Advanced Bisphenanthroline Amine Ligand Synthesis for Commercial Scale-up of Complex Coordination Compounds
The chemical landscape for advanced functional materials is constantly evolving, driven by the need for more efficient and versatile transition metal complexes. Patent CN116178396A introduces a significant breakthrough in this domain with the disclosure of a novel bisphenanthroline amine transition metal ion ligand, specifically N-(1,10-phenanthroline-2-yl)-N-(2-(pyridin-2-yl)ethyl)-1,10-phenanthroline-2-amine. This compound, characterized by the molecular formula C31H22N6 and a molecular weight of 478.56, represents a critical advancement in ligand design for coordinating 3d and 4d transition metal ions. The synthesis methodology outlined in the patent addresses long-standing limitations in the availability of diverse phenanthroline-based ligands, which are essential for applications ranging from semiconductor materials to homogeneous catalysis. By providing a robust and reproducible preparation method, this technology opens new avenues for the development of high-performance metal complexes that are vital for modern industrial processes. The strategic importance of this molecule lies in its ability to form stable coordination bonds, thereby enhancing the physicochemical properties of the resulting metal complexes for use in high-value sectors such as sustainable energy and biological detection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-phenanthroline ligands has been fraught with challenges that hinder their widespread commercial adoption and application in high-precision industries. Conventional methods often suffer from limited structural diversity, restricting the types of transition metal ions that can be effectively complexed for specific industrial tasks. Many existing synthetic routes rely on harsh reaction conditions or expensive catalysts that are difficult to remove, leading to impurities that can compromise the performance of the final metal complex in sensitive applications like solar cell manufacturing. Furthermore, the scalability of traditional phenanthroline derivative synthesis is often poor, with low yields and complex purification steps that drive up production costs significantly. The lack of readily available ligands capable of forming stable complexes with a broad range of 3d and 4d transition metals has created a bottleneck in the development of new functional materials. These limitations not only slow down research and development cycles but also increase the supply chain risk for manufacturers who rely on consistent, high-purity ligand supplies for their production lines.
The Novel Approach
The methodology presented in patent CN116178396A offers a transformative solution to these persistent challenges by introducing a streamlined two-step synthesis route that prioritizes both efficiency and structural integrity. This novel approach utilizes readily available starting materials, specifically 2-chloro-1,10-phenanthroline and 2-(2-pyridyl)ethylamine, to construct the complex bisphenanthroline amine framework with high precision. The process is designed to operate under inert gas protection, which minimizes side reactions and ensures the formation of the desired product with minimal impurity profiles. By avoiding the use of exotic or prohibitively expensive reagents, this method significantly lowers the barrier to entry for large-scale production, making it an attractive option for cost reduction in specialty chemical manufacturing. The ability to synthesize this ligand with a defined molecular structure allows for predictable complexation behavior, which is crucial for R&D directors seeking to optimize the performance of transition metal catalysts. This new route effectively solves the problem of limited ligand variety, enabling the creation of tailored metal complexes that meet the rigorous demands of modern electronic and pharmaceutical applications.
Mechanistic Insights into Bisphenanthroline Amine Ligand Formation
The formation of the N-(1,10-phenanthroline-2-yl)-N-(2-(pyridin-2-yl)ethyl)-1,10-phenanthroline-2-amine ligand involves a sophisticated sequence of nucleophilic substitutions and coupling reactions that require precise control over reaction parameters. The first step entails the reaction of 2-chloro-1,10-phenanthroline with 2-(2-pyridyl)ethylamine in a toluene solvent system, heated to 120°C for 24 hours under an inert atmosphere. This thermal activation facilitates the displacement of the chlorine atom by the amine group, forming a stable intermediate known as N-(2-(pyridin-2-yl)ethyl)-1,10-phenanthroline-2-amine. The second step is even more critical, involving the deprotonation of the intermediate amine using sodium hydride, followed by coupling with a second equivalent of 2-chloro-1,10-phenanthroline. This step constructs the final bisphenanthroline architecture, creating a rigid and electron-rich environment ideal for coordinating transition metal ions. The use of sodium hydride as a base is particularly advantageous as it drives the reaction to completion while minimizing the formation of unwanted by-products that could interfere with downstream metal complexation.
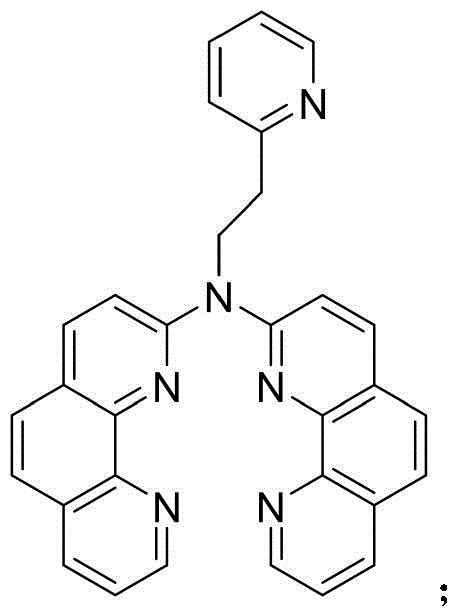
Controlling the impurity profile during this synthesis is paramount for ensuring the high purity required for electronic and pharmaceutical applications. The patent specifies a rigorous purification protocol that includes extraction with dichloromethane and subsequent silica gel column chromatography using a gradient elution of dichloromethane and methanol. This meticulous purification process is designed to remove unreacted starting materials, inorganic salts, and any side products formed during the high-temperature coupling steps. The resulting ligand exhibits a high degree of chemical homogeneity, which is essential for forming well-defined metal complexes with consistent physicochemical properties. For R&D directors, this level of purity assurance means that the ligand can be reliably used in sensitive catalytic cycles without the risk of catalyst poisoning or performance degradation. The structural integrity of the bisphenanthroline core ensures that the resulting metal complexes maintain their stability under various operational conditions, making them suitable for long-term use in industrial reactors or energy conversion devices.
How to Synthesize N-(1,10-phenanthroline-2-yl)-N-(2-(pyridin-2-yl)ethyl)-1,10-phenanthroline-2-amine Efficiently
Implementing this synthesis route in a commercial setting requires a clear understanding of the operational parameters and safety protocols associated with handling reactive reagents like sodium hydride. The process is designed to be scalable, moving from laboratory benchtop quantities to industrial production volumes without compromising on yield or quality. The initial step involves heating the reactants in toluene, a common industrial solvent, which simplifies the solvent recovery and recycling processes. The subsequent coupling reaction requires careful monitoring of gas evolution to ensure the complete consumption of the base before heating, which is a critical safety and quality control measure. Detailed standardized synthesis steps are essential for maintaining batch-to-batch consistency, which is a key requirement for qualifying as a reliable transition metal ligand supplier in the global market.
- React 2-chloro-1,10-phenanthroline with 2-(2-pyridyl)ethylamine in toluene at 120°C for 24 hours under inert gas to form the intermediate amine.
- Couple the intermediate with 2-chloro-1,10-phenanthroline using sodium hydride in toluene at 120°C for 4 hours, followed by extraction and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel ligand synthesis route offers substantial strategic benefits that extend beyond simple cost metrics. The streamlined nature of the two-step process reduces the overall complexity of the manufacturing workflow, which directly translates to enhanced supply chain reliability and reduced lead time for high-purity catalyst precursors. By utilizing common solvents like toluene and dichloromethane, the process leverages existing infrastructure in most fine chemical manufacturing facilities, eliminating the need for specialized equipment investments. This compatibility with standard industrial practices ensures that production can be ramped up quickly to meet fluctuating market demands, providing a buffer against supply disruptions. Furthermore, the high yield of the intermediate step and the robust purification protocol minimize waste generation, aligning with modern environmental compliance standards and reducing the burden on waste management systems.
- Cost Reduction in Manufacturing: The elimination of complex and expensive catalytic systems in the ligand synthesis itself leads to significant cost optimization in the production of these high-value intermediates. By avoiding the use of precious metal catalysts for the ligand construction, the process removes the need for costly metal removal and recovery steps, which are often major cost drivers in fine chemical synthesis. The use of sodium hydride, while requiring careful handling, is a cost-effective reagent that drives the reaction efficiently, reducing the overall consumption of raw materials per unit of product. This economic efficiency allows for more competitive pricing structures, enabling cost reduction in specialty chemical manufacturing for downstream users who incorporate these ligands into their final products. The simplified purification process also reduces solvent consumption and energy usage, further contributing to the overall economic viability of the production route.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-chloro-1,10-phenanthroline ensures a stable and secure supply chain that is less susceptible to raw material shortages. Unlike synthetic routes that depend on exotic or single-source reagents, this method utilizes commodity chemicals that are widely produced and traded in the global market. This diversity of supply sources mitigates the risk of production halts due to vendor-specific issues, ensuring continuous availability for critical applications in the semiconductor and pharmaceutical sectors. The robustness of the synthesis protocol also means that production can be easily transferred between different manufacturing sites without significant re-validation, providing flexibility in logistics and inventory management. This reliability is crucial for procurement managers who need to guarantee the uninterrupted flow of materials for their production lines.
- Scalability and Environmental Compliance: The synthesis route is inherently designed for scalability, with reaction conditions that can be safely managed in large-scale reactors using standard engineering controls. The use of toluene as the primary solvent allows for efficient distillation and recycling, minimizing the environmental footprint of the manufacturing process. The purification steps, while thorough, do not generate hazardous waste streams that require specialized disposal, simplifying compliance with environmental regulations. This ease of scale-up ensures that the commercial scale-up of complex coordination compounds can be achieved without encountering the typical bottlenecks associated with process intensification. The ability to produce large quantities of high-purity ligand supports the growing demand for advanced materials in renewable energy and electronics, positioning this technology as a sustainable choice for long-term industrial growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this bisphenanthroline amine ligand. These answers are derived directly from the patent data to ensure accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this ligand into existing product lines or research projects. The information provided covers key aspects such as molecular properties, reaction conditions, and potential applications, offering a comprehensive overview for stakeholders.
Q: What is the molecular weight of the synthesized ligand?
A: The molecular weight of the bisphenanthroline amine transition metal ion ligand is precisely 478.56 g/mol, with a molecular formula of C31H22N6.
Q: What are the critical reaction conditions for this synthesis?
A: The synthesis requires strict inert gas protection and heating in anhydrous toluene at 120°C, utilizing sodium hydride as a base in the second coupling step.
Q: What are the primary industrial applications of this ligand?
A: This ligand is designed for complexing 3d and 4d transition metal ions, finding utility in semiconductor materials, solar cell manufacturing, and homogeneous catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(1,10-phenanthroline-2-yl)-N-(2-(pyridin-2-yl)ethyl)-1,10-phenanthroline-2-amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ligands play in the advancement of transition metal chemistry and materials science. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to handle complex organic syntheses under inert conditions allows us to reproduce the high-quality standards outlined in patent CN116178396A, providing our partners with a reliable source of this advanced ligand. By partnering with us, you gain access to a supply chain that prioritizes consistency, quality, and technical support, enabling you to focus on your core innovation goals without worrying about material availability.
We invite you to collaborate with us to optimize your supply chain and unlock the full potential of this technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and process needs. We encourage you to reach out to request specific COA data and route feasibility assessments to ensure that this ligand meets your exact performance criteria. Whether you are developing new catalysts for sustainable energy or advanced materials for electronics, NINGBO INNO PHARMCHEM is equipped to support your journey with premium chemical solutions and expert guidance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
