Advanced Anhydrous Synthesis of Ethambutol Hydrochloride for Commercial Scale-up
Introduction to Advanced Ethambutol Hydrochloride Manufacturing
The global pharmaceutical landscape continues to demand robust, scalable, and cost-effective solutions for critical anti-tuberculosis agents, with Ethambutol Hydrochloride remaining a cornerstone therapy. As detailed in patent CN108218724B, a significant technological breakthrough has been achieved in the synthesis of this vital compound, addressing long-standing inefficiencies in traditional manufacturing protocols. This novel methodology shifts the paradigm from corrosive, multi-step neutralization processes to a streamlined, anhydrous condensation and direct acidification route. For R&D Directors and Supply Chain Heads, this represents a pivotal opportunity to optimize production lines for higher purity and reduced operational expenditure. The process leverages precise control over reaction parameters, specifically the molar ratios of chiral starting materials and strict moisture control, to achieve yields exceeding 80% with ignition residues as low as 0.01%. By integrating these advanced synthetic strategies, manufacturers can secure a more reliable supply of high-purity pharmaceutical intermediates while mitigating the environmental and equipment risks associated with legacy chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ethambutol Hydrochloride has been plagued by significant operational bottlenecks that inflate costs and complicate scale-up. Traditional routes typically involve a condensation reaction followed by a mandatory neutralization step using strong bases such as sodium hydroxide to handle the hydrochloric acid generated during the process. This approach introduces severe drawbacks, including substantial corrosion to reaction vessels due to the presence of strong alkalis and subsequent saline byproducts. Furthermore, the generation of inorganic salts creates a complex waste stream that requires extensive treatment, increasing the environmental footprint and disposal costs. The necessity for neutralization also complicates the recovery of unreacted starting materials, particularly the expensive chiral S-(+)-2-amino-1-butanol, often leading to lower overall atom economy. Additionally, older methods utilizing gaseous precursors like 1,2-epoxy-3-butene pose safety hazards and equipment complexity issues regarding feeding control, while routes starting from ethyl (S)-2-aminobutyrate suffer from prohibitive raw material costs and availability constraints.
The Novel Approach
The innovative method disclosed in the patent data fundamentally reengineers the synthesis pathway by eliminating the problematic neutralization step entirely. Instead of neutralizing with base, the process utilizes a direct acidification strategy where an alcoholic solution of hydrogen chloride is introduced to the condensation mixture. This shift not only removes the source of equipment corrosion and solid salt waste but also simplifies the downstream processing significantly. By maintaining anhydrous conditions throughout the reaction, the method facilitates the efficient recovery and recycling of the valuable S-(+)-2-amino-1-butanol from the mother liquor without the need for complex dehydration steps like azeotropic distillation with toluene. The result is a cleaner reaction profile with fewer impurities, a simplified workflow that shortens the production cycle, and a dramatic reduction in the consumption of auxiliary chemicals. This approach aligns perfectly with modern green chemistry principles, offering a sustainable alternative that enhances both economic viability and operational safety for large-scale manufacturing facilities.
Mechanistic Insights into Anhydrous Condensation and Direct Salification
At the heart of this synthesis lies a precise nucleophilic substitution mechanism where the amino group of S-(+)-2-amino-1-butanol attacks the electrophilic carbon of 1,2-dichloroethane. The reaction is conducted at elevated temperatures, typically between 120°C and 130°C, to overcome the activation energy barrier while maintaining the stability of the chiral center. Critical to the success of this mechanism is the strict control of the molar ratio between the amine and the dichloroalkane, optimized at approximately 9.36:1 to 10:1. This excess of the chiral amine serves a dual purpose: it drives the equilibrium towards the formation of the desired diamine product and acts as a scavenger for the HCl generated, although the primary acidification is handled later. The integrity of the stereocenter is paramount, necessitating the use of starting materials with a specific rotation between +9.3° and +10.9°. Any deviation in optical purity at this stage would propagate through the synthesis, compromising the therapeutic efficacy of the final API.
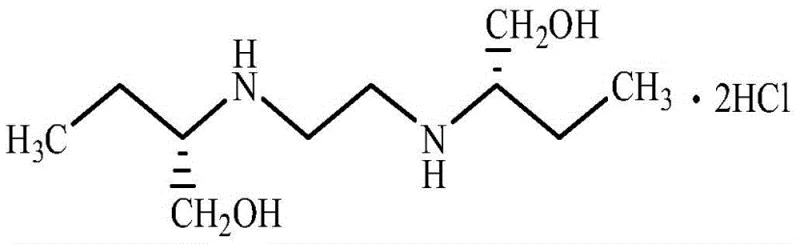
Following the condensation, the transition to the hydrochloride salt is managed through the addition of alcoholic HCl, preferably in ethanol, under controlled low-temperature conditions (10°C to 50°C). This step is meticulously designed to occur in an environment with water content below 3wt%, ideally less than 1.5wt%. The absence of water is mechanistically crucial because it prevents the hydrolysis of the intermediate and ensures that the unreacted S-(+)-2-amino-1-butanol remains in a state suitable for direct recovery and reuse in subsequent batches. The crystallization process, occurring at 5°C to 15°C, allows for the formation of well-defined crystals with a particle size distribution of 90-110 mesh, which is optimal for downstream formulation. This mechanistic control over hydration and acidity ensures that the final product achieves a purity of over 99% with negligible ignition residue, meeting the stringent specifications required for pharmaceutical grade intermediates.
How to Synthesize Ethambutol Hydrochloride Efficiently
The implementation of this synthesis route requires careful attention to thermal management and reagent quality to maximize yield and purity. The process begins with the charging of high-optical-purity S-(+)-2-amino-1-butanol into a reactor, followed by the slow addition of 1,2-dichloroethane under heating. Maintaining the temperature window of 120-130°C for a duration of 3 hours ensures complete conversion while minimizing side reactions. Upon completion, the mixture is cooled, and absolute ethanol is added to facilitate the subsequent salification. The addition of alcoholic HCl must be controlled to maintain a pH between 3 and 3.5, ensuring complete salt formation without excessive acidity that could degrade the product. Finally, the product is isolated via filtration and washing, while the mother liquor is processed to recover the valuable chiral amine. For the detailed standardized operating procedures and specific equipment settings required for this synthesis, please refer to the guide below.
- Condense S-(+)-2-amino-1-butanol with 1,2-dichloroethane at 120-130°C under anhydrous conditions to form the free base.
- Add an alcoholic HCl solution directly to the reaction mixture to form the hydrochloride salt, avoiding sodium hydroxide neutralization.
- Crystallize the product at low temperature (5-15°C) and recover unreacted aminobutanol from the mother liquor for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method translates into tangible strategic advantages that extend beyond simple yield improvements. The elimination of the sodium hydroxide neutralization step removes a significant variable cost associated with base consumption and the handling of corrosive hazardous materials. Furthermore, by avoiding the generation of inorganic salt waste, the facility reduces its burden on wastewater treatment systems and lowers compliance costs related to environmental discharge. The ability to recover and reuse the expensive chiral starting material, S-(+)-2-amino-1-butanol, directly from the mother liquor without complex purification significantly lowers the raw material cost per kilogram of finished product. This closed-loop material flow enhances supply chain resilience by reducing dependency on fresh raw material shipments and mitigating the impact of market price volatility for chiral building blocks.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for purchasing and storing large quantities of sodium hydroxide, thereby reducing raw material inventory costs. Additionally, the removal of the neutralization step decreases the consumption of hydrochloric acid, as the acid is used more efficiently for direct salification rather than being partially consumed in neutralizing base. The reduction in equipment corrosion extends the lifespan of reactors and piping, leading to lower capital expenditure on maintenance and replacement over time. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: By simplifying the process flow and removing complex separation steps associated with salt removal, the production cycle time is significantly shortened. This increased throughput capability allows manufacturers to respond more rapidly to fluctuations in market demand for anti-tuberculosis medications. The robustness of the anhydrous process also reduces the risk of batch failures due to moisture sensitivity or pH excursions, ensuring a more consistent and reliable supply of high-quality product to downstream API manufacturers. This reliability is critical for maintaining uninterrupted production schedules in the highly regulated pharmaceutical sector.
- Scalability and Environmental Compliance: The process is inherently safer and more environmentally friendly, as it avoids the use of strong corrosive bases and minimizes the generation of solid and liquid waste streams. This aligns with increasingly stringent global environmental regulations, reducing the risk of regulatory fines or shutdowns. The simplicity of the operation makes it highly scalable from pilot plant to commercial production volumes without the need for specialized corrosion-resistant alloys that would otherwise be required for handling hot caustic solutions. This ease of scale-up ensures that supply can be ramped up quickly to meet global health demands without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing operations. The answers highlight the critical control points that differentiate this method from conventional approaches.
Q: How does the new synthesis method reduce equipment corrosion?
A: The novel process eliminates the traditional neutralization step using strong bases like sodium hydroxide. By proceeding directly to acidification with alcoholic HCl, the method avoids the generation of corrosive saline byproducts and reduces the overall chemical aggressiveness towards reactor vessels.
Q: What is the impact of water content on the reaction yield?
A: Maintaining anhydrous conditions is critical. The patent specifies that water content should be controlled below 3wt%, preferably below 1.5wt%. Excess water hinders the recovery of the expensive chiral starting material, S-(+)-2-amino-1-butanol, and can negatively affect the crystallization quality and yield of the final hydrochloride salt.
Q: Why is the specific rotation of the starting material important?
A: Using S-(+)-2-amino-1-butanol with a high specific rotation (between +9.3° and +10.9°) ensures the stereochemical integrity of the final API intermediate. High optical purity in the feedstock correlates directly with higher product purity and reduced impurity profiles in the final Ethambutol Hydrochloride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethambutol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the pharmaceutical industry. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the anhydrous synthesis of Ethambutol Hydrochloride can be seamlessly transferred to industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch conforms to the highest international standards. Our infrastructure is designed to handle complex chiral syntheses with precision, making us an ideal partner for companies seeking to optimize their supply chain for anti-tuberculosis agents.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing setup. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and reliability in your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
