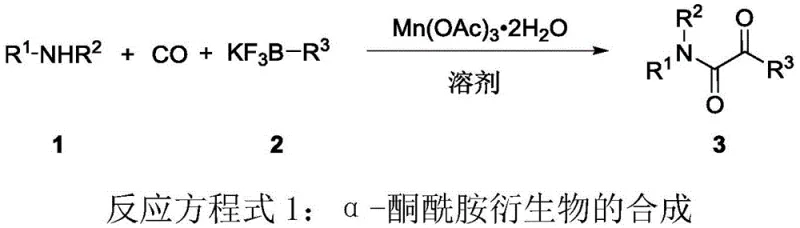
Revolutionizing Alpha-Ketoamide Production: Scalable Manganese-Catalyzed Synthesis for Pharmaceutical Supply Chains
Patent CN115572239A introduces a groundbreaking one-pot synthesis method for α-ketoamide compounds, leveraging trivalent manganese acetate as a catalyst under carbon monoxide atmosphere. This innovative approach addresses critical limitations in conventional palladium-based systems by eliminating expensive metal catalysts and expanding substrate compatibility to include aryl amines. The process achieves high yields and selectivity through a radical-based mechanism, offering significant advantages for pharmaceutical intermediate manufacturing. By utilizing simple and readily available starting materials such as amines and alkyl boron reagents, this method provides a scalable pathway for producing structurally diverse α-ketoamide derivatives essential in drug development. The patent demonstrates robust operational parameters including optimal temperature at 60°C and pressure at 60 atmospheres, ensuring reproducibility across various molecular scaffolds. This advancement represents a paradigm shift in synthetic methodology with profound implications for cost-effective production of bioactive compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional palladium-catalyzed dicarbonylation methods suffer from severe constraints including prohibitive costs associated with palladium-phosphine ligand systems and narrow substrate scope limited exclusively to alkyl amines with electron-donating substituents. These processes fail completely when applied to aryl amines or electron-withdrawing electrophiles, creating significant barriers for synthesizing complex pharmaceutical intermediates like Telaprevir derivatives. Additionally, the requirement for specialized purification steps to remove toxic metal residues increases production complexity while generating hazardous waste streams that complicate environmental compliance. The inherent sensitivity of palladium systems to reaction conditions often results in inconsistent yields and unwanted by-products such as simple amides, necessitating costly separation procedures that undermine commercial viability for large-scale manufacturing operations.
The Novel Approach
The patented manganese-catalyzed process overcomes these limitations through its unique radical-based mechanism that operates effectively with both alkyl and aryl amines across diverse electronic profiles. By employing inexpensive trivalent manganese acetate dihydrate as catalyst under standardized carbon monoxide pressure (60 atm) at moderate temperatures (60°C), it achieves consistent high yields without requiring precious metal handling infrastructure. This method eliminates the need for specialized ligands while maintaining exceptional selectivity that prevents amide by-product formation through controlled radical pathways. The use of simple solvents like 1,2-dichloroethane and readily available alkyl boron reagents creates an inherently scalable process that significantly reduces both capital expenditure and operational complexity compared to conventional approaches.
Mechanistic Insights into Manganese-Promoted Dicarbonylation Amination
The reaction proceeds through a radical-mediated dicarbonylation pathway where trivalent manganese acetate initiates single-electron transfer to generate nitrogen-centered radicals from amine substrates. This critical step enables nucleophilic attack on carbon monoxide followed by transmetalation with alkyl boron reagents, forming key acyl radical intermediates that undergo further carbonylation before reductive elimination. The manganese catalyst cycles between +3 and +2 oxidation states during this process, facilitating continuous radical generation without requiring external oxidants or reductants. This mechanism fundamentally differs from palladium-catalyzed pathways by avoiding oxidative addition steps that limit substrate scope, thereby accommodating electron-deficient aryl amines that would poison traditional transition metal catalysts.
Impurity control is achieved through the selective radical recombination pathway that prevents competing hydrolysis or over-carbonylation reactions common in conventional methods. The absence of acidic or basic conditions eliminates potential racemization issues while the mild reaction temperature (60°C) prevents thermal degradation of sensitive functional groups. Crucially, the mechanism inherently suppresses amide formation by favoring double carbonylation over single insertion events, resulting in consistently high-purity products without requiring additional purification steps. This selectivity profile directly addresses critical quality attributes demanded by pharmaceutical manufacturers for complex intermediate synthesis.
How to Synthesize Alpha-Ketoamide Efficiently
This patented methodology represents a significant advancement in alpha-ketoamide production through its streamlined one-pot process that eliminates multiple purification steps required by conventional approaches. The integration of readily available starting materials with standardized reaction parameters creates an ideal platform for technology transfer to manufacturing environments. Detailed operational procedures have been validated across diverse substrate classes as demonstrated in the patent examples, providing robust protocols for immediate implementation in commercial settings. The following standardized synthesis steps outline the precise methodology required to achieve optimal results in industrial-scale production.
- Prepare the reaction mixture by combining amine, alkyl boron reagent, and trivalent manganese acetate dihydrate in a high-pressure reactor with 1,2-dichloroethane solvent.
- Subject the mixture to carbon monoxide atmosphere at 60 atmospheres pressure and heat to 60°C for 16 hours to facilitate the dicarbonylation amination.
- After reaction completion, isolate the alpha-ketoamide derivative through column chromatography to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative process delivers transformative benefits for procurement and supply chain operations by addressing fundamental pain points in pharmaceutical intermediate manufacturing. The elimination of precious metal catalysts removes significant cost drivers while enhancing supply chain resilience through reliance on globally available raw materials. Standardized reaction conditions enable seamless technology transfer between development and production facilities, reducing qualification timelines and minimizing scale-up risks that typically plague complex syntheses. These advantages collectively create substantial value across the procurement lifecycle while strengthening strategic sourcing capabilities.
- Cost Reduction in Manufacturing: The substitution of expensive palladium-phosphine systems with non-toxic manganese catalysts eliminates both raw material costs and downstream purification expenses associated with metal residue removal. This fundamental shift in catalytic approach creates substantial cost savings through simplified process design that avoids specialized equipment requirements while maintaining high product quality standards essential for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials including common amines and alkyl boron reagents ensures consistent supply availability without dependence on constrained specialty chemicals. The robust reaction profile tolerates minor variations in raw material quality while maintaining performance specifications, significantly reducing supply chain vulnerability compared to conventional methods requiring highly purified precursors.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production due to its straightforward operational parameters and absence of hazardous intermediates. Simplified waste streams resulting from non-toxic catalyst usage facilitate environmental compliance while reducing disposal costs, making this approach particularly suitable for sustainable manufacturing initiatives within regulated industries.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of the patented methodology and its implementation requirements. These insights derive directly from experimental data presented in the patent documentation regarding reaction parameters, substrate scope limitations, and quality control outcomes observed during validation studies.
Q: How does this method overcome limitations of palladium-catalyzed systems for alpha-ketoamide synthesis?
A: The manganese-catalyzed process eliminates expensive palladium-phosphine catalysts while maintaining high selectivity through radical-based mechanisms. It accommodates electron-withdrawing substituents and aryl amines that fail in conventional systems, significantly broadening substrate scope without requiring specialized ligands or purification steps for metal residues.
Q: What advantages does trivalent manganese offer over traditional catalysts in pharmaceutical intermediate production?
A: Manganese provides substantial cost reduction as it is non-toxic and inexpensive compared to palladium systems. Its compatibility with diverse amine sources including aryl amines enables synthesis of previously inaccessible derivatives, while the radical mechanism prevents amide by-product formation ensuring higher purity without additional separation processes.
Q: How does this process ensure supply chain reliability for complex pharmaceutical intermediates?
A: The method utilizes readily available starting materials under standardized conditions (60 atm CO, 60°C), enabling seamless scale-up from laboratory to commercial production. The absence of precious metal catalysts eliminates supply constraints while consistent yields across varied substrates provide predictable output for continuous manufacturing requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketoamide Supplier
Our company leverages this patented technology to deliver high-purity alpha-ketoamide intermediates with exceptional consistency across diverse molecular architectures. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. Our integrated manufacturing platform ensures seamless transition from development to full-scale production without compromising quality or delivery timelines, providing pharmaceutical clients with reliable access to critical building blocks for complex drug substances.
We invite procurement teams to initiate technical discussions by requesting our Customized Cost-Saving Analysis which details specific implementation pathways for your target molecules. Contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your manufacturing requirements.
