Advanced Synthesis of Broad-Spectrum Antibacterial Triazole Intermediates for Commercial Scale-Up
The pharmaceutical industry is currently facing a critical challenge with the rising prevalence of drug-resistant fungal strains, particularly Candida krusei, which has shown significant resistance to conventional treatments like fluconazole. In response to this urgent medical need, patent CN102199128A introduces a novel class of high-efficiency antibacterial triazole compounds designed to overcome these limitations through structural innovation. The core technology involves the organic combination of o-hydroxyphenyl and 1,2,3-triazole molecular fragments, resulting in 1-substituted phenyl-4-substituted phenyl-5-(2-hydroxyl substituted benzyl) amino-1,2,3-triazole derivatives. These compounds have demonstrated exceptional broad-spectrum bactericidal effects, particularly against Candida albicans and Escherichia coli, while maintaining a low toxicity profile. For procurement and R&D teams seeking reliable pharmaceutical intermediates supplier partnerships, this technology represents a significant opportunity to diversify pipelines with next-generation antifungal agents that meet stringent efficacy requirements.
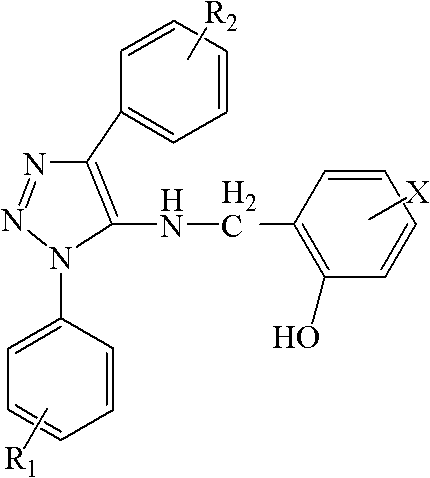
The structural versatility of these compounds allows for extensive optimization of biological activity by varying substituents such as halogens, methyl groups, or methoxy groups at specific positions on the phenyl rings. This modularity is crucial for medicinal chemists aiming to fine-tune pharmacokinetic properties without altering the core scaffold. Furthermore, the synthesis pathway described in the patent is notably concise, avoiding the multi-step complexities often associated with heterocyclic drug synthesis. By leveraging a straightforward five-step sequence starting from readily available functionalized anilines, manufacturers can achieve high yields while adhering to green chemistry principles. This efficiency translates directly into cost reduction in pharmaceutical intermediates manufacturing, making it an attractive candidate for commercial scale-up of complex polymer additives or active pharmaceutical ingredients where supply chain stability is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing triazole-based antifungals often rely on cumbersome cyclization reactions that require harsh conditions, expensive catalysts, or difficult-to-remove heavy metal residues. Many existing processes involve the use of hydrazine hydrate or other hazardous reagents that pose significant safety risks during large-scale production. Additionally, conventional routes frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult to separate, thereby reducing overall yield and increasing waste disposal costs. The reliance on transition metal catalysts also introduces the risk of metal contamination in the final API, necessitating additional purification steps that drive up production expenses. For supply chain heads, these factors create vulnerabilities in terms of lead time and regulatory compliance, as removing trace metals to meet ICH Q3D guidelines can be a bottleneck in the manufacturing process.
The Novel Approach
The methodology outlined in CN102199128A offers a transformative alternative by utilizing a diazotization-azidation-cyclization strategy that eliminates the need for transition metal catalysts entirely. This approach begins with the conversion of substituted anilines into diazonium salts, followed by azidation to form substituted azidobenzenes, which then undergo a thermal cyclization with phenylacetonitriles. This sequence not only simplifies the reaction workflow but also ensures high atom economy and minimal byproduct formation. The subsequent condensation with salicylaldehydes and mild reduction using sodium borohydride further enhances the environmental profile of the process. By avoiding toxic reagents and operating under relatively mild temperatures, this novel approach significantly reduces the environmental footprint and operational hazards associated with triazole synthesis. For organizations focused on reducing lead time for high-purity pharmaceutical intermediates, this streamlined process offers a robust pathway to rapid clinical supply.
Mechanistic Insights into Diazotization-Azidation Cyclization
The core of this synthetic strategy lies in the efficient construction of the 1,2,3-triazole ring via a [3+2] cycloaddition-like mechanism involving an organic azide and an activated nitrile. The process initiates with the diazotization of the aniline derivative at 0-5°C, where precise temperature control is critical to prevent the decomposition of the unstable diazonium intermediate. Following this, the addition of sodium azide facilitates the substitution of the diazo group with an azido group, generating the key 1,3-dipole required for ring closure. In the presence of a base catalyst such as sodium methoxide, the azide reacts with the methylene group of the phenylacetonitrile, leading to the formation of the triazole ring with high regioselectivity. This mechanistic pathway is advantageous because it avoids the formation of 1,5-disubstituted isomers, which are common impurities in copper-catalyzed click chemistry reactions, thus simplifying downstream purification.
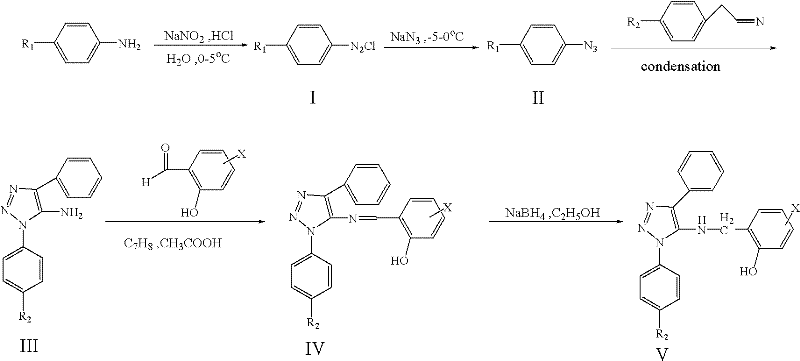
Following the formation of the amino-triazole core, the synthesis proceeds through a condensation reaction with substituted salicylaldehyde to form a Schiff base intermediate. This step is carefully monitored using TLC to ensure complete consumption of the starting amine, preventing the carryover of unreacted materials into the final reduction step. The final transformation involves the reduction of the imine bond using sodium borohydride in ethanol, a reagent chosen for its selectivity and safety profile compared to more aggressive reducing agents like lithium aluminum hydride. The reaction conditions are maintained between 5°C and 40°C to control the exotherm and ensure the integrity of the hydroxyl group on the benzyl moiety. This careful control of reaction parameters ensures that the final product retains the critical o-hydroxybenzyl structure responsible for the enhanced antibacterial activity, while minimizing the formation of over-reduced or degraded byproducts.
How to Synthesize 1-Substituted Phenyl-4-Substituted Phenyl-5-(2-Hydroxyl Substituted Benzyl) Amino-1,2,3-Triazole Efficiently
To achieve optimal results in the production of these high-value intermediates, strict adherence to the patented protocol is essential, particularly regarding stoichiometry and temperature management during the diazotization and azidation phases. The process requires the use of high-purity starting materials to prevent the introduction of trace impurities that could complicate the crystallization of the final product. Operators must utilize real-time analytical monitoring, such as TLC or HPLC, to determine the exact endpoint of each reaction stage, especially the cyclization step which can take up to 60 hours depending on the substituents. Detailed standard operating procedures should be established for the handling of sodium azide, given its toxicity, ensuring that all safety protocols are followed to protect personnel and equipment. The following guide outlines the critical operational parameters derived from the patent examples to assist technical teams in replicating this high-yield synthesis.
- Perform diazotization of substituted aniline at 0-5°C using sodium nitrite and acid to form the diazonium salt.
- Convert the diazonium salt to substituted azidobenzene via reaction with sodium azide at -10°C to 0°C.
- Execute ring closure by reacting azidobenzene with substituted phenylacetonitrile using a base catalyst to form the amino-triazole core.
- Condense the amino-triazole with substituted salicylaldehyde to form the Schiff base intermediate.
- Reduce the Schiff base using sodium borohydride to yield the final 1-substituted phenyl-4-substituted phenyl-5-(2-hydroxyl substituted benzyl) amino-1,2,3-triazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers substantial advantages in terms of cost efficiency and supply chain resilience, addressing key pain points for procurement managers in the fine chemical sector. The elimination of expensive noble metal catalysts removes a significant cost driver and simplifies the supply chain by reducing dependency on scarce resources. Furthermore, the use of commodity chemicals such as aniline derivatives, phenylacetonitriles, and salicylaldehydes ensures that raw material sourcing is stable and less susceptible to market volatility. The process design inherently supports scalability, as the reaction conditions do not require specialized high-pressure or cryogenic equipment beyond standard ice-bath cooling, which is easily implementable in existing multipurpose reactors. This flexibility allows manufacturers to rapidly adjust production volumes in response to market demand without significant capital expenditure.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing sodium borohydride for the final reduction step instead of more expensive and hazardous alternatives, while the solvent system relies on recyclable alcohols and hydrocarbons like ethanol and toluene. The high yields reported in the patent examples, often exceeding 90% in the final reduction step, minimize material waste and maximize throughput per batch. Additionally, the purification strategy relies primarily on recrystallization rather than column chromatography, which is prohibitively expensive at an industrial scale, thereby drastically lowering the cost of goods sold. These factors combine to create a highly economical manufacturing process that enhances profit margins for commercial partners.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals for the starting materials, the risk of supply disruption is significantly mitigated compared to routes requiring custom-synthesized building blocks. The robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supply, such as cooling water temperature fluctuations, ensuring consistent batch-to-bquality. The simplified workup procedures, involving filtration and washing, reduce the processing time per batch, allowing for faster turnaround and improved inventory turnover rates. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: The synthesis aligns with green chemistry principles by avoiding the generation of heavy metal waste streams, which simplifies wastewater treatment and reduces environmental compliance costs. The solvent choices are compatible with standard recovery systems, allowing for high rates of solvent recycling and further reducing the environmental footprint. The absence of high-pressure hydrogenation steps enhances plant safety and lowers insurance and maintenance costs associated with high-risk operations. Consequently, this route facilitates the commercial scale-up of complex pharmaceutical intermediates with minimal regulatory hurdles related to environmental discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their production portfolios. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of adopting this route for large-scale manufacturing and for anticipating any specific technical requirements that may arise during technology transfer.
Q: What is the primary advantage of this triazole synthesis route regarding impurity control?
A: The process utilizes rigorous TLC tracking at every stage, particularly during the cyclization and reduction steps, ensuring that raw material spots disappear completely before proceeding. This minimizes carry-over impurities and simplifies the final recrystallization purification.
Q: How does this method address the issue of drug resistance in existing antifungal treatments?
A: By organically combining the o-hydroxyphenyl fragment with the 1,2,3-triazole molecular fragment, the resulting compounds exhibit a novel structure that demonstrates strong antibacterial activity against Candida albicans and Escherichia coli, overcoming resistance seen in traditional fluconazole treatments.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route employs common solvents like ethanol and toluene, avoids expensive transition metal catalysts, and operates under mild temperature conditions (mostly 0°C to reflux), making it highly scalable and compliant with green chemistry standards for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Substituted Phenyl-4-Substituted Phenyl-5-(2-Hydroxyl Substituted Benzyl) Amino-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates, guaranteeing that every batch complies with international quality standards. We understand the critical nature of supply continuity in the pharmaceutical sector and have established robust contingency plans to mitigate risks associated with raw material availability and logistical challenges. By partnering with us, you gain access to a team of experts dedicated to optimizing process parameters for maximum yield and minimal environmental impact, aligning with your corporate sustainability goals.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Our engineers are ready to conduct a Customized Cost-Saving Analysis to demonstrate how implementing this synthesis route can enhance your bottom line while securing a stable supply of high-quality intermediates. Let us collaborate to accelerate the development of next-generation antifungal therapies and bring these vital medicines to patients faster.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
