Advanced Ion-Exchange Synthesis of D-Calcium Pantothenate for Commercial Scale Vitamin Production
The global demand for high-purity vitamins and nutritional supplements continues to surge, driving the need for more efficient and environmentally sustainable manufacturing processes. Patent CN101948402B introduces a groundbreaking methodology for the preparation of D-calcium pantothenate, widely known as Vitamin B5, which addresses critical bottlenecks in traditional synthesis routes. This innovative approach replaces conventional calcification agents with a sophisticated ion-exchange technique, fundamentally altering the reaction landscape to eliminate water generation during the crucial acylation phase. By leveraging 732-type calcium cationic exchange resins, the process achieves a cleaner reaction profile that significantly enhances yield and simplifies downstream purification. For industry leaders seeking a reliable vitamin B5 supplier, understanding this technological shift is paramount, as it represents a move towards greener chemistry without compromising on the stringent purity specifications required for pharmaceutical and food-grade applications. The implications of this patent extend beyond mere laboratory success, offering a robust framework for industrial scalability that aligns with modern regulatory and environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of D-calcium pantothenate has relied heavily on the reaction between D-pantoyl lactone and calcium beta-alanine, where the latter is typically prepared using calcium oxide or calcium hydroxide as the calcifying agent. While these alkaline earth metal compounds are inexpensive and readily available, their usage introduces a significant chemical disadvantage: the generation of water as a stoichiometric byproduct during the salt formation. This incidental water presence creates a hostile environment for the subsequent acylation reaction, which is highly sensitive to moisture and requires strictly anhydrous conditions to proceed efficiently. Consequently, manufacturers often struggle with suboptimal reaction kinetics and reduced conversion rates, typically capping overall yields at approximately 80%, which is economically inefficient for large-scale operations. Furthermore, the presence of water necessitates energy-intensive drying steps and complicates the crystallization process, leading to potential issues with product morphology and purity that can delay batch release times.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent circumvents these moisture-related challenges by decoupling the calcium introduction from the initial nitrile hydrolysis step. Instead of directly forming the calcium salt with lime, the process first hydrolyzes 3-aminopropionitrile using sodium hydroxide or potassium hydroxide to form the corresponding sodium or potassium salt of beta-alanine. This intermediate is then subjected to a cationic exchange process using a specialized 732-type resin loaded with calcium ions, effectively swapping the alkali metal for calcium in a controlled, anhydrous methanol medium. This strategic pivot ensures that no water is produced during the formation of the reactive calcium beta-alanine species, thereby creating ideal conditions for the subsequent acylation with D-pantoyl lactone. The result is a streamlined workflow that not only boosts theoretical yields but also drastically simplifies the operational complexity, making it an attractive option for cost reduction in nutritional ingredients manufacturing by minimizing waste and energy consumption.
Mechanistic Insights into Ion-Exchange Mediated Acylation
The core of this synthetic breakthrough lies in the precise manipulation of ionic species within a methanol solvent system, leveraging the high charge density of the 732-type cationic exchange resin. The process initiates with the base-catalyzed hydrolysis of 3-aminopropionitrile, where the nitrile group is converted into a carboxylate salt under elevated temperatures ranging from 96°C to 100°C. As illustrated in the reaction scheme below, the interaction between the nitrile and the strong base (NaOH or KOH) proceeds smoothly to generate 3-alanine sodium or 3-alanine potassium, releasing ammonia which is subsequently removed via vacuum distillation to prevent interference with downstream steps.
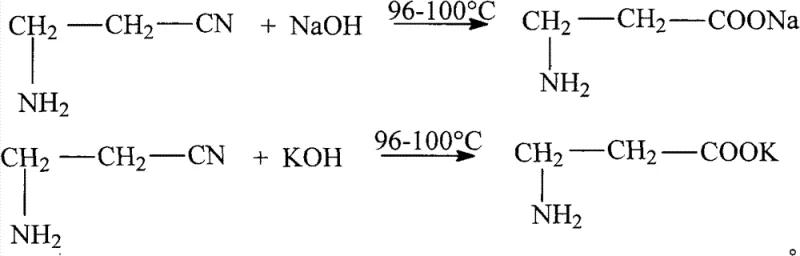
Following the hydrolysis and pH adjustment, the crude salt mixture is dissolved in methanol, exploiting the differential solubility where the organic alanine salts remain in solution while inorganic halides precipitate out and are filtered away. The filtrate, now a purified methanol solution of the alkali metal alanine salt, is passed through the calcium-loaded resin. The resin's sulfonic acid groups, pre-saturated with calcium ions, exhibit a strong affinity for the alanine anions, facilitating a rapid ion exchange that releases the calcium salt of beta-alanine into the methanol phase. This high-purity D-calcium pantothenate precursor is then reacted with D-pantoyl lactone at controlled low temperatures between 0°C and 10°C. The absence of water allows the ring-opening acylation to proceed with high fidelity, minimizing side reactions such as hydrolysis of the lactone, which is a common impurity source in traditional wet methods. Finally, crystallization is induced at temperatures between -15°C and -10°C, yielding the final product with exceptional structural integrity and minimal residual solvents.
How to Synthesize D-Calcium Pantothenate Efficiently
Implementing this advanced synthesis route requires careful attention to stoichiometry and thermal management to ensure reproducibility and safety at scale. The patent outlines a specific sequence where the molar ratio of 3-aminopropionitrile to the hydroxide base is maintained between 1:1.1 and 1:1.4 to ensure complete conversion while minimizing excess base that would require neutralization. Detailed standard operating procedures regarding the loading capacity of the resin, the flow rate of the methanol solution, and the precise timing of the crystallization seeding are critical for maximizing the efficiency of the commercial scale-up of complex vitamins. Operators must adhere strictly to the temperature gradients specified, particularly during the exothermic hydrolysis and the endothermic crystallization phases, to maintain batch-to-batch consistency.
- Hydrolyze 3-aminopropionitrile with NaOH or KOH at 96-100°C to form 3-alanine salts.
- Purify the resulting salt mixture via methanol dissolution and filtration to remove inorganic halides.
- Perform ion exchange using 732-type calcium resin to convert the sodium/potassium salt to 3-alanine calcium.
- React the 3-alanine calcium solution with D-pantolactone at low temperature (-15 to -10°C) to crystallize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ion-exchange technology offers compelling strategic advantages that extend well beyond simple yield improvements. By eliminating the need for calcium oxide or hydroxide in the primary reaction vessel, the process removes a major source of inorganic waste and simplifies the effluent treatment profile, aligning perfectly with increasingly stringent environmental regulations. The ability to recycle the 732-type calcium cationic exchange resin multiple times significantly reduces the consumption of consumable materials, translating into direct operational expenditure savings over the lifecycle of the production campaign. Moreover, the simplified purification steps, which rely on solubility differences rather than complex extraction or distillation sequences, reduce the overall processing time and equipment footprint required per kilogram of output.
- Cost Reduction in Manufacturing: The elimination of water generation during the calcification step removes the necessity for extensive drying protocols and azeotropic distillation, which are energy-intensive and costly. By avoiding the use of stoichiometric amounts of lime that generate solid waste, the process reduces the burden on waste disposal systems and lowers the cost associated with handling hazardous byproducts. Furthermore, the high selectivity of the ion-exchange resin minimizes the formation of difficult-to-remove impurities, reducing the need for expensive recrystallization cycles and increasing the overall throughput of the manufacturing facility.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to greater supply chain stability by reducing the risk of batch failures due to moisture sensitivity. Traditional methods often suffer from variability caused by ambient humidity or incomplete drying of reagents, whereas this anhydrous approach provides a more forgiving and consistent reaction environment. This reliability ensures that production schedules can be met with greater precision, reducing lead time for high-purity vitamins and allowing suppliers to respond more agilely to fluctuations in market demand without compromising on quality standards.
- Scalability and Environmental Compliance: The process is inherently scalable, as the ion-exchange step can be easily expanded by increasing resin bed volume without altering the fundamental reaction kinetics. The absence of heavy metal catalysts or toxic reagents simplifies the regulatory approval process for new facilities and ensures compliance with global food and pharmaceutical safety standards. Additionally, the recycling of the resin and the minimization of solvent usage through efficient recovery systems contribute to a lower carbon footprint, making the manufacturing process more sustainable and attractive to eco-conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel D-calcium pantothenate synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational parameters and benefits. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement specialists assessing the long-term value proposition of suppliers utilizing this technology.
Q: Why is the ion-exchange method superior to traditional calcium oxide calcification?
A: Traditional methods using calcium oxide or hydroxide generate water as a byproduct during the formation of calcium beta-alanine. This water interferes with the subsequent acylation reaction with D-pantolactone, often lowering yields to around 80%. The ion-exchange method avoids water generation entirely, ensuring a smoother acylation process and higher overall yields.
Q: Can the 732-type calcium cationic exchange resin be reused?
A: Yes, the process is designed for sustainability and cost-efficiency. The 732-type calcium cationic exchange resin can be recovered and recycled after the ion exchange step, significantly reducing solid waste generation and raw material consumption compared to single-use reagents.
Q: What are the critical temperature controls for the acylation step?
A: Precise temperature control is vital for product quality. The acylation reaction between 3-alanine calcium and D-pantolactone should occur between 0°C and 10°C, followed by crystallization at temperatures between -15°C and -10°C to maximize recovery and purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Calcium Pantothenate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic routes like the ion-exchange method described in CN101948402B for producing high-quality Vitamin B5. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical nutritional ingredients. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of D-calcium pantothenate adheres to the highest international pharmacopeial standards. We are committed to leveraging cutting-edge chemistry to deliver products that not only meet but exceed the performance expectations of the global pharmaceutical and food industries.
We invite you to collaborate with us to optimize your supply chain and achieve superior cost efficiencies through our advanced manufacturing capabilities. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and innovation in your vitamin B5 sourcing strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
