Scalable Semi-Synthesis of 14β-Hydroxy-1,14-Carbonate-Deacetylbaccatin III for Global Pharma Supply Chains
The pharmaceutical industry's relentless pursuit of next-generation antineoplastic agents has placed a premium on the efficient synthesis of complex taxane intermediates. Patent CN1254470C introduces a groundbreaking preparation method for 14β-hydroxy-1,14-carbonate-deacetylbaccatin III, a pivotal precursor for novel taxane derivatives with enhanced pharmacological profiles. Historically, the reliance on natural extraction from scarce plant sources created severe bottlenecks in the supply chain for these high-value compounds. This patented technology fundamentally shifts the paradigm by utilizing 10-deacetylbaccatin III, an abundant and renewable starting material derived from Taxus baccata, thereby unlocking a scalable and economically viable pathway for global API manufacturing. For R&D directors and procurement strategists, this innovation represents a critical opportunity to secure a reliable pharmaceutical intermediate supplier partnership that mitigates raw material volatility while ensuring consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for accessing 14β-hydroxy-1,14-carbonate-deacetylbaccatin III derivatives have been plagued by inherent inefficiencies rooted in raw material scarcity. Prior art, such as EP 559,019, describes the isolation of the necessary 14β-hydroxy-deacetylbaccatin III precursor directly from the leaves of Taxus wallichiana. This natural source is geographically limited and yields only trace amounts of the target compound, rendering the process incapable of supporting commercial-scale demand. Furthermore, the extraction process is labor-intensive and subject to seasonal and agricultural variances, leading to unpredictable lead times and fluctuating costs. From a process chemistry perspective, relying on such low-abundance natural products complicates impurity profiling and batch-to-batch consistency, posing significant risks for regulatory filings and long-term supply continuity.
The Novel Approach
In stark contrast, the methodology disclosed in CN1254470C leverages 10-deacetylbaccatin III, which can be isolated in large quantities from the widely cultivated European yew (Taxus baccata). This strategic switch in starting materials transforms the economic landscape of taxane synthesis. The process initiates with the selective protection of the 7- and 10-hydroxyl groups, exploiting their differential reactivity to mask these positions while leaving the C-1 and C-13 hydroxyls free for subsequent functionalization. By employing trichloroacetyl or trialkylsilyl protecting groups, the synthesis achieves a level of chemoselectivity that was previously difficult to maintain. This approach not only bypasses the scarcity issues of the old method but also sets the stage for high-yielding downstream transformations, effectively addressing the need for cost reduction in API manufacturing.
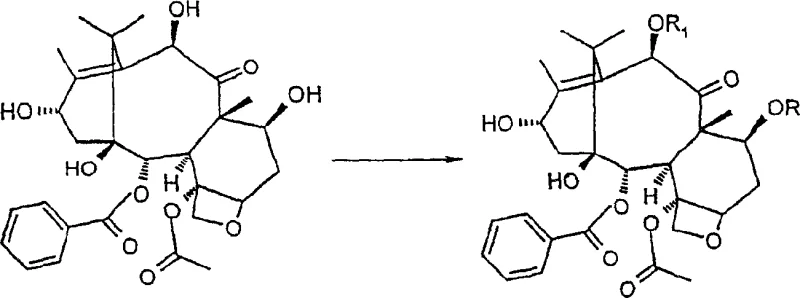
Mechanistic Insights into MnO2-Mediated Oxidation and Carbonate Formation
The core of this synthetic breakthrough lies in the precise oxidative functionalization of the taxane core. The protocol utilizes activated manganese dioxide (MnO2) to effect a two-step oxidation sequence. Initially, the reagent selectively oxidizes the C-13 hydroxyl group to a ketone. Under prolonged reaction conditions, typically extending up to 72 hours in acetonitrile, the system facilitates a further oxidation that introduces a hydroxyl group at the C-14 beta position. This transformation is mechanistically fascinating as it navigates the steric congestion of the taxane ring system to install oxygen functionality at a hindered bridgehead-adjacent carbon. The use of MnO2 is particularly advantageous due to its mildness, which prevents over-oxidation or degradation of the sensitive oxetane ring and ester side chains, ensuring the structural integrity required for high-purity pharmaceutical intermediates.
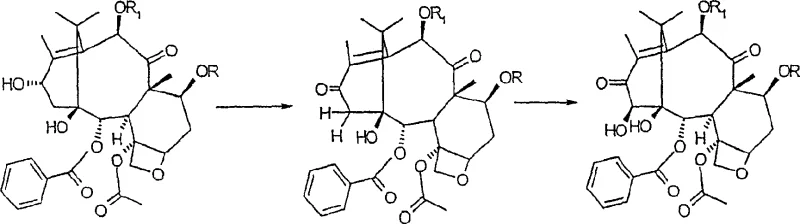
Following oxidation, the adjacent hydroxyl groups at the C-1 and C-14 positions undergo carbonate formation using phosgene or triphosgene in a dichloromethane and toluene mixture. This cyclization is critical for locking the conformation of the A-ring, which is essential for the biological activity of downstream derivatives. Subsequently, the C-13 carbonyl is reduced using sodium borohydride in methanol. This reduction step is highly regioselective, targeting the C-13 ketone while leaving the C-9 carbonyl untouched, and stereoselective, yielding almost exclusively the 13-alpha isomer. The final deprotection of the 7- and 10-positions reveals the target molecule. This sequence demonstrates a masterful control over chemo-, regio-, and stereoselectivity, minimizing the formation of diastereomeric impurities that would otherwise complicate purification.
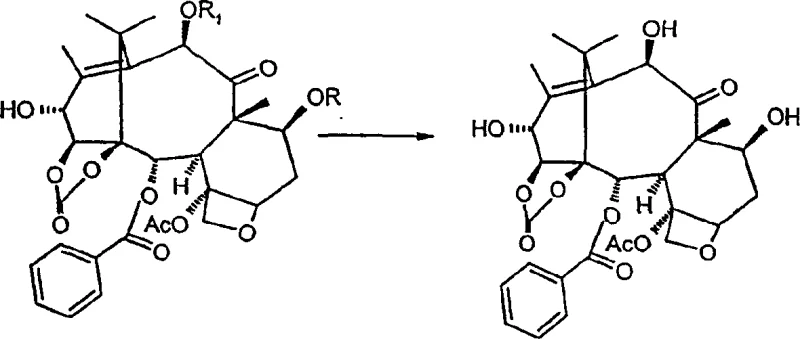
How to Synthesize 14β-Hydroxy-1,14-Carbonate-Deacetylbaccatin III Efficiently
Executing this synthesis requires careful attention to reaction conditions, particularly during the protection and oxidation phases. The patent outlines a preferred embodiment where 10-deacetylbaccatin III is reacted with trichloroacetyl chloride in dichloromethane, utilizing triethylamine and a catalytic amount of DMAP. This specific modification eliminates the need for pyridine as a solvent, which is a significant process improvement. The detailed standardized synthetic steps, including precise stoichiometry, temperature controls, and workup procedures for each of the five stages, are provided in the technical guide below to ensure reproducibility and safety in your pilot plant operations.
- Selective protection of the 7- and 10-hydroxyl groups of 10-Deacetylbaccatin III using trichloroacetyl or silyl reagents to differentiate reactivity.
- Two-step oxidation using activated manganese dioxide to introduce a carbonyl at C-13 and a hydroxyl group at the C-14 beta position.
- Formation of the 1,14-carbonate bridge followed by stereoselective reduction of the C-13 carbonyl and final deprotection to yield the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the transition to this semi-synthetic route offers profound strategic benefits beyond mere technical elegance. The primary advantage is the decoupling of production from scarce natural resources. By anchoring the supply chain on 10-deacetylbaccatin III, which is commercially available in metric ton quantities, manufacturers can guarantee commercial scale-up of complex diterpenes without the risk of crop failure or geopolitical supply disruptions associated with rare yew species. This stability translates directly into more predictable forecasting and inventory management, allowing pharmaceutical companies to plan long-term production schedules with confidence.
- Cost Reduction in Manufacturing: The elimination of pyridine in favor of a dichloromethane-based system catalyzed by DMAP significantly lowers solvent costs and waste disposal fees. Pyridine is not only expensive but also requires rigorous removal to meet residual solvent limits, adding extra distillation steps. The new method simplifies the workup, as the trichloroacetate protecting groups can be removed concurrently with the reduction step or under mild conditions, streamlining the overall process flow and reducing utility consumption.
- Enhanced Supply Chain Reliability: Sourcing 10-deacetylbaccatin III from Taxus baccata ensures a robust and continuous supply line. Unlike the extraction of precursors from Taxus wallichiana, which is limited by geography and biomass availability, the European yew is widely cultivated for ornamental and forestry purposes. This abundance means that reducing lead time for high-purity intermediates is achievable, as raw material procurement is no longer the critical path bottleneck. Suppliers can maintain higher safety stocks of the starting material, buffering against market fluctuations.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind. The use of common solvents like dichloromethane, ethyl acetate, and methanol facilitates easy solvent recovery and recycling. Furthermore, the avoidance of heavy metal catalysts and the use of manganese dioxide, which can be filtered and potentially regenerated, aligns with modern green chemistry principles. This reduces the environmental footprint of the manufacturing process, aiding in regulatory compliance and sustainability reporting for corporate stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in CN1254470C, providing clarity on process robustness and product quality attributes.
Q: Why is 10-Deacetylbaccatin III preferred over 14β-Hydroxybaccatin III as a starting material?
A: 10-Deacetylbaccatin III is abundantly available from the needles of Taxus baccata, whereas 14β-Hydroxybaccatin III must be extracted in low yields from the scarce leaves of Taxus wallichiana, making the former far more sustainable for commercial scale-up.
Q: How does the new process improve environmental compliance compared to conventional methods?
A: The novel protocol replaces pyridine, a solvent with significant residual emission concerns, with a dichloromethane and triethylamine system catalyzed by DMAP, drastically reducing hazardous waste and simplifying solvent recovery.
Q: What ensures the stereochemical purity at the C-13 position during reduction?
A: The reduction of the C-13 carbonyl is performed using sodium borohydride in methanol, which proceeds with high regioselectivity and stereoselectivity to almost exclusively yield the desired 13-alpha isomer while leaving the C-9 carbonyl intact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 14β-Hydroxy-1,14-Carbonate-Deacetylbaccatin III Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel oncology therapies depends on the reliability of the underlying chemical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 14β-hydroxy-1,14-carbonate-deacetylbaccatin III meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence ensures that your development timelines are met without compromise on quality.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your project economics. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential efficiencies this method offers for your specific portfolio. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, securing a competitive edge in the global taxane market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
