Scaling Advanced CA-4 Derivative Synthesis for Global Pharmaceutical Supply Chains
Scaling Advanced CA-4 Derivative Synthesis for Global Pharmaceutical Supply Chains
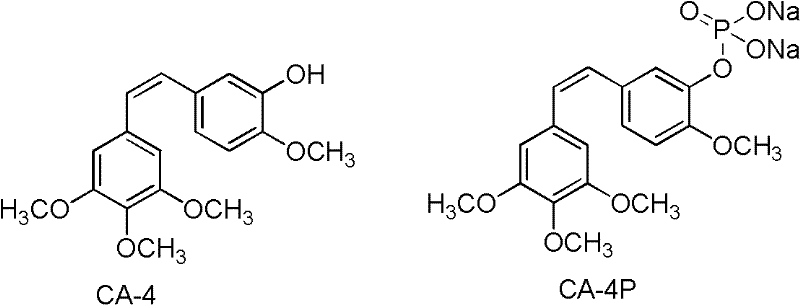
The pharmaceutical industry continuously seeks novel angiogenesis inhibitors to combat various cancers and chronic inflammatory diseases, and patent CN102219811A presents a significant breakthrough in this domain by disclosing a class of Combretastatin A-4 (CA-4) derivatives. These novel compounds address the critical limitation of poor water solubility inherent in the parent CA-4 molecule, which has historically restricted its clinical application despite potent anti-tumor activity. By conjugating the CA-4 core with specific amino sugar moieties through dicarboxylic acid linkers, this technology creates multi-target angiogenesis inhibitors with enhanced pharmacological profiles. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural versatility of these derivatives is paramount for integrating them into modern oncology pipelines. This report analyzes the technical feasibility and commercial viability of producing these high-value intermediates at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to utilizing Combretastatin A-4 have been severely hampered by its physicochemical properties, specifically its extremely low water solubility which complicates formulation and delivery. Conventional methods often rely on simple salt formation or basic prodrug strategies that may not provide sufficient stability or targeted release mechanisms required for effective tumor vascular disruption. Furthermore, existing microtubule inhibitors like vinblastine and paclitaxel, while effective, suffer from narrow anti-tumor spectrums and significant toxicity profiles that limit their therapeutic window. The reliance on these older generations of drugs creates a bottleneck in clinical development where resistance mechanisms quickly emerge, necessitating the search for compounds with different binding modes or improved pharmacokinetics. Without structural modification, the native CA-4 scaffold remains a promising but practically difficult candidate for widespread commercial deployment in solid dosage forms or injectables.
The Novel Approach
The innovative strategy outlined in the patent data involves a sophisticated molecular engineering approach where the hydroxyl group of the CA-4 molecule is linked to amino sugar molecules via succinic, malonic, glutaric, or adipic acid arms. This structural modification not only potentially enhances water solubility but also introduces a multi-target mechanism that inhibits human umbilical vein endothelial cell proliferation more effectively than the parent compound. By varying the length of the carbon chain linker and the specific configuration of the sugar moiety, chemists can fine-tune the metabolic stability and tissue distribution of the drug. This modularity allows for the creation of a diverse library of candidates from a common set of intermediates, providing a robust platform for drug discovery teams to optimize efficacy while managing toxicity. Such a approach represents a significant leap forward in designing next-generation vascular disrupting agents.
Mechanistic Insights into Amide Coupling and Linker Chemistry
The core of this synthesis relies on a precise amide coupling reaction between a carboxylic acid-functionalized CA-4 intermediate and various protected amino sugars. The process typically utilizes activating agents such as EDCI (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide) alongside HOBt (Hydroxybenzotriazole) in polar aprotic solvents like DMF. This activation strategy ensures high conversion rates while minimizing the risk of racemization at chiral centers, which is critical for maintaining the stereochemical integrity required for biological activity. The reaction proceeds under mild conditions, often at room temperature, which preserves the sensitive cis-stilbene geometry of the CA-4 core that is essential for tubulin binding. Understanding this mechanistic nuance is vital for process chemists aiming to replicate the high yields reported in the patent examples without degrading the valuable stilbene scaffold.
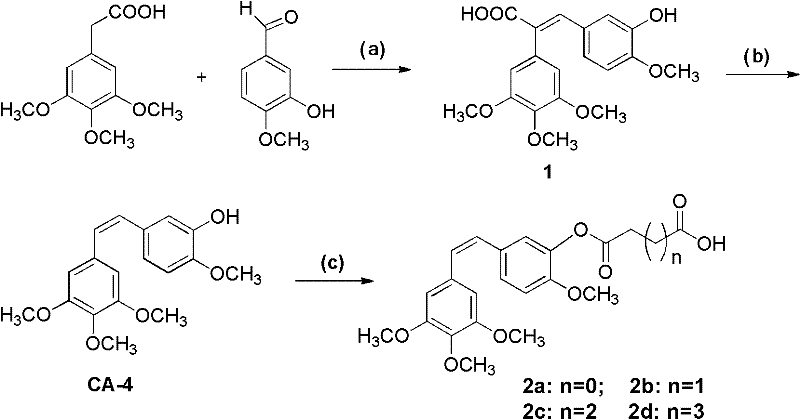
Impurity control in this synthesis is managed through the careful selection of protecting groups on the sugar moieties, such as acetyl groups, which can be selectively removed or retained depending on the desired final properties. The use of dicarboxylic anhydrides in the earlier steps to functionalize the CA-4 phenol group introduces a stable ester linkage that resists premature hydrolysis in circulation. This stability is crucial for ensuring that the active moiety reaches the tumor vasculature before metabolic cleavage occurs. Additionally, the purification protocols described involve standard silica gel chromatography and recrystallization techniques, indicating that the impurities generated are chemically distinct enough to be separated without requiring exotic or prohibitively expensive purification technologies. This predictability in impurity profiles greatly simplifies the regulatory path for these intermediates.
How to Synthesize CA-4 Derivatives Efficiently
The synthesis of these complex angiogenesis inhibitors follows a logical progression starting from readily available aromatic aldehydes and phenylacetic acids. The initial Perkin condensation establishes the carbon backbone, followed by a decarboxylation step to generate the stilbene core, and finally, the conjugation with the sugar component. Detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that ensure reproducibility. Operators must pay close attention to temperature control during the decarboxylation phase to prevent isomerization of the double bond. The following protocol outlines the critical parameters for achieving the high purity standards required for pharmaceutical applications.
- Perform Perkin condensation of trimethoxyphenylacetic acid with methoxybenzaldehyde using acetic anhydride and triethylamine.
- Execute decarboxylation using copper powder in quinoline at elevated temperatures to form the stilbene core.
- Couple the resulting acid intermediate with protected amino sugars using EDCI and HOBt in DMF solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described offers substantial advantages due to its reliance on commodity chemicals and established reaction types. The starting materials, including various methoxy-substituted benzaldehydes and dicarboxylic anhydrides, are widely available from global chemical suppliers, reducing the risk of raw material shortages. This abundance ensures that production schedules can be maintained without significant delays caused by sourcing bottlenecks. Furthermore, the reaction conditions do not require extreme pressures or cryogenic temperatures, allowing the process to be run in standard glass-lined or stainless steel reactors found in most multipurpose chemical manufacturing facilities. This compatibility with existing infrastructure significantly lowers the barrier to entry for contract manufacturing organizations looking to offer this technology.
- Cost Reduction in Manufacturing: The elimination of rare transition metal catalysts in the final coupling steps significantly reduces the cost burden associated with catalyst recovery and heavy metal removal. By utilizing organic coupling reagents like EDCI and HOBt, the process avoids the need for expensive scavengers or specialized filtration equipment required for palladium or copper residues. This simplification of the downstream processing directly translates to lower operational expenditures and a more favorable cost of goods sold. Additionally, the high yields reported in the patent examples suggest minimal waste generation, further enhancing the economic efficiency of the overall manufacturing campaign.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the stocking of key intermediates, such as the functionalized CA-4 acid, which can be rapidly converted into various final derivatives based on demand. This flexibility enables manufacturers to respond quickly to changing market needs without retooling entire production lines. The robustness of the chemical bonds formed during the synthesis ensures that the intermediates have a reasonable shelf life, facilitating inventory management and reducing the risk of spoilage during storage and transport. Such stability is a critical factor for maintaining a continuous supply to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The solvents employed in this process, such as ethyl acetate, acetone, and ethanol, are generally considered greener alternatives to chlorinated solvents and are easier to recover and recycle. This alignment with environmental best practices simplifies the permitting process for new production facilities and reduces the costs associated with waste disposal. The absence of highly toxic reagents also improves workplace safety profiles, lowering insurance premiums and regulatory compliance burdens. As the industry moves towards more sustainable manufacturing practices, this synthetic route positions itself as a forward-looking solution for large-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these CA-4 derivatives. They are derived from the specific technical disclosures and experimental data provided in the patent documentation. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their portfolios. The answers reflect the current state of the art as described in the intellectual property.
Q: What is the primary advantage of these CA-4 derivatives over the parent compound?
A: The primary advantage is significantly improved water solubility and bioavailability through sugar conjugation, addressing the clinical limitations of native CA-4.
Q: Are the reagents used in this synthesis pathway commercially scalable?
A: Yes, the pathway utilizes common industrial reagents such as succinic anhydride, triethylamine, and standard protecting groups, ensuring robust supply chain continuity.
Q: How does the amide coupling step impact impurity profiles?
A: The use of EDCI/HOBt mediation allows for mild reaction conditions that minimize racemization and side reactions, leading to a cleaner crude product profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable CA-4 Derivatives Supplier
NINGBO INNO PHARMCHEM stands ready to support your development programs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in handling sensitive stilbene chemistry and carbohydrate conjugations, ensuring that stringent purity specifications are met for every batch. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify the structural integrity and isotopic purity of these complex intermediates. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and regulatory compliance, mitigating the risks associated with early-stage process development.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Our engineers can provide a Customized Cost-Saving Analysis to demonstrate how optimizing this synthetic route can benefit your bottom line. Whether you need kilogram quantities for preclinical studies or metric tons for commercial launch, we have the capacity and capability to deliver. Let us help you accelerate your timeline to market with a reliable and efficient supply of these critical oncology intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
