Advanced Rhodium Catalysis for Scalable Polysubstituted Aminoisoquinoline Manufacturing
Advanced Rhodium Catalysis for Scalable Polysubstituted Aminoisoquinoline Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high efficiency and purity. Patent CN111808071B discloses a groundbreaking method for synthesizing polysubstituted aminoisoquinoline compounds through the rhodium-catalyzed cyclization of pyridine derivatives and alkynes. This technology leverages pentamethylcyclopentadienyl rhodium complexes to facilitate C-H bond activation under relatively mild conditions, offering a significant departure from traditional synthetic routes that often suffer from harsh requirements and limited substrate scope. For R&D directors and procurement specialists, this innovation represents a pivotal shift towards more sustainable and cost-effective manufacturing of valuable isoquinoline intermediates, which are ubiquitous in drug molecules and natural products. The ability to achieve high selectivity and yield without extreme conditions positions this chemistry as a cornerstone for next-generation API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for isoquinoline derivatives predominantly rely on intramolecular cyclization of pre-functionalized substrates, a strategy that imposes severe constraints on molecular design and process economics. These conventional methods typically demand rigorous reaction conditions, including high temperatures and the use of sensitive reagents that necessitate strict anhydrous and anaerobic environments, thereby inflating operational costs and safety risks. Furthermore, the requirement for specific functional groups on the starting materials limits the versatility of the synthesis, often resulting in poor reaction activity and narrow substrate applicability. The generation of significant by-products in these older processes complicates downstream purification, leading to lower overall yields and increased waste disposal burdens that are increasingly untenable in modern green chemistry frameworks. Consequently, the industry has long sought a catalytic system capable of bypassing these limitations through direct C-H functionalization.
The Novel Approach
The novel approach detailed in the patent utilizes a rhodium-catalyzed system that directly activates C-H bonds on 2-aminopyridine derivatives, enabling their coupling with internal alkynes to form the desired isoquinoline core with exceptional efficiency. By employing a directing group strategy involving carbonyl moieties and sterically hindered groups like methyls, the process effectively controls regioselectivity and prevents the pyridine nitrogen from coordinating with and deactivating the metal catalyst. This mechanistic refinement allows the reaction to proceed smoothly in common organic solvents at temperatures ranging from 30°C to 150°C, eliminating the need for extreme conditions. The result is a streamlined process that delivers target products with selectivity exceeding 90% and minimizes by-product formation, drastically reducing the complexity and cost of purification steps required to obtain high-purity final materials.
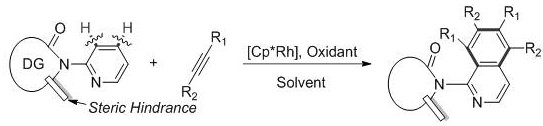
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this technological breakthrough lies in the sophisticated interplay between the rhodium catalyst and the engineered substrate architecture, specifically designed to overcome the inherent coordination issues of pyridine nitrogen. The introduction of a directing group (DG), such as a carbonyl in indolone or isoindolone structures, guides the rhodium center to the specific C-H bond intended for activation, ensuring precise regiocontrol during the cyclization event. Simultaneously, the incorporation of bulky steric hindrance groups, such as gem-dimethyl motifs, plays a critical dual role: it not only enhances the reactivity of the proximal C-H bond but also physically shields the pyridine nitrogen, preventing it from acting as a catalyst poison. This delicate balance allows the catalytic cycle to turnover efficiently, facilitating the insertion of the alkyne and subsequent reductive elimination to forge the new carbon-carbon bonds that construct the polysubstituted isoquinoline framework.
From an impurity control perspective, this mechanism offers distinct advantages by suppressing side reactions that typically plague transition metal-catalyzed processes. The high selectivity observed, consistently above 90% across various substrate combinations, indicates that the catalyst system effectively discriminates between competing reaction pathways, minimizing the formation of regioisomers or oligomeric by-products. The use of oxidants like silver acetate or copper salts further ensures the regeneration of the active rhodium species without introducing excessive oxidative degradation of the sensitive organic substrates. For quality control teams, this translates to a cleaner reaction profile where the primary challenge shifts from separating complex mixtures to simply removing residual catalyst and solvent, thereby simplifying the analytical burden and ensuring consistent batch-to-batch purity profiles essential for regulatory compliance.
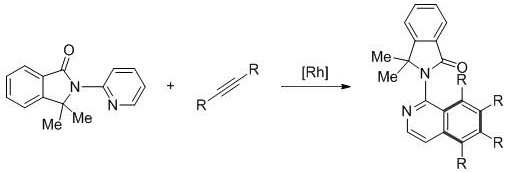
How to Synthesize Polysubstituted Aminoisoquinoline Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable heterocycles, emphasizing operational simplicity and broad substrate tolerance. The process begins with the combination of 2-aminopyridine derivatives and internal alkynes in a molar ratio typically between 1:2 and 1:5, dissolved in a suitable organic solvent such as 1,2-dichloroethane or toluene. To this mixture, a pentamethylcyclopentadienyl rhodium complex catalyst is added at a loading of 1-10 mol%, along with a stoichiometric amount of an oxidant like silver acetate to drive the catalytic cycle. The reaction is then heated to temperatures between 30°C and 150°C for a duration of 1 to 24 hours, depending on the specific reactivity of the substrates involved, after which the solvent is removed and the product is purified via standard silica gel chromatography.
- Combine 2-aminopyridine derivatives and internal alkyne compounds in a molar ratio of 1: 2 to 1:5 within an organic solvent such as 1,2-dichloroethane.
- Add a pentamethylcyclopentadienyl rhodium complex catalyst (1-10 mol%) and a suitable oxidant like silver acetate to the reaction mixture.
- Heat the reaction mixture to temperatures between 30°C and 150°C for 1 to 24 hours, then purify the crude product via silica column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed methodology offers tangible benefits that extend far beyond the laboratory bench, directly impacting the bottom line through process intensification and risk mitigation. The elimination of harsh reaction conditions and the tolerance for a wide range of functional groups mean that raw material sourcing becomes more flexible, allowing for the use of commercially available building blocks without the need for expensive custom synthesis of pre-functionalized precursors. This flexibility significantly reduces the lead time associated with raw material acquisition and lowers the overall cost of goods sold by minimizing the number of synthetic steps required to reach the target intermediate. Furthermore, the high selectivity and reduced by-product profile translate into higher throughput and less waste generation, aligning perfectly with sustainability goals and reducing the environmental compliance costs associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The streamlined nature of this catalytic process eliminates the need for multiple protection and deprotection steps often required in traditional isoquinoline synthesis, resulting in substantial cost savings in terms of reagents, labor, and time. By avoiding the use of expensive pre-functionalized substrates and reducing the complexity of the purification workflow, manufacturers can achieve a more economical production model that remains competitive even at large scales. The ability to run reactions in common solvents without strict inert atmosphere requirements further lowers capital expenditure on specialized equipment and utility costs, making the process economically viable for both pilot and commercial-scale operations.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-aminopyridine derivatives and internal alkynes ensures a stable and resilient supply chain that is less susceptible to disruptions caused by the scarcity of exotic reagents. The robustness of the reaction conditions, which do not demand ultra-dry or oxygen-free environments, reduces the risk of batch failures due to environmental factors, thereby enhancing overall production reliability and on-time delivery performance. This stability allows supply chain planners to forecast production schedules with greater confidence, ensuring a continuous flow of high-quality intermediates to downstream API manufacturing sites without unexpected delays.
- Scalability and Environmental Compliance: The mild reaction parameters and simple workup procedure make this technology highly scalable, facilitating a smooth transition from gram-scale laboratory optimization to multi-ton commercial production without significant process re-engineering. The reduction in by-product formation and the use of less hazardous reagents contribute to a greener manufacturing footprint, helping companies meet increasingly stringent environmental regulations and corporate sustainability targets. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible and forward-thinking partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their existing manufacturing portfolios. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is critical for assessing the feasibility of adopting this method for specific project requirements and for anticipating any potential challenges during the technology transfer phase.
Q: What represents the primary advantage of this rhodium-catalyzed method over traditional synthesis?
A: The primary advantage lies in the utilization of C-H bond activation which avoids the need for pre-functionalized substrates, thereby significantly simplifying the synthetic route and improving atom economy compared to traditional intramolecular cyclization methods.
Q: How does the process ensure high selectivity for the target polysubstituted product?
A: High selectivity exceeding 90% is achieved through the strategic introduction of directing groups such as carbonyls and sterically hindered groups like methyls, which effectively regulate reaction activity and prevent catalyst poisoning by the pyridine nitrogen.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly suitable for industrial scale-up due to its mild reaction conditions that do not require strict anhydrous or anaerobic environments, coupled with a straightforward workup procedure involving simple solvent removal and column purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Aminoisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the rhodium-mediated C-H activation described in CN111808071B for the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in a practical, GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of polysubstituted aminoisoquinoline meets the exacting standards required by global regulatory bodies, providing our partners with the confidence needed to advance their drug development programs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs, offering a Customized Cost-Saving Analysis that quantifies the potential economic benefits for your supply chain. By requesting specific COA data and route feasibility assessments from our experts, you can gain a deeper understanding of how our capabilities align with your requirements for high-purity isoquinoline derivatives. Let us collaborate to optimize your manufacturing strategy, reduce lead times, and secure a reliable supply of critical intermediates that drive your success in the competitive pharmaceutical marketplace.
