777B can be seamlessly transitioned from the lab to the market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of indoline-5-ylmethylamine dihydrochloride meets the exacting standards required for biological efficacy. We are committed to leveraging our technical expertise to optimize this metal-free synthesis, delivering a product that supports your R&D goals while enhancing your supply chain efficiency.
We invite you to collaborate with us to explore how this advanced synthesis method can benefit your specific product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating the tangible economic value of switching to this superior route. We encourage you to reach out to request specific COA data and route feasibility assessments, allowing you to validate the quality and compatibility of our intermediates with your downstream processes. By partnering with us, you gain access to a reliable supply of high-performance chemicals backed by decades of industry expertise and a commitment to continuous innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
