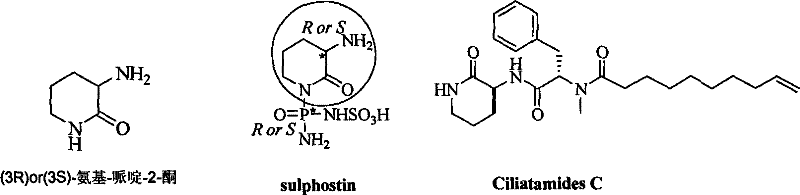
Scalable Asymmetric Synthesis of Ciliatamides C for Commercial Pharmaceutical Production
Scalable Asymmetric Synthesis of Ciliatamides C for Commercial Pharmaceutical Production
The pharmaceutical industry constantly seeks robust, scalable routes for complex marine natural products that exhibit potent biological activity. Patent CN101270076B introduces a groundbreaking asymmetric synthetic technology for the preparation of (3S, 9S)-Ciliatamides C, a marine natural product known for its inhibitory activity against HeLa cell toxicity. This patent represents a significant leap forward in the field of chiral piperidine compounds, shifting the paradigm from expensive, scarce starting materials to abundant, natural chiral pools. By utilizing L-Glutamic acid or D-Glutamic acid as the foundational raw material, this method addresses the critical bottlenecks of cost and availability that have historically limited the clinical research and commercial application of such potent compounds. The technical breakthrough lies not only in the high yield and excellent stereoselectivity but also in the operational simplicity that facilitates easier separation and purification processes. For R&D directors and procurement managers alike, this patent offers a viable pathway to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity compounds without the prohibitive costs associated with traditional extraction or complex synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of the key fragment (3R) or (3S)-amido-piperidine-2-one relied heavily on Ornithine as the starting raw material, a strategy fraught with significant chemical and economic disadvantages. The conventional first strategy involved the use of classical thionyl chloride for esterification followed by ring-closure under strongly alkaline conditions, a process that often resulted in substantial product solubility in the aqueous phase, thereby creating immense difficulty in separation and recovery. Furthermore, the second strategy, which utilized acetyl chloride for protection and sodium methylate for cyclization, while avoiding some isolation difficulties, still suffered from the inherent high cost and limited availability of non-natural D-Ornithine required for the R-configuration synthesis. Both traditional strategies shared common critical shortcomings, including the tendency for product racemization under highly basic ring-closure conditions, which drastically reduced the overall yield and optical purity. Additionally, the selectivity for amino protection in the product molecule was notoriously poor, leading to complex impurity profiles that required extensive and costly downstream purification. These technical barriers effectively limited the commercial viability of producing Ciliatamides C, making it difficult for any reliable pharmaceutical intermediates supplier to offer the compound at a price point suitable for widespread drug development.
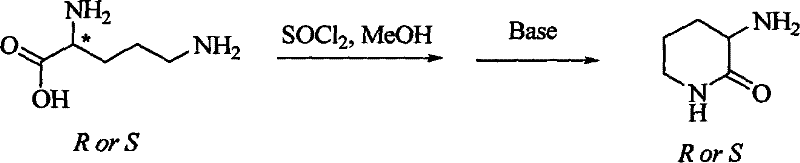
The Novel Approach
In stark contrast to the limitations of Ornithine-based routes, the novel approach detailed in CN101270076B leverages the abundant and inexpensive natural compounds L-Glutamic acid or D-Glutamic acid as the starting raw materials. This strategic shift to a chiral pool synthesis not only drastically simplifies the sourcing of raw materials but also inherently establishes the correct stereochemistry from the outset, minimizing the risk of racemization. The new method employs a series of mild and controlled reactions, including Boc protection, selective reduction, and azide substitution, which collectively ensure high stereoselectivity and operational safety. By avoiding the use of harsh reagents like thionyl chloride and strong bases for cyclization, the process significantly reduces the formation of by-products and simplifies the work-up procedures. This results in a synthetic route that is not only chemically elegant but also economically superior, offering substantial cost savings in pharmaceutical intermediates manufacturing. The ability to synthesize both (3R) and (3S) configurations from readily available glutamic acid enantiomers provides unparalleled flexibility for producing diverse analogs, making this method a cornerstone for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Glutamic Acid-Derived Cyclization
The core of this synthetic breakthrough lies in the meticulous transformation of the linear glutamic acid chain into the cyclic piperidine structure through a sequence of highly selective steps. The process begins with the esterification and Boc-protection of the amino group, which serves to mask the reactivity of the amine while activating the carboxylic acid for subsequent reduction. The reduction of the distal carboxylic acid to a primary alcohol, followed by mesylation, creates an excellent leaving group that is primed for nucleophilic substitution. The subsequent displacement of the mesylate with sodium azide is a critical step that introduces the nitrogen atom required for ring closure while inverting the configuration at the chiral center if necessary, ensuring the correct stereochemical outcome. Finally, the catalytic hydrogenation of the azide group generates a free amine in situ, which immediately attacks the activated ester to form the six-membered piperidine-2-one ring. This intramolecular cyclization is driven by the thermodynamic stability of the ring and the proximity of the reacting groups, resulting in high yields and minimal epimerization. The use of mild acidic conditions for the final deprotection step ensures that the sensitive piperidine ring remains intact, preserving the optical purity of the final product.
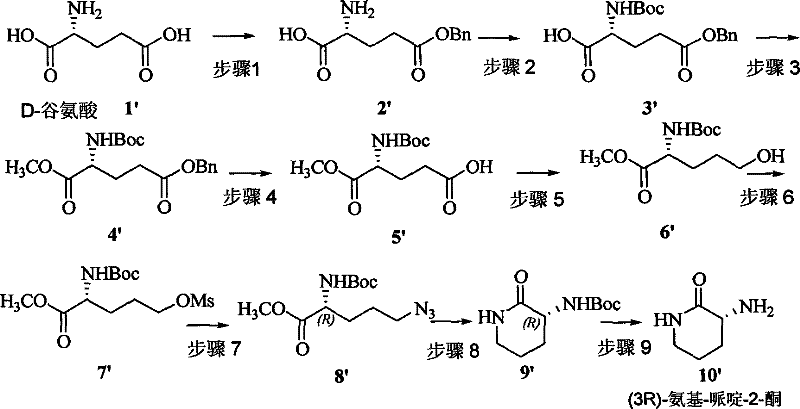
Impurity control is another critical aspect of this mechanism, as the stepwise nature of the synthesis allows for the removal of by-products at each stage before they can propagate through the sequence. The use of column chromatography after key steps, such as the methylation and azide substitution, ensures that only the desired intermediates proceed to the next reaction, thereby maintaining a high level of chemical integrity throughout the process. The selection of solvents like DMF, methanol, and ethyl acetate is optimized to maximize solubility of the intermediates while facilitating easy extraction and separation of inorganic salts and reagents. Furthermore, the avoidance of strong bases during the cyclization step prevents the base-catalyzed racemization that plagues conventional methods, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications. This rigorous control over the reaction environment and intermediate quality underscores the robustness of the method, making it an ideal candidate for technology transfer to large-scale production facilities where consistency and reliability are paramount.
How to Synthesize (3S)-amido-piperidine-2-one Efficiently
The synthesis of this critical chiral building block is achieved through a streamlined nine-step sequence that transforms inexpensive L-Glutamic acid into the target piperidine derivative with high efficiency. The process begins with the protection of the amino group and esterification of the carboxylic acid, followed by a series of functional group interconversions that prepare the molecule for cyclization. Key operations include the reduction of the side-chain acid to an alcohol, activation as a mesylate, and displacement with azide, culminating in a reductive cyclization that forms the piperidine ring. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, temperatures, and work-up procedures required to replicate the high yields reported in the patent embodiments. This route is designed to be scalable, utilizing common laboratory and industrial equipment without the need for specialized high-pressure or cryogenic setups.
- Protect L-Glutamic Acid via esterification and Boc-protection to establish the chiral backbone.
- Perform selective reduction and mesylation to prepare the chain for cyclization.
- Execute azide substitution and subsequent catalytic hydrogenation to form the piperidine ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers transformative advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring continuity of supply for critical drug intermediates. The shift from expensive, specialty amino acids like Ornithine to commodity chemicals like Glutamic acid represents a fundamental reduction in raw material costs, which directly impacts the bottom line of the manufacturing process. Additionally, the simplified operational conditions, which avoid hazardous reagents and extreme temperatures, reduce the safety burden and energy consumption associated with production, further driving down the overall cost of goods. The high yields and excellent stereoselectivity reported in the patent mean that less raw material is wasted, and less time is spent on purification, leading to a more efficient use of manufacturing capacity. For supply chain leaders, the availability of Glutamic acid from multiple global suppliers ensures that production is not held hostage by the scarcity of a single specialized starting material, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive non-natural amino acids and hazardous reagents like thionyl chloride leads to significant cost optimization in the production of chiral piperidine compounds. By utilizing readily available Glutamic acid, manufacturers can avoid the price volatility associated with specialty fine chemicals, ensuring a more stable and predictable cost structure. The high yields achieved at each step minimize material loss, meaning that less starting material is required to produce the same amount of final product, which amplifies the cost savings. Furthermore, the simplified purification processes reduce the consumption of solvents and chromatography media, contributing to lower operational expenses and a smaller environmental footprint.
- Enhanced Supply Chain Reliability: Sourcing L-Glutamic acid or D-Glutamic acid is significantly easier and more reliable than sourcing Ornithine, as Glutamic acid is a high-volume commodity chemical produced globally for the food and feed industries. This abundance ensures that manufacturers can secure long-term supply contracts without the risk of shortages that often plague niche chemical markets. The robustness of the synthetic route also means that production can be easily scaled up or shifted between different manufacturing sites without the need for specialized equipment or highly trained personnel. This flexibility allows supply chain managers to build a more resilient network of suppliers, reducing the risk of disruption and ensuring consistent delivery of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The use of mild reaction conditions and common solvents makes this process highly amenable to scale-up from laboratory to commercial production without significant re-engineering. The avoidance of heavy metals and hazardous gases simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. The high atom economy of the route, driven by the efficient use of the chiral pool starting material, minimizes the generation of chemical waste, aligning with the principles of green chemistry. This environmental compatibility not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing operation, making it more attractive to environmentally conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, based on the detailed data provided in the patent specifications. These answers are designed to clarify the feasibility of the process for large-scale manufacturing and to highlight the specific advantages over traditional methods. Understanding these details is crucial for technical teams evaluating the potential for technology transfer and for procurement teams assessing the long-term viability of the supply chain.
Q: Why is L-Glutamic Acid preferred over Ornithine for this synthesis?
A: L-Glutamic Acid is a naturally abundant, low-cost commodity chemical, whereas Ornithine is significantly more expensive and often requires complex sourcing. The glutamic acid route also avoids harsh thionyl chloride conditions, reducing safety risks and racemization.
Q: What is the stereoselectivity of the new cyclization method?
A: The patent describes excellent stereoselectivity, leveraging the inherent chirality of the starting amino acid. This minimizes the formation of unwanted enantiomers, simplifying downstream purification and ensuring high optical purity for the final API intermediate.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method utilizes common solvents like methanol and ethyl acetate and avoids cryogenic conditions or exotic catalysts. The operational simplicity and high yields described in the embodiments indicate strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ciliatamides C Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent technologies like CN101270076B into commercial reality, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in the nuances of asymmetric synthesis and chiral pool strategies, ensuring that the high stereoselectivity and yields described in the patent are maintained during scale-up. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Ciliatamides C or its intermediates meets the exacting standards required for pharmaceutical development. Our commitment to technical excellence means that we can navigate the challenges of process optimization, from solvent recovery to impurity profiling, to deliver a product that is both cost-effective and of the highest quality.
We invite you to collaborate with us to unlock the full potential of this innovative synthetic route for your drug development pipeline. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that identifies specific opportunities to optimize your supply chain based on this technology. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. Let us help you reduce lead time for high-purity pharmaceutical intermediates and secure a competitive advantage in the global market through our proven expertise in process chemistry and manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
