Advanced Synthesis of Key Agrochemical Intermediates for Commercial Scale-Up and Cost Efficiency
The chemical industry is constantly evolving, driven by the need for more efficient and environmentally sustainable manufacturing processes. A significant advancement in this domain is detailed in patent CN101139322A, which outlines a novel preparation method for 5-amino-1-(2,6-dichloro-4-trifluoromethylphenyl)-3-cyanopyrazole. This compound serves as a critical intermediate in the synthesis of high-efficiency, broad-spectrum insecticides, specifically fluorine-containing nitrile derivatives. The traditional methods for producing this key molecular scaffold often involve complex multi-step procedures that generate substantial waste and incur high operational costs. By leveraging a streamlined diazotization and cyclization pathway, this technology addresses the pressing demands for cost reduction in agrochemical intermediate manufacturing. For R&D Directors and Procurement Managers alike, understanding the nuances of this synthesis is vital for securing a reliable agrochemical intermediate supplier capable of delivering high-purity materials consistently. The innovation lies not just in the chemical transformation but in the holistic optimization of reaction conditions, solvent usage, and post-processing steps, ensuring that the final product meets stringent quality specifications required for downstream pharmaceutical and agrochemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazole derivatives like the target compound has relied heavily on mixed acid systems, typically involving a combination of sulfuric acid and glacial acetic acid. While these methods can achieve the desired chemical transformation, they are plagued by significant inefficiencies that hinder commercial viability. The use of glacial acetic acid introduces a layer of complexity in the post-reaction workup, as separating the acetic acid from the sulfuric acid waste stream is energetically intensive and chemically challenging. This difficulty in recycling leads to the generation of large volumes of spent acid, which poses a severe environmental burden and increases disposal costs. Furthermore, the solubility characteristics of the reactants in these mixed systems often necessitate harsher reaction conditions, which can degrade the quality of the intermediate and lead to the formation of unwanted by-products. For a procurement manager, these inefficiencies translate directly into higher raw material costs and unpredictable supply chains. The inability to effectively recycle the acid medium means that every batch requires fresh inputs of expensive reagents, eroding profit margins and making the final insecticide product less competitive in the global market. Additionally, the environmental regulations surrounding the discharge of mixed acid waste are becoming increasingly stringent, forcing manufacturers to invest heavily in treatment facilities or face regulatory penalties.
The Novel Approach
In contrast, the method disclosed in the patent utilizes a simplified reaction medium consisting primarily of sulfuric acid with a concentration range of 40% to 98%. This strategic shift eliminates the need for glacial acetic acid entirely, thereby removing the primary source of separation difficulty and waste generation. By operating within this optimized acidic environment, the diazotization of 2,6-dichloro-4-trifluoromethylaniline proceeds with greater ease and control. The subsequent cyclization step, involving the addition of ethyl 2,3-dicyanopropionate under weakly alkaline conditions in an organic solvent, is designed to maximize yield while minimizing side reactions. This approach not only maintains the ease of reaction but also significantly improves the overall yield, with experimental data showing results consistently above 70%. For supply chain heads, this translates to a more robust and scalable process. The ability to recycle the sulfuric acid directly, without the contamination of acetic acid, drastically reduces the consumption of raw materials and lowers the overall production cost. This novel approach represents a paradigm shift towards greener chemistry, where economic efficiency and environmental stewardship go hand in hand, making it an attractive option for companies seeking a reliable agrochemical intermediate supplier.
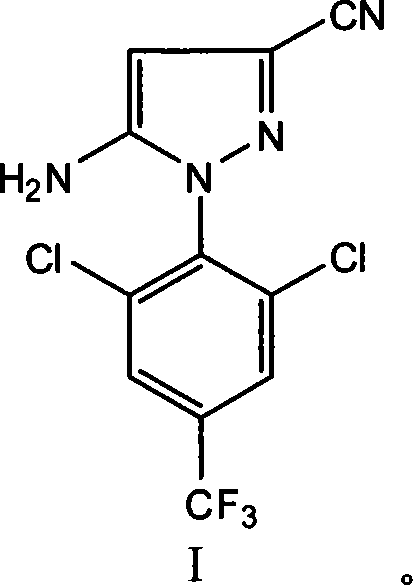
Mechanistic Insights into Diazotization and Cyclization
The core of this synthesis lies in the precise control of the diazotization reaction, which is the foundational step for constructing the pyrazole ring. The process begins with the dissolution of 2,6-dichloro-4-trifluoromethylaniline in sulfuric acid, where it forms a stable salt. The addition of sodium nitrite solution at controlled temperatures between 0°C and 70°C generates the diazonium salt in situ. The temperature control is critical here; too low, and the reaction kinetics are sluggish, while too high risks the decomposition of the unstable diazonium species. The patent specifies an optimal range of 20°C to 60°C for this step, ensuring a balance between reaction rate and stability. Once the diazotization is complete, the reaction mixture is cooled to 15°C to 30°C before the introduction of the cyclization agent, ethyl 2,3-dicyanopropionate. This temperature drop is essential to prevent premature side reactions and to ensure that the coupling occurs selectively at the desired position on the aromatic ring. The mechanistic pathway involves the nucleophilic attack of the active methylene group of the dicyanopropionate on the diazonium ion, followed by intramolecular cyclization to form the pyrazole core. This sequence is highly sensitive to the pH of the medium, which is why the subsequent step involves adjusting the pH to greater than 9 using ammonia water in an organic solvent. This alkaline environment facilitates the final ring closure and ensures the formation of the 5-amino-3-cyano substitution pattern.
Impurity control is another critical aspect of this mechanism that appeals to R&D Directors focused on purity and impurity profiles. The use of sulfuric acid as the sole acid medium minimizes the formation of acetylated by-products that are common in mixed acid systems. Furthermore, the specific molar ratios of reactants—1:1 to 1.2 for the aniline to sodium nitrite, and 1:1 to 1.1 for the aniline to dicyanopropionate—are tuned to ensure complete conversion of the starting material while minimizing excess reagents that could lead to impurities. The post-treatment process involves filtering off black solid materials, which are likely polymeric by-products or tarry residues, before washing the organic layer. This physical separation step is crucial for removing high-molecular-weight impurities that could affect the color and stability of the final product. The final recrystallization from toluene and normal hexane further purifies the compound, ensuring that the melting point falls within the narrow range of 140-142°C, indicative of high chemical purity. This rigorous control over the reaction mechanism and purification steps ensures that the resulting high-purity agrochemical intermediates meet the strict specifications required for the synthesis of active pharmaceutical ingredients and advanced agrochemicals.
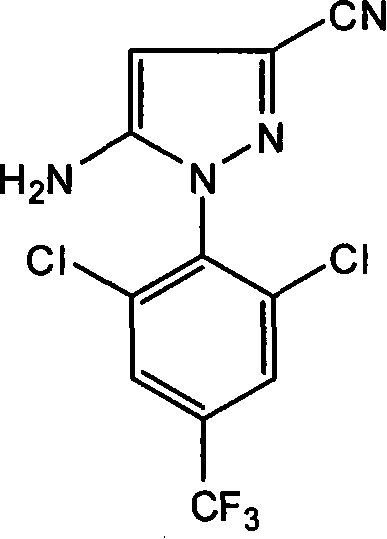
How to Synthesize 5-Amino-1-(2,6-Dichloro-4-Trifluoromethylphenyl)-3-Cyanopyrazole Efficiently
Implementing this synthesis route requires careful attention to the specific operational parameters outlined in the patent to ensure reproducibility and safety on a commercial scale. The process is designed to be straightforward, utilizing standard chemical engineering unit operations such as stirred tank reactors, extraction units, and crystallizers. The key to efficiency lies in the precise management of the acid concentration and the temperature profiles during the diazotization and cyclization phases. Operators must ensure that the sulfuric acid concentration is maintained within the 70% to 80% range for optimal results, as deviations can impact the solubility of the aniline salt and the stability of the diazonium intermediate. The addition of reagents must be controlled via metering pumps to maintain the exothermic nature of the reactions within safe limits. Following the reaction, the extraction process using solvents like chloroform, ethylene dichloride, or toluene is critical for isolating the product from the aqueous acid phase. The detailed standardized synthesis steps, including specific stirring times, washing protocols, and drying procedures, are essential for achieving the reported yields and purity levels. For technical teams looking to adopt this technology, adhering to these standardized protocols is the first step towards successful commercial scale-up of complex agrochemical intermediates.
- Diazotization of 2,6-dichloro-4-trifluoromethylaniline in 40-98% sulfuric acid with sodium nitrite at 0-70°C.
- Adjustment of reaction temperature to 15-30°C and addition of ethyl 2,3-dicyanopropionate.
- Cyclization in organic solvent under weakly alkaline conditions at 25-50°C followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers substantial benefits that extend beyond the laboratory bench. For procurement managers, the primary advantage is the significant reduction in raw material costs. By eliminating glacial acetic acid from the process, the bill of materials is simplified, and the reliance on volatile organic acids is removed. This not only lowers the direct cost of goods sold but also reduces the logistical burden associated with storing and handling corrosive acids. The ability to recycle the sulfuric acid medium further enhances cost efficiency, as the spent acid can be reconcentrated and reused in subsequent batches, creating a closed-loop system that minimizes waste. This aligns perfectly with the goal of cost reduction in agrochemical intermediate manufacturing, allowing companies to offer more competitive pricing to their downstream customers. Moreover, the simplified waste stream means that environmental compliance costs are drastically reduced. There is no need for complex separation units to recover acetic acid, and the volume of hazardous waste requiring disposal is significantly lower. This translates to lower operational expenditures and a smaller environmental footprint, which is increasingly important for maintaining a social license to operate in the chemical industry.
- Cost Reduction in Manufacturing: The elimination of glacial acetic acid and the ability to recycle sulfuric acid directly lead to a streamlined cost structure. Without the need for expensive acid recovery systems or the purchase of additional neutralizing agents for acetic acid, the overall manufacturing overhead is reduced. This qualitative improvement in process efficiency allows for better margin protection even in fluctuating raw material markets. The reduction in solvent consumption, achieved through optimized extraction ratios, further contributes to lower variable costs per kilogram of product. By focusing on reagent efficiency and waste minimization, the process inherently drives down the cost of production without compromising on quality.
- Enhanced Supply Chain Reliability: The use of common, readily available solvents like toluene and chloroform ensures that the supply chain is not vulnerable to shortages of specialized reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations, means that production schedules are less likely to be disrupted by minor process variations. This reliability is crucial for reducing lead time for high-purity agrochemical intermediates, as it allows for consistent batch-to-batch turnover. Suppliers can maintain higher inventory levels with confidence, knowing that the process is stable and scalable. This stability fosters stronger partnerships between manufacturers and their clients, as the risk of supply interruption is minimized.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard equipment that can be easily scaled from pilot plant to full commercial production. The reduction in "three wastes" (waste gas, waste water, and waste residue) simplifies the environmental permitting process and reduces the ongoing cost of waste treatment. The ability to handle the reaction in standard stainless steel or glass-lined reactors without the need for exotic materials of construction further facilitates scale-up. This environmental compliance ensures long-term sustainability, protecting the supply chain from future regulatory tightening and ensuring continuous operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this critical intermediate. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their broader supply chain strategies. The focus is on practical implementation, quality assurance, and the tangible benefits of adopting this optimized synthetic route.
Q: What are the primary advantages of using sulfuric acid over mixed acid systems in this synthesis?
A: Using sulfuric acid as the sole medium eliminates the need for glacial acetic acid, significantly reducing raw material costs and simplifying the recovery of spent acid, which enhances environmental compliance.
Q: How does this method improve yield compared to conventional techniques?
A: The optimized conditions, specifically the controlled temperature range during diazotization and the specific molar ratios, allow for yields exceeding 70%, which is a substantial improvement over older methods that suffered from lower efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes common solvents like chloroform or toluene and avoids complex catalysts, making it highly scalable and easier to handle in large reactor volumes compared to laboratory-only protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Amino-1-(2,6-Dichloro-4-Trifluoromethylphenyl)-3-Cyanopyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our expertise in CDMO services allows us to adapt complex synthetic pathways like the one described in CN101139322A to meet your specific volume and quality requirements. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5-amino-1-(2,6-dichloro-4-trifluoromethylphenyl)-3-cyanopyrazole meets the highest industry standards. We understand that consistency is key in the agrochemical and pharmaceutical sectors, and our robust quality management systems are designed to deliver exactly that.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that can help you make informed decisions about your sourcing strategy. Whether you are looking to secure a long-term supply agreement or need support with process development, we are committed to being your trusted partner in chemical manufacturing. Contact us today to discuss how we can support your project with our advanced synthesis capabilities and dedication to excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
