Advanced Manufacturing of 1,5-Benzothiazepines: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously seeks novel heterocyclic scaffolds that offer enhanced biological activity while maintaining manufacturability, and the technology disclosed in patent CN101337948B represents a significant advancement in this domain. This patent details the synthesis and application of specific 1,5-benzothiazepine compounds characterized by the presence of carbalkoxy, carboxyl, and carboxylate salt functional groups, which distinguish them from conventional analogs. These structural modifications are not merely academic exercises but are critical engineering decisions that directly influence the solubility, bioavailability, and ultimate therapeutic efficacy of the final drug product. For R&D directors and procurement specialists evaluating new supply chains, understanding the underlying chemistry of these seven-membered sulfur-nitrogen heterocycles is essential for assessing their potential in antifungal drug development. The ability to produce these complex molecules with high purity and consistent quality is a cornerstone of modern pharmaceutical manufacturing, and the methods described herein provide a robust pathway for achieving these goals without relying on scarce or prohibitively expensive reagents.
Furthermore, the strategic importance of this technology lies in its direct application to combating resistant fungal strains, a growing concern in global healthcare. The specific substitution patterns allowed within the general formulas, including various alkyl and aryl groups at the R1 and R2 positions, offer a vast chemical space for medicinal chemists to optimize potency and reduce toxicity. This flexibility is crucial for creating a diverse library of candidates during the lead optimization phase of drug discovery. By leveraging this patented methodology, manufacturers can ensure a steady supply of high-value intermediates that meet the stringent regulatory requirements of major markets. The following analysis delves deep into the technical nuances of this synthesis, comparing it against legacy methods to highlight the operational efficiencies and commercial advantages it offers to forward-thinking chemical enterprises.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for benzothiazepine derivatives often suffer from significant drawbacks that impede their transition from laboratory curiosity to commercial reality. Many conventional methods rely on harsh reaction conditions, such as extremely high temperatures or the use of strong, corrosive acids that pose safety risks and complicate waste management protocols in large-scale facilities. Additionally, older methodologies frequently struggle with regioselectivity, leading to complex mixtures of isomers that require extensive and yield-reducing purification steps like column chromatography, which are economically unviable for ton-scale production. The lack of functional group tolerance in these legacy processes often limits the structural diversity of the final products, restricting the ability of pharmaceutical companies to explore structure-activity relationships effectively. Moreover, the absence of polar functional groups like carboxylates in many traditional analogs results in poor aqueous solubility, creating substantial formulation challenges downstream that increase development time and cost. These cumulative inefficiencies create bottlenecks in the supply chain, making it difficult to secure reliable sources of high-quality intermediates needed for clinical trials and eventual market launch.
The Novel Approach
In stark contrast, the methodology outlined in CN101337948B introduces a streamlined and versatile approach that directly addresses the shortcomings of previous technologies. By focusing on the condensation of substituted o-aminothiophenols with specific alpha,beta-unsaturated ketones, this novel route achieves the formation of the seven-membered ring under remarkably mild conditions, typically ranging from 0°C to 50°C. This temperature window is highly favorable for industrial processing as it reduces energy consumption and minimizes the risk of thermal degradation of sensitive intermediates. The introduction of ester and carboxyl groups at the 2-position of the benzothiazepine ring is a game-changer, as it provides a handle for further derivatization and significantly improves the physicochemical properties of the molecule. 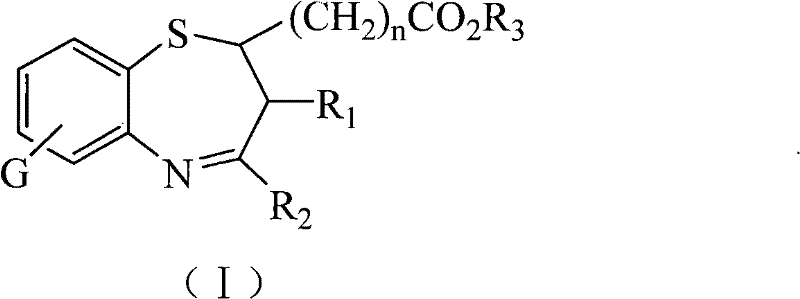
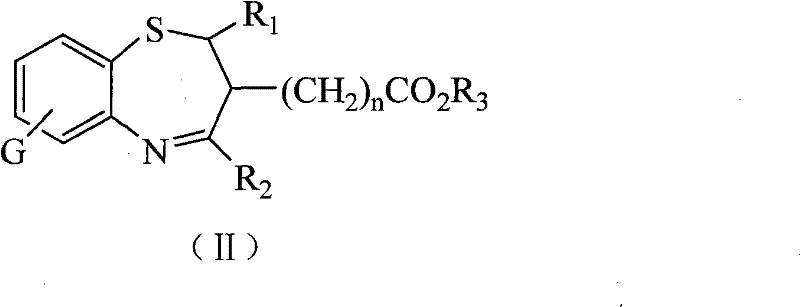 As illustrated in the structural formulas, the variability in the R groups allows for precise tuning of the molecule's lipophilicity and electronic properties. This level of control ensures that the resulting compounds not only exhibit potent antifungal activity against pathogens like Candida albicans but also possess the necessary stability for long-term storage and distribution. The process eliminates the need for exotic catalysts, relying instead on readily available bases and acids, which simplifies the procurement landscape and enhances supply chain resilience.
As illustrated in the structural formulas, the variability in the R groups allows for precise tuning of the molecule's lipophilicity and electronic properties. This level of control ensures that the resulting compounds not only exhibit potent antifungal activity against pathogens like Candida albicans but also possess the necessary stability for long-term storage and distribution. The process eliminates the need for exotic catalysts, relying instead on readily available bases and acids, which simplifies the procurement landscape and enhances supply chain resilience.
Mechanistic Insights into Condensation and Cyclization
The core of this synthetic innovation lies in the mechanistic pathway that facilitates the construction of the 1,5-benzothiazepine scaffold with high fidelity. The reaction initiates with a nucleophilic attack by the sulfur atom of the o-aminothiophenol on the beta-carbon of the alpha,beta-unsaturated ketone, a Michael-type addition that sets the stage for ring closure. This step is critical because the nucleophilicity of the thiol group must be carefully balanced against the electrophilicity of the enone system to prevent polymerization or side reactions. Following the initial addition, an intramolecular cyclization occurs involving the amino group, closing the seven-membered ring and establishing the characteristic benzothiazepine core. The presence of the ester side chain plays a subtle yet vital role in stabilizing the transition states through electronic effects, ensuring that the cyclization proceeds efficiently even with sterically hindered substrates. Understanding this mechanism is paramount for process chemists who need to troubleshoot potential issues during scale-up, such as incomplete conversion or the formation of byproducts. The ability to drive this reaction to completion without requiring expensive transition metal catalysts is a testament to the elegance of the design, offering a greener and more sustainable alternative to metal-mediated coupling reactions often seen in fine chemical synthesis.
Beyond the ring formation, the subsequent functionalization steps are equally critical for defining the commercial viability of the process. The hydrolysis of the ester group to the corresponding carboxylic acid or its salt form is achieved through standard saponification conditions using sodium or potassium hydroxide. This transformation is highly selective, leaving the sensitive imine bond within the heterocyclic ring intact, which demonstrates the robustness of the core structure under basic conditions. The final acidification step allows for the isolation of the free acid, which can then be purified through recrystallization, a unit operation that is highly scalable and cost-effective compared to chromatographic techniques. This sequence of reactions ensures that the final product meets the rigorous purity specifications required for pharmaceutical applications, minimizing the burden on quality control laboratories. By controlling the pH and temperature during these workup stages, manufacturers can effectively manage the impurity profile, ensuring that the levels of residual starting materials and side products remain well below regulatory thresholds. This meticulous attention to detail in the reaction mechanism translates directly into a more reliable and predictable manufacturing process.
How to Synthesize 1,5-Benzothiazepines Efficiently
Implementing this synthesis in a production environment requires a clear understanding of the operational parameters that govern yield and quality. The process begins with the careful selection of solvents, with anhydrous methanol, DMSO, or dichloromethane being preferred due to their ability to dissolve both the polar amino-thiophenols and the organic ketones effectively. Maintaining the reaction temperature within the specified 0-50°C range is crucial; deviations can lead to slower kinetics or increased impurity formation, impacting the overall economics of the batch. Once the cyclization is complete, the isolation of the intermediate ester is typically achieved through filtration or solvent removal, followed by recrystallization to ensure high purity before proceeding to the hydrolysis step. 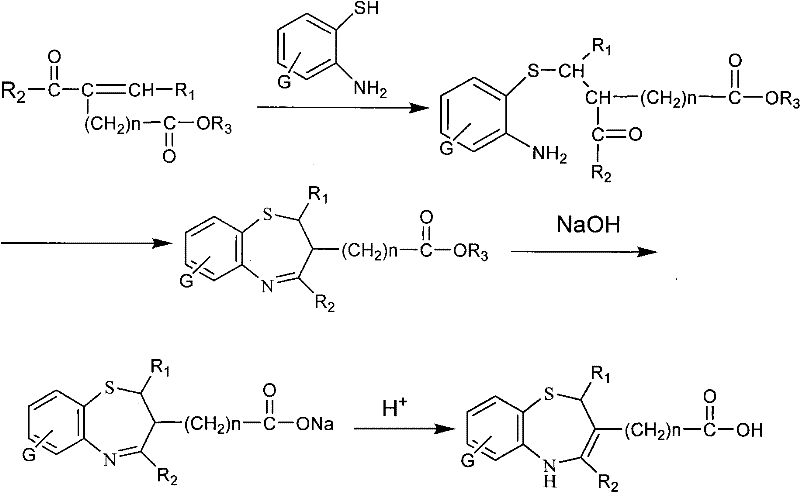 The visual representation of the reaction pathway highlights the simplicity of the transformation, moving from readily available starting materials to the complex heterocyclic target in just a few logical steps. This simplicity is a key driver for cost reduction, as it reduces the number of unit operations and the associated labor and equipment time. For technical teams looking to adopt this route, it is essential to validate the stoichiometry and mixing rates to ensure homogeneity, particularly when scaling from grams to kilograms. The final acidification step requires precise pH monitoring to ensure complete precipitation of the product without co-precipitating inorganic salts, which could compromise the purity of the final API intermediate.
The visual representation of the reaction pathway highlights the simplicity of the transformation, moving from readily available starting materials to the complex heterocyclic target in just a few logical steps. This simplicity is a key driver for cost reduction, as it reduces the number of unit operations and the associated labor and equipment time. For technical teams looking to adopt this route, it is essential to validate the stoichiometry and mixing rates to ensure homogeneity, particularly when scaling from grams to kilograms. The final acidification step requires precise pH monitoring to ensure complete precipitation of the product without co-precipitating inorganic salts, which could compromise the purity of the final API intermediate.
- Condensation and Cyclization: React substituted o-aminothiophenols with alpha,beta-unsaturated ketones in solvents like methanol or DMSO at 0-50°C to form the benzothiazepine ring.
- Hydrolysis: Treat the resulting ester intermediate with aqueous sodium hydroxide or potassium hydroxide to convert the ester group into a carboxylate salt.
- Acidification: Adjust the pH to less than 3 using acetic acid or hydrochloric acid to isolate the final free carboxylic acid form of the 1,5-benzothiazepine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthesis route offers compelling advantages that extend beyond mere technical feasibility. The reliance on commodity chemicals such as o-aminothiophenols and simple unsaturated ketones means that the raw material supply chain is robust and less susceptible to the volatility often seen with specialized reagents. This stability is crucial for supply chain heads who need to guarantee continuity of supply for long-term production campaigns. Furthermore, the avoidance of precious metal catalysts eliminates a significant cost center and removes the need for complex metal scavenging steps, which are often required to meet strict residual metal limits in pharmaceutical products. The mild reaction conditions also contribute to lower energy costs and reduced wear and tear on reactor vessels, extending the lifespan of capital equipment. These factors combine to create a manufacturing process that is not only chemically efficient but also economically resilient, providing a buffer against market fluctuations and raw material price spikes. For procurement managers, this translates into a more predictable cost structure and the ability to negotiate better terms with suppliers due to the standardized nature of the inputs.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of standard hydrolysis reagents significantly lowers the direct material costs associated with production. By avoiding complex purification methods like preparative HPLC and relying instead on crystallization, the process reduces solvent consumption and waste disposal costs, leading to substantial overall savings. The high yields reported in the patent examples suggest that material throughput is optimized, minimizing the loss of valuable intermediates during synthesis. This efficiency allows for a more competitive pricing model for the final intermediate, making it an attractive option for cost-sensitive generic drug development projects. Additionally, the simplified workflow reduces labor hours per batch, further enhancing the economic viability of large-scale manufacturing operations.
- Enhanced Supply Chain Reliability: The use of widely available solvents like methanol and DMSO ensures that production is not held hostage by the scarcity of niche chemicals. This accessibility means that multiple suppliers can be qualified for raw materials, reducing the risk of single-source dependency and enhancing supply chain security. The robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling companies to leverage global production networks to optimize logistics and reduce lead times. The stability of the intermediates also facilitates easier storage and transportation, reducing the need for specialized cold-chain logistics. For supply chain leaders, this reliability is invaluable, as it ensures that production schedules can be met consistently without unexpected delays caused by material shortages or process failures.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to pilot plant and full commercial production without significant re-engineering. The mild temperatures and pressures involved reduce the safety risks associated with exothermic runaways, making it easier to obtain regulatory approvals for new manufacturing sites. Furthermore, the reduced use of hazardous reagents and the generation of less toxic waste align with modern green chemistry principles, helping companies meet increasingly stringent environmental regulations. This compliance not only avoids potential fines but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B negotiations. The ability to produce high volumes with a smaller environmental footprint is a strategic advantage that resonates with stakeholders across the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 1,5-benzothiazepine derivatives. These answers are derived directly from the technical specifications and experimental data provided in the source patent, ensuring accuracy and relevance for industry professionals. Understanding these details is critical for making informed decisions about integrating this technology into your existing product pipeline. The clarity provided here aims to bridge the gap between theoretical chemistry and practical manufacturing realities, offering actionable insights for your team.
Q: What are the primary antifungal targets for these 1,5-benzothiazepine derivatives?
A: According to patent CN101337948B, these compounds exhibit significant inhibitory effects against Candida albicans and Cryptococcus neoformans, making them valuable for developing new antifungal therapies.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like methanol and DMSO and operates at mild temperatures (0-50°C), avoiding extreme conditions that hinder scalability, thus ensuring reliable commercial supply.
Q: How does the carboxyl functional group impact the compound's utility?
A: The introduction of carboxyl and carboxylate salt groups enhances water solubility and bioavailability compared to traditional benzothiazepines, addressing key formulation challenges in drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Benzothiazepines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new antifungal therapies. Our team of expert chemists has extensively evaluated the synthesis route described in CN101337948B and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of 1,5-benzothiazepines we deliver meets the highest international standards. We understand that in the pharmaceutical industry, there is no room for error, and our commitment to quality assurance is unwavering. By partnering with us, you gain access to a supply chain that is not only reliable but also deeply knowledgeable about the specific chemical challenges associated with heterocyclic synthesis.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out to us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities align with your project goals. Whether you are in the early stages of lead optimization or preparing for commercial launch, our expertise can help you navigate the complexities of chemical sourcing. Let us be your partner in turning innovative chemistry into life-saving medicines, ensuring that you have the reliable support you need to succeed in a competitive global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
