Advanced Synthesis of Benzothiazole-Tröger's Base Derivatives for High-Performance Fluorescent Materials
The chemical landscape for advanced functional materials is constantly evolving, driven by the need for molecules that combine structural rigidity with superior optical performance. Patent CN111440178A introduces a groundbreaking class of Benzothiazole-Schiff base-Tröger's base derivatives that address critical limitations in current fluorescent probe technology. These novel compounds are engineered by covalently linking arylvinyl-containing benzothiazole fragments to a rigid Tröger's base (TB) scaffold through imine (C=N) bonds. This strategic molecular design significantly extends the conjugated system while maintaining a unique V-shaped geometry that prevents detrimental pi-pi stacking in the solid state. The resulting materials exhibit remarkable Aggregation-Induced Emission (AIE) characteristics, large Stokes shifts, and robust thermal stability, positioning them as ideal candidates for next-generation optoelectronic devices and biological imaging applications. Furthermore, preliminary biological screening indicates that specific analogs within this series possess potent antifungal activity, expanding their utility beyond mere diagnostics into therapeutic domains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional fluorescent probes often suffer from the Aggregation-Caused Quenching (ACQ) effect, where fluorescence intensity drastically diminishes upon aggregation or in the solid state due to strong intermolecular pi-pi interactions. This phenomenon severely limits their application in high-concentration environments or solid-state devices like OLEDs. Moreover, many conventional synthesis routes for heterocyclic Schiff bases rely on harsh reaction conditions, toxic solvents, or expensive transition metal catalysts that complicate downstream purification and increase environmental burdens. The lack of structural rigidity in many linear fluorophores also leads to non-radiative decay pathways via intramolecular rotation, reducing quantum yields. Consequently, there is a pressing industrial demand for molecular architectures that can maintain high luminosity in aggregated states while being synthesized through cost-effective and environmentally benign processes.
The Novel Approach
The methodology outlined in CN111440178A overcomes these hurdles by utilizing the intrinsic V-shape of the Tröger's base core to sterically hinder close packing of the fluorophores. By integrating benzothiazole units, known for their high fluorescence quantum yields and electron-transporting capabilities, the inventors have created a hybrid system that leverages the Restriction of Intramolecular Motion (RIM) mechanism. The synthesis avoids noble metal catalysts, relying instead on classical organic transformations such as acid-catalyzed cyclization and base-mediated condensation. The final products are obtained through simple reflux in ethanol followed by filtration, eliminating the need for complex chromatographic separations on a large scale. This approach not only enhances the photophysical properties, yielding yellow-green emission with large Stokes shifts, but also streamlines the manufacturing process, making it highly attractive for commercial scale-up of complex heterocyclic compounds.
Mechanistic Insights into Tröger's Base Cyclization and Schiff Base Formation
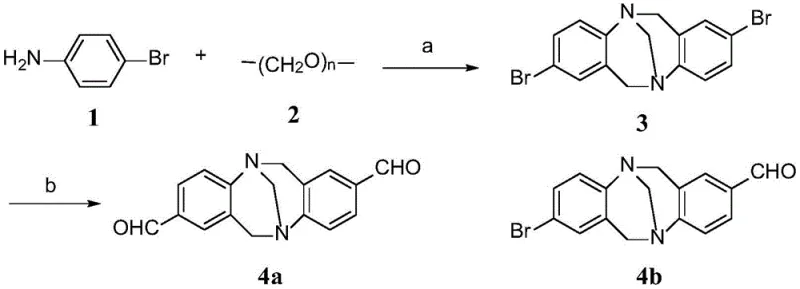
The synthesis begins with the construction of the rigid Tröger's base skeleton, a process that demands precise control over reaction conditions to ensure high regioselectivity and yield. As illustrated in the reaction scheme below, p-bromoaniline reacts with paraformaldehyde in the presence of trifluoroacetic acid at low temperatures, gradually warming to room temperature over several days. This acid-catalyzed condensation forms the methylene-bridged diazocine ring system characteristic of Tröger's base. Subsequent functionalization involves lithiation at cryogenic temperatures (-78°C) using n-butyllithium, followed by quenching with DMF to introduce formyl groups at the desired positions. This step is critical for creating the reactive handles necessary for the final assembly. The ability to selectively mono- or di-formylate the core allows for the creation of diverse libraries of derivatives, providing flexibility in tuning the electronic properties of the final material.
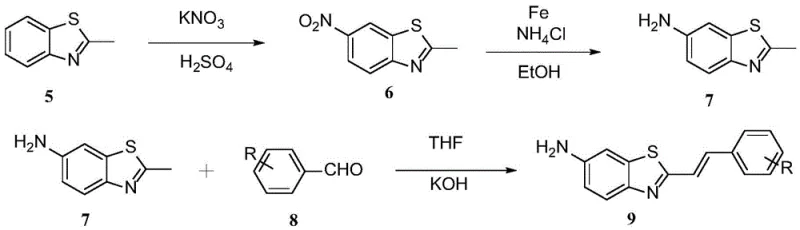
Parallel to the core synthesis, the benzothiazole wings are constructed through a sequence involving nitration, reduction, and Knoevenagel condensation. 2-Methylbenzothiazole is first nitrated to introduce an electron-withdrawing group, which is subsequently reduced to an amine using iron powder. This amine then undergoes a condensation reaction with various substituted benzaldehydes in the presence of potassium hydroxide to form the styryl-benzothiazole moiety. The final assembly involves the condensation of the aldehyde-functionalized Tröger's base with the amino-functionalized benzothiazole arms. This Schiff base formation is driven by the removal of water under reflux conditions in ethanol. The resulting imine linkages extend the conjugation length across the entire molecule, facilitating efficient intramolecular charge transfer (ICT) which is responsible for the observed solvatochromism and enhanced fluorescence. The rigidification of the molecular structure upon aggregation restricts the rotation of these bonds, thereby activating the AIE mechanism and leading to bright solid-state emission.
How to Synthesize Benzothiazole-Schiff base-Tröger's base Derivatives Efficiently
The preparation of these high-value intermediates follows a logical, step-wise progression that balances chemical complexity with operational simplicity. The process starts with the formation of the central scaffold, followed by the independent synthesis of the fluorescent arms, and concludes with a convergent coupling step. This modular approach allows for the easy variation of substituents on the benzothiazole ring to fine-tune emission wavelengths and biological activity without altering the core synthetic strategy. The final condensation step is particularly robust, proceeding cleanly in a green solvent like ethanol, which aligns with modern principles of sustainable chemistry. For detailed operational parameters, stoichiometry, and workup procedures required to achieve the reported yields and purity levels, please refer to the standardized synthesis guide below.
- Synthesize the Tröger's base core (Intermediate 3) via acid-catalyzed cyclization of p-bromoaniline and paraformaldehyde, followed by lithiation and formylation to obtain dialdehyde intermediates 4a or 4b.
- Prepare the benzothiazole vinyl arms (Intermediate 9/9') through nitration of 2-methylbenzothiazole, reduction to the amine, and Knoevenagel condensation with substituted benzaldehydes.
- Execute the final condensation by refluxing the Tröger's base aldehyde with the benzothiazole amine in ethanol, followed by filtration and recrystallization to isolate the target Schiff base derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthetic route described in this patent offers significant advantages over traditional methods for producing complex fluorophores. The reliance on commodity chemicals such as p-bromoaniline, paraformaldehyde, and substituted benzaldehydes ensures a stable and cost-effective raw material supply chain. Unlike routes requiring specialized organometallic reagents or rare earth catalysts, this process utilizes widely available acids and bases, mitigating the risk of supply disruptions and price volatility associated with critical raw materials. Furthermore, the isolation of the final products via simple filtration and washing eliminates the need for expensive silica gel column chromatography, which is often a bottleneck in large-scale manufacturing. This simplification of the downstream processing directly translates to reduced production costs and shorter cycle times, enhancing the overall economic viability of the project.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging steps and rigorous heavy metal testing, which are mandatory for pharmaceutical grade intermediates. Additionally, the use of ethanol as the primary solvent for the final step reduces solvent recovery costs and environmental compliance burdens compared to chlorinated or aromatic solvents. The high yields reported for the final condensation step indicate efficient atom economy, minimizing waste generation and maximizing the output per batch. These factors collectively contribute to a substantially lower cost of goods sold (COGS), making these materials competitive for high-volume applications in the fine chemical industry.
- Enhanced Supply Chain Reliability: The synthetic pathway is composed of robust reactions that are less sensitive to minor fluctuations in temperature or moisture, ensuring consistent batch-to-bquality. The starting materials are bulk chemicals with multiple global suppliers, reducing dependency on single-source vendors. The straightforward workup procedures, involving standard extraction and recrystallization techniques, can be easily implemented in existing multipurpose chemical plants without the need for specialized equipment. This adaptability ensures that production can be scaled up rapidly to meet market demand, providing a reliable source of high-purity fluorescent probe materials for downstream users.
- Scalability and Environmental Compliance: The process avoids the generation of hazardous waste streams associated with heavy metal catalysis or harsh oxidizing agents. The primary byproducts are water and inorganic salts, which are easier to treat and dispose of in accordance with environmental regulations. The solid nature of the intermediates and final products facilitates safe handling, storage, and transportation, reducing the risks associated with liquid hazardous materials. The inherent thermal stability of the Tröger's base core further ensures safety during scale-up, allowing for operation at elevated temperatures if necessary to drive reaction kinetics without compromising product integrity or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these aspects is crucial for evaluating the feasibility of integrating these materials into your specific product development pipelines.
Q: What are the key optical properties of these Benzothiazole-Tröger's base derivatives?
A: These derivatives exhibit excellent Aggregation-Induced Emission (AIE) properties, large Stokes shifts, and strong yellow-green solid-state luminescence, making them superior to traditional ACQ fluorophores for solid-state applications.
Q: Does this synthesis route require expensive transition metal catalysts?
A: No, the synthesis primarily relies on acid catalysis (trifluoroacetic acid, sulfuric acid) and base-mediated condensations (KOH), avoiding the need for costly palladium or other transition metal catalysts, which simplifies purification and reduces heavy metal residues.
Q: What is the antifungal potential of these compounds?
A: Specific derivatives, such as compound 11j, have demonstrated significant inhibitory activity against Aspergillus fumigatus (AF1160) with low MIC values, indicating strong potential for development into novel antifungal pharmaceutical agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiazole-Schiff base-Tröger's base Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of these AIE-active materials in the fields of bioimaging and optoelectronics. Our team of expert chemists has thoroughly analyzed the synthetic route disclosed in CN111440178A and is fully prepared to support your development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early research phase or full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling and structural confirmation via NMR and MS, guaranteeing that every batch meets the highest international standards.
We invite you to collaborate with us to optimize this chemistry for your specific applications. Whether you require custom analogs with modified emission profiles or need to secure a stable supply of the standard derivatives for antifungal drug development, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project. We encourage you to reach out for specific COA data and route feasibility assessments to see how we can accelerate your time-to-market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
