Advanced Synthesis of High Thermal Stability Carbazolyl Stilbene Derivatives for Next-Generation OLED Applications
Advanced Synthesis of High Thermal Stability Carbazolyl Stilbene Derivatives for Next-Generation OLED Applications
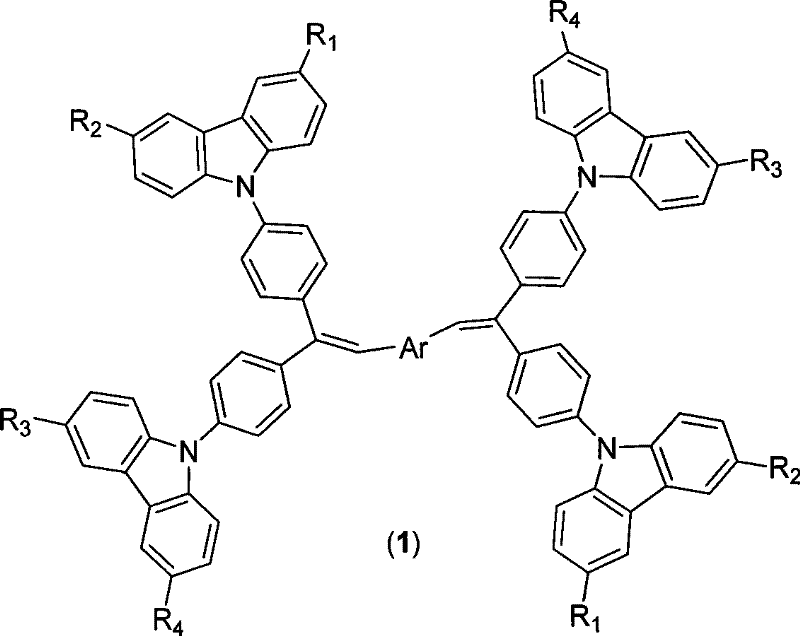
The rapid evolution of Organic Light Emitting Diode (OLED) technology demands materials that not only offer high luminous efficiency but also possess exceptional thermal stability to withstand the rigors of vacuum deposition and prolonged operation. Patent CN101343539B introduces a groundbreaking class of organic luminescent materials featuring a carbazolyl stilbene derivative structure, specifically designed to address the critical bottlenecks of low glass transition temperature (Tg) and aggregation-caused quenching found in earlier generations of blue and green emitters. This proprietary synthesis route leverages robust N-arylation and olefination chemistries to construct complex molecular architectures that exhibit aggregation-induced emission (AIE) characteristics, ensuring high brightness even in the solid state. By integrating bulky carbazole units with rigid stilbene linkers, the resulting compounds achieve Tg values significantly higher than industry standards, directly translating to enhanced device longevity and operational stability for display and lighting manufacturers seeking reliable OLED material suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional small molecule OLED emitters, such as the widely used DPVBi, have historically struggled with inherent thermal instability, characterized by glass transition temperatures as low as 64°C. This thermal fragility leads to morphological changes in the emissive layer during device operation or fabrication, causing crystallization that drastically reduces luminous efficiency and shortens the operational lifespan of the display panel. Furthermore, many conventional fluorophores suffer from severe fluorescence quenching when transitioning from solution to the solid state, a phenomenon known as aggregation-caused quenching (ACQ), which necessitates complex doping strategies to maintain performance. The synthesis of these older materials often involves multi-step sequences with difficult purification requirements, frequently relying on preparative column chromatography that is impractical for ton-scale manufacturing, thereby inflating costs and limiting supply chain reliability for high-purity electronic chemical manufacturing.
The Novel Approach
The innovative methodology outlined in the patent data circumvents these issues by constructing a rigid, sterically hindered backbone that physically prevents molecular packing and crystallization, thereby elevating the glass transition temperature to over 170°C in many embodiments. This approach utilizes a modular synthetic strategy where carbazole units are first attached to a central benzophenone core via nucleophilic aromatic substitution, creating a stable ketone intermediate that serves as a versatile building block. Subsequent conversion of this ketone into a stilbene derivative through Wittig or Horner-Wadsworth-Emmons olefination introduces the necessary conjugation for luminescence while maintaining the steric bulk required for amorphous film formation. Crucially, the final products exhibit aggregation-induced emission, meaning their fluorescence intensity actually increases in the aggregated solid state, effectively turning a traditional liability of solid-state physics into a performance asset for next-generation optoelectronic devices.
Mechanistic Insights into N-Arylation and Wittig Olefination
The core of this synthesis relies on a highly efficient N-arylation reaction where the nitrogen atom of the carbazole ring attacks the electron-deficient aromatic ring of a di-fluorobenzophenone derivative. This nucleophilic aromatic substitution is facilitated by strong bases like potassium tert-butoxide in polar aprotic solvents such as DMF at elevated temperatures around 110°C. The mechanism proceeds through a Meisenheimer complex intermediate, where the electron-withdrawing carbonyl group activates the fluorine leaving groups for displacement by the carbazolyl anion. This step is critical for establishing the thermal stability of the final molecule, as the resulting C-N bonds are robust and the bulky carbazole substituents provide significant steric hindrance. The reaction conditions are optimized to ensure complete conversion of the starting materials, minimizing the presence of mono-substituted impurities that could act as charge traps in the final OLED device, thus ensuring a clean impurity profile essential for high-performance electronic materials.
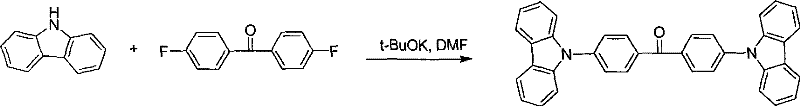
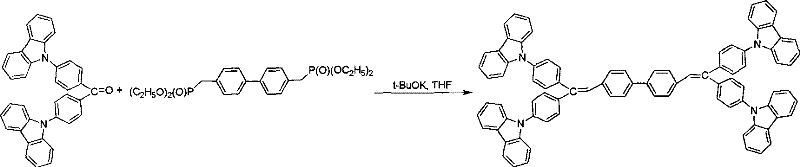
Following the formation of the ketone intermediate, the synthesis proceeds via a carbon-carbon bond-forming olefination to generate the stilbene double bond. This transformation typically employs phosphonate esters which, upon deprotonation by a base like t-BuOK in dry THF, generate a reactive ylide species. This ylide attacks the carbonyl carbon of the ketone intermediate, forming a betaine or oxaphosphetane intermediate that subsequently collapses to release the phosphine oxide byproduct and form the desired alkene. The stereochemistry of this reaction can be tuned to favor the E-isomer, which generally offers better planarity and conjugation for improved charge transport properties. The use of phosphonate reagents rather than traditional phosphonium salts often provides better control over the reaction and easier removal of byproducts, as the phosphine oxides formed are often less soluble or can be washed away, simplifying the downstream purification process significantly compared to traditional Wittig protocols.
How to Synthesize Carbazolyl Stilbene Derivatives Efficiently
The synthesis protocol described in the patent offers a streamlined pathway that is highly amenable to scale-up, beginning with the preparation of the bis-carbazolyl ketone intermediate followed by the final olefination step. The process is designed to minimize unit operations, utilizing precipitation and filtration as the primary isolation techniques rather than resource-intensive chromatography. This operational simplicity is a key factor in reducing the overall cost of goods sold (COGS) and improving the environmental footprint of the manufacturing process. For detailed standard operating procedures regarding stoichiometry, specific temperature ramps, and workup protocols, please refer to the structured guide below which outlines the critical process parameters derived from the experimental embodiments.
- Perform N-arylation by reacting carbazole with difluorobenzophenone in DMF using potassium tert-butoxide at elevated temperatures to form the bis-carbazolyl ketone intermediate.
- Conduct a Wittig or Horner-Wadsworth-Emmons reaction between the ketone intermediate and a bis-phosphonate ylide reagent in dry THF under argon protection.
- Isolate the final product via filtration and washing with solvents like ethanol or acetone, avoiding complex column chromatography for scalable purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic advantages, primarily driven by the accessibility of raw materials and the simplicity of the purification workflow. The starting materials, such as carbazole and difluorobenzophenone, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency and ensuring supply continuity even during market fluctuations. The elimination of complex chromatographic purification steps not only reduces solvent consumption and waste disposal costs but also drastically shortens the production cycle time, allowing for faster turnaround on custom orders and improved responsiveness to market demand. This efficiency translates directly into a more resilient supply chain capable of supporting the high-volume requirements of the consumer electronics and display industries without compromising on the stringent purity specifications required for electronic grade materials.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions like Suzuki or Heck couplings for similar structures, replacing them with cost-effective base-mediated reactions. By avoiding precious metals like palladium, the process removes the necessity for rigorous and costly metal scavenging steps to meet ppm-level impurity limits, resulting in significant savings in both reagent costs and processing time. Furthermore, the high yields reported in the embodiments, often exceeding 75% for the final coupling step, maximize atom economy and reduce the volume of raw materials needed per kilogram of finished product, driving down the variable cost of production substantially.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate standard industrial solvents and do not require extreme cryogenic temperatures or ultra-high vacuum, makes the process highly transferable to existing multipurpose chemical manufacturing facilities. This flexibility allows for rapid qualification of contract manufacturing organizations (CMOs) and diversifies the potential manufacturing base, reducing the lead time for high-purity OLED material production. The stability of the intermediates also allows for potential stocking strategies, where the ketone precursor can be synthesized and stored in bulk, decoupling the upstream supply chain from the final formulation steps and providing a buffer against demand volatility.
- Scalability and Environmental Compliance: The reliance on precipitation and filtration for product isolation significantly reduces the volume of organic waste generated compared to column chromatography, aligning with increasingly strict environmental regulations and corporate sustainability goals. The solvents used, such as THF, ethanol, and acetone, are readily recyclable through standard distillation processes, further enhancing the green chemistry profile of the manufacture. This ease of scale-up ensures that the transition from gram-scale R&D batches to multi-kilogram pilot runs and eventually to metric ton commercial production can be achieved with minimal process re-engineering, securing a stable long-term supply for downstream device manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of these carbazolyl stilbene derivatives in industrial applications. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for R&D and procurement professionals evaluating this technology for integration into their supply chains.
Q: What is the primary advantage of carbazolyl stilbene derivatives in OLED devices?
A: These materials exhibit high glass transition temperatures (Tg), often exceeding 170°C, which ensures thermal stability during vacuum deposition and significantly extends device lifespan compared to traditional materials like DPVBi.
Q: How does the aggregation-induced emission (AIE) property benefit solid-state applications?
A: Unlike conventional fluorophores that suffer from quenching in the solid state, AIE-active carbazolyl stilbenes maintain or increase luminous efficiency when aggregated, making them ideal for the solid thin films used in OLED emissive layers.
Q: Is the synthesis process suitable for large-scale commercial production?
A: Yes, the patented method utilizes straightforward N-arylation and olefination reactions with easy purification steps like precipitation and filtration, eliminating the need for expensive and time-consuming chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazolyl Stilbene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance luminescent materials play in the advancement of OLED technology, and we are uniquely positioned to support your development and production needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of carbazolyl stilbene derivative meets the exacting standards required for electronic chemical manufacturing, providing you with a reliable OLED material supplier partner you can trust.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this thermally stable material platform. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your supply chain is optimized for both performance and cost-efficiency in the competitive landscape of organic electronics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
