Advanced Lycorine Derivatives: Scalable Synthesis for High-Purity Antitumor Intermediates
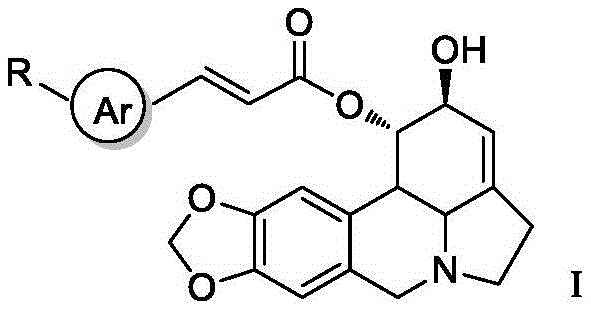
The pharmaceutical landscape is constantly evolving in the search for more effective antitumor agents, and recent advancements in natural product modification have yielded promising candidates. Patent CN113416199B introduces a novel series of lycorine beta-aryl acrylate derivatives that demonstrate exceptional potential in oncology applications. These compounds are structurally engineered by modifying the 2-hydroxyl position of the lycorine alkaloid core, a strategic change that significantly amplifies their biological efficacy against various human tumor cell lines. The innovation lies not just in the biological activity but in the robust chemical architecture that allows for diverse substitution patterns on the aryl ring, enabling fine-tuning of pharmacokinetic properties. For R&D directors and procurement specialists, this represents a critical opportunity to access high-purity pharmaceutical intermediates that bridge the gap between natural product discovery and clinical viability. The structural versatility offered by this patent provides a solid foundation for developing next-generation targeted therapies with improved safety profiles.
The development of these derivatives addresses a significant need in the industry for reliable pharmaceutical intermediates supplier solutions that can deliver complex molecules with consistent quality. By leveraging the inherent bioactivity of lycorine and enhancing it through synthetic organic chemistry, this technology offers a pathway to drugs that may overcome resistance mechanisms seen with traditional chemotherapy agents. The detailed structural elucidation provided in the patent ensures that manufacturing partners can replicate the synthesis with high fidelity, reducing the risk of batch-to-batch variability. This level of documentation is crucial for regulatory compliance and accelerates the timeline from preclinical research to investigational new drug applications. Furthermore, the broad scope of substituents allowed on the aryl ring means that a single platform technology can generate a library of candidates, maximizing the return on investment for pharmaceutical developers seeking to diversify their oncology pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to utilizing lycorine in antitumor therapy have often been hampered by limitations in potency and selectivity, which restrict their clinical utility despite their natural origin. Unmodified lycorine, while possessing inherent antiviral and anti-inflammatory properties, frequently requires high concentrations to achieve therapeutic effects against aggressive tumor cell strains, leading to potential off-target toxicity. Conventional modification strategies often lack the precision to target specific molecular sites on the complex alkaloid skeleton without affecting other critical functional groups, resulting in complex mixtures that are difficult to purify. This lack of chemoselectivity increases production costs and complicates the scale-up process, making it challenging for supply chain heads to secure consistent volumes of material. Additionally, older synthetic routes may rely on harsh conditions or expensive catalysts that are not sustainable for long-term commercial manufacturing, creating bottlenecks in the supply of cost reduction in pharmaceutical intermediates manufacturing. The inability to systematically vary the side chains limits the ability to optimize the drug-like properties necessary for oral bioavailability and metabolic stability.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles by employing a highly selective esterification strategy at the 2-hydroxyl position, preserving the integrity of the rest of the molecule while introducing potent pharmacophores. This method allows for the introduction of beta-aryl acrylate groups which have been shown to significantly enhance cytotoxicity against a wide range of cancer types, including lung, liver, and gastric cancers. The use of a protecting group strategy ensures that the reaction proceeds with high regioselectivity, minimizing the formation of by-products and simplifying the downstream purification process. This precision translates directly into higher yields and reduced waste, aligning with the goals of green chemistry and environmental compliance in modern chemical manufacturing. By enabling the use of diverse aryl acrylic acids, this approach provides a modular platform for rapid structure-activity relationship studies, allowing researchers to quickly identify lead compounds with optimal therapeutic indices. The robustness of this synthetic route makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates, ensuring that supply can meet the demands of clinical trials and eventual market launch.
Mechanistic Insights into Esterification and Protecting Group Strategy
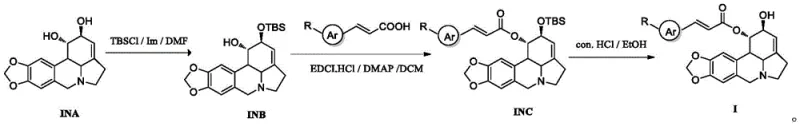
The core of this synthetic innovation lies in the meticulous control of reactivity through the use of tert-butyldimethylsilyl (TBS) protecting groups, which shield the 2-hydroxyl moiety during intermediate steps to prevent unwanted side reactions. This protection is critical because the lycorine skeleton contains multiple reactive sites that could otherwise interfere with the esterification process, leading to a mixture of regioisomers that are difficult to separate. By temporarily masking the hydroxyl group, chemists can direct the coupling reaction specifically to the desired position using standard carbodiimide coupling agents like EDCI in the presence of DMAP. This catalytic system facilitates the formation of the ester bond under mild conditions, preserving the stereochemistry of the chiral centers within the lycorine core which are essential for biological activity. The subsequent deprotection step using concentrated hydrochloric acid is equally important, as it cleanly removes the silyl group without degrading the sensitive ester linkage or the alkaloid framework. This sequence of protection, coupling, and deprotection exemplifies a classic yet highly effective strategy in complex molecule synthesis that ensures high purity and structural fidelity.
Understanding the structure-activity relationship (SAR) is vital for R&D teams aiming to optimize these compounds for specific therapeutic indications. The patent data indicates that the nature of the substituents on the aryl ring of the acrylate moiety plays a pivotal role in determining the antitumor potency. Electron-withdrawing groups such as halogens and nitro groups at specific positions on the phenyl ring have been observed to enhance the inhibitory activity against tumor cell lines compared to electron-donating groups. This suggests that the electronic properties of the side chain influence the interaction of the molecule with its biological target, possibly affecting binding affinity or cellular uptake. Furthermore, the steric bulk of the substituents can impact the conformation of the molecule, potentially optimizing its fit within the active site of the target enzyme or receptor. By systematically varying these substituents, researchers can fine-tune the pharmacological profile of the derivatives, balancing potency with safety and metabolic stability. This deep mechanistic understanding allows for the rational design of next-generation analogs that could overcome drug resistance mechanisms prevalent in current cancer therapies.
How to Synthesize Lycorine Beta-Aryl Acrylate Efficiently
The synthesis of these high-value intermediates follows a streamlined three-step protocol that is amenable to both laboratory scale and industrial production. The process begins with the selective protection of lycorine, followed by the coupling with the appropriate beta-aryl acrylic acid, and concludes with the removal of the protecting group to reveal the final active pharmaceutical ingredient. Each step has been optimized to maximize yield and minimize impurity formation, ensuring that the final product meets the rigorous quality standards required for pharmaceutical applications. The use of common reagents and solvents throughout the sequence simplifies the procurement process and reduces the overall cost of goods sold. Detailed operational parameters regarding temperature, reaction time, and workup procedures are provided to ensure reproducibility across different manufacturing sites. This standardized approach reduces the technical risk associated with technology transfer and enables rapid scaling to meet commercial demand. For detailed step-by-step instructions and specific reaction conditions, please refer to the technical guide below.
- Selectively protect the 2-hydroxyl group of lycorine using tert-butyldimethylsilyl chloride to form intermediate INB.
- Perform esterification between intermediate INB and beta-aryl acrylic acid using EDCI and DMAP to obtain intermediate INC.
- Remove the silane protecting group from intermediate INC using concentrated hydrochloric acid to yield the final lycorine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for oncology intermediates. The reliance on readily available starting materials such as lycorine and substituted cinnamic acids ensures a stable supply chain that is not dependent on scarce or geographically restricted resources. This availability mitigates the risk of supply disruptions and allows for better long-term planning of production schedules. Furthermore, the synthetic steps utilize standard organic transformations that do not require specialized equipment or exotic catalysts, making it easy for multiple qualified manufacturers to produce the material. This competition among suppliers can drive down costs and improve service levels, providing significant cost reduction in pharmaceutical intermediates manufacturing. The high selectivity of the reaction also means that less raw material is wasted in the form of by-products, contributing to a more sustainable and economically efficient process. These factors combined create a resilient supply chain capable of supporting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts or high-pressure equipment, relying instead on cost-effective organic reagents that are widely available in the bulk chemical market. This simplification of the process chemistry directly translates to lower operational expenditures and reduced capital investment requirements for manufacturing facilities. Additionally, the high yield and purity achieved through the protecting group strategy minimize the need for extensive purification steps, further reducing solvent consumption and waste disposal costs. By streamlining the production process, manufacturers can achieve substantial cost savings that can be passed on to the end client or reinvested into further R&D efforts. The economic efficiency of this route makes it a highly attractive option for large-scale production of antitumor agents.
- Enhanced Supply Chain Reliability: The use of commodity chemicals as starting materials ensures that the supply chain is robust and less susceptible to fluctuations in the availability of specialized precursors. Lycorine can be sourced from natural extracts or synthetic routes, providing multiple avenues for securing raw materials in case of disruptions in one supply line. The modular nature of the synthesis allows for the easy substitution of different aryl acrylic acids without changing the core process, enabling flexible production planning based on market demand for specific derivatives. This flexibility is crucial for maintaining continuity of supply in the face of changing regulatory requirements or shifts in therapeutic trends. A reliable supply of high-quality intermediates is essential for keeping clinical trials on schedule and ensuring timely market entry for new drugs.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at atmospheric pressure, which simplifies the engineering requirements for scaling up from kilogram to tonne quantities. This ease of scale-up reduces the time and cost associated with process validation and regulatory approval for commercial manufacturing. Moreover, the process generates minimal hazardous waste compared to traditional methods that might use heavy metals or toxic solvents, aligning with increasingly stringent environmental regulations. The ability to produce these compounds in an environmentally friendly manner enhances the corporate social responsibility profile of the manufacturing partner and reduces the risk of regulatory penalties. Sustainable manufacturing practices are becoming a key differentiator in the pharmaceutical industry, and this route positions the product favorably in that regard.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these lycorine derivatives. They are based on the specific technical disclosures and experimental data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this technology into their development pipelines. The answers reflect the current state of knowledge regarding the stability, activity, and synthesis of these compounds. For more specific technical data or custom synthesis requests, direct consultation with the technical team is recommended.
Q: What is the primary structural modification in these lycorine derivatives?
A: The primary modification involves the esterification of the 2-hydroxyl group of the lycorine core with various beta-aryl acrylic acid moieties, which significantly enhances antitumor potency compared to the parent compound.
Q: How does the synthesis route ensure high purity for pharmaceutical use?
A: The route utilizes a selective protection-deprotection strategy using TBSCl, which prevents side reactions at other hydroxyl positions, ensuring the final product meets stringent purity specifications required for clinical applications.
Q: Are the starting materials for this synthesis commercially available?
A: Yes, the starting material lycorine and the various substituted beta-aryl acrylic acids are readily available from standard chemical suppliers, facilitating a reliable and continuous supply chain for manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lycorine Beta-Aryl Acrylate Supplier
NINGBO INNO PHARMCHEM stands ready to support your development efforts with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in the nuances of alkaloid chemistry and protecting group strategies, ensuring that every batch of lycorine beta-aryl acrylate derivatives meets stringent purity specifications. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every compound we produce. Our commitment to quality and reliability makes us the preferred partner for pharmaceutical companies seeking to advance their oncology portfolios. We understand the critical importance of timeline and consistency in drug development and are dedicated to delivering materials that enable your success.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can assist in optimizing your supply chain. Request a Customized Cost-Saving Analysis to understand how our manufacturing capabilities can reduce your overall project costs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to meet your exacting standards. Let us help you accelerate your path to clinical success with our high-quality pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
