Advanced Metal-Free Synthesis of Polysubstituted 2-Cyclopentenyl Ethyl Acetate Intermediates for Pharma
Advanced Metal-Free Synthesis of Polysubstituted 2-Cyclopentenyl Ethyl Acetate Intermediates for Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex cyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN109232314B, which discloses a novel synthetic route for polysubstituted 2-cyclopentenyl ethyl acetate compounds. These structural motifs are ubiquitous in natural products and drug lead compounds exhibiting potent biological activities, including antifungal, anti-HIV, and thrombin inhibitory properties. The disclosed technology represents a paradigm shift from traditional transition-metal catalyzed processes to a more sustainable, base-mediated cascade reaction. By leveraging simple chain-like starting materials, this method directly assembles the cyclopentene core with high efficiency, bypassing the need for pre-functionalized cyclic precursors that often plague conventional synthetic strategies. This innovation not only streamlines the synthetic pathway but also aligns perfectly with the modern industry's demand for greener, more economical manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted cyclopentene frameworks has relied heavily on the use of noble metal catalysts, such as palladium, rhodium, or gold complexes, which drive cyclization reactions through sophisticated organometallic mechanisms. While effective in laboratory settings, these traditional approaches suffer from severe drawbacks when translated to industrial scale, primarily due to the exorbitant cost of the catalysts and the stringent requirement for their removal to meet pharmaceutical purity standards. Furthermore, conventional routes often necessitate multi-step sequences involving the preparation of specialized cyclic starting materials, which increases the overall process mass intensity and generates substantial chemical waste. The operational complexity is further compounded by the need for inert atmospheres, anhydrous conditions, and sensitive temperature controls, all of which contribute to higher capital expenditure and longer production lead times. Additionally, the presence of residual heavy metals in the final active pharmaceutical ingredient (API) is a critical regulatory concern, requiring additional downstream purification steps that erode profit margins and delay time-to-market for new drug candidates.
The Novel Approach
In stark contrast to the legacy methods, the technology described in patent CN109232314B introduces a streamlined, one-pot synthesis that utilizes readily available linear precursors to construct the target cyclopentene scaffold in a single operational sequence. This novel approach employs inexpensive inorganic bases, such as potassium carbonate or cesium carbonate, in common organic solvents like acetonitrile or acetone, thereby eliminating the dependency on precious metal catalysts entirely. The reaction initiates at ambient temperature and proceeds to completion upon mild heating to 80°C, demonstrating remarkable tolerance to various functional groups on the aromatic rings of the substrates. By avoiding the preparation of cyclic starting materials and utilizing a direct condensation-cyclization strategy, this method significantly reduces the number of unit operations and the associated solvent consumption. The simplicity of the work-up procedure, which involves standard extraction and flash column chromatography, ensures that the process is not only chemically efficient but also operationally straightforward, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their production portfolios.
Mechanistic Insights into Base-Catalyzed Cascade Cyclization
The core of this innovative synthesis lies in a meticulously orchestrated cascade reaction initiated by the deprotonation of active methylene compounds, specifically malononitrile or ethyl cyanoacetate, by the inorganic base. This initial step generates a nucleophilic carbanion species that attacks the electrophilic center of the ethyl 4-chloroacetoacetate, forming a key intermediate that sets the stage for the subsequent cyclization. The introduction of the substituted 1,2-allenone triggers a complex series of bond-forming events, including Michael addition and intramolecular aldol-type condensations, which ultimately forge the five-membered carbocyclic ring. The beauty of this mechanism is its reliance on thermodynamic driving forces rather than kinetic activation by expensive catalysts, allowing the reaction to proceed smoothly under relatively mild thermal conditions. The versatility of the system is evidenced by its compatibility with a wide range of substituents on the allenone moiety, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as cyano, chloro, and bromo, without compromising the integrity of the final product.
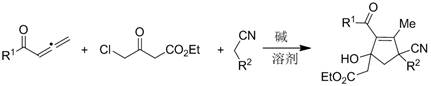
From an impurity control perspective, the mild nature of the reaction conditions plays a pivotal role in ensuring high product purity and minimizing the formation of difficult-to-remove byproducts. Unlike harsh acidic or strongly basic conditions that can lead to polymerization or decomposition of sensitive functional groups, the use of weak inorganic bases like potassium carbonate provides a buffered environment that favors the desired cyclization pathway. The absence of transition metals eliminates the risk of metal-catalyzed side reactions, such as oxidative coupling or isomerization, which are common pitfalls in noble metal-catalyzed processes. Furthermore, the high regioselectivity observed in the formation of the polysubstituted cyclopentene ring suggests a well-defined transition state that minimizes the generation of regioisomers. This inherent selectivity simplifies the downstream purification process, as the crude reaction mixture typically contains fewer impurities, allowing for efficient isolation of the target compound via standard silica gel chromatography. For R&D teams focused on process development, this mechanistic clarity offers a robust platform for optimizing reaction parameters to achieve even higher yields and purity profiles suitable for GMP manufacturing.
How to Synthesize Polysubstituted 2-Cyclopentenyl Ethyl Acetate Efficiently
The practical implementation of this synthesis is designed to be accessible to laboratories equipped with standard organic synthesis infrastructure, requiring no specialized high-pressure reactors or glovebox facilities. The protocol begins with the dissolution of the active methylene component and the chloroacetoacetate derivative in a polar aprotic solvent, followed by the addition of the base to generate the reactive nucleophile in situ. After a brief stirring period at room temperature to ensure complete formation of the initial adduct, the substituted 1,2-allenone is introduced, and the reaction mixture is heated to promote the cyclization cascade. Monitoring the reaction progress via thin-layer chromatography (TLC) allows for precise determination of the endpoint, typically achieved within 1.5 hours at 80°C. Upon completion, the reaction is quenched with a saturated ammonium chloride solution, and the product is extracted into an organic phase, dried, and concentrated to yield the crude material. For a detailed breakdown of the specific stoichiometric ratios, solvent volumes, and purification gradients used in the exemplary embodiments, please refer to the standardized synthesis guide below.
- Dissolve ethyl 4-chloroacetoacetate and malononitrile (or ethyl cyanoacetate) in an organic solvent such as acetonitrile or acetone.
- Add a base like potassium carbonate and stir at room temperature for 20 minutes to initiate the condensation reaction.
- Introduce the substituted 1,2-allenone, raise the temperature to 80°C, and react for 1.5 hours followed by standard extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis technology translates into tangible strategic advantages that directly impact the bottom line and operational resilience. The elimination of noble metal catalysts removes a significant variable cost component from the bill of materials, while also mitigating the supply risk associated with the fluctuating prices and geopolitical availability of metals like palladium and platinum. Moreover, the use of commodity chemicals such as potassium carbonate and acetonitrile ensures a stable and diversified supply base, reducing the likelihood of production stoppages due to raw material shortages. The simplified operational workflow, characterized by ambient temperature initiation and short reaction times, enhances throughput capacity and allows for better utilization of existing reactor assets without the need for costly retrofitting. These factors collectively contribute to a more agile and cost-competitive manufacturing model that can respond rapidly to market demands.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive transition metal catalysts, which traditionally account for a substantial portion of the raw material costs in complex heterocycle synthesis. By substituting these with low-cost inorganic bases, the direct material cost is drastically reduced, enabling more competitive pricing for the final intermediates. Additionally, the simplified work-up procedure eliminates the need for specialized scavenger resins or complex filtration steps required to remove trace metals, further lowering processing costs. The high yields reported across various substrate scopes indicate excellent atom economy, minimizing waste disposal costs and maximizing the output per batch. This economic efficiency makes the process highly attractive for cost reduction in pharmaceutical intermediate manufacturing, allowing companies to maintain healthy margins even in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk commodity chemicals for both reagents and solvents significantly de-risks the supply chain compared to processes dependent on specialized or proprietary catalysts. Ethyl 4-chloroacetoacetate, malononitrile, and substituted allenones are commercially accessible from multiple global suppliers, ensuring continuity of supply and preventing single-source bottlenecks. The robustness of the reaction conditions, which tolerate a variety of functional groups and do not require stringent anhydrous or inert environments, reduces the sensitivity of the process to minor variations in raw material quality. This resilience is crucial for maintaining consistent production schedules and meeting delivery commitments to downstream API manufacturers. Consequently, this method supports reducing lead time for high-purity pharmaceutical intermediates by streamlining the procurement and production phases.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, the process aligns well with green chemistry principles by avoiding toxic heavy metals and utilizing benign solvents that are easily recovered and recycled. The absence of metal residues simplifies the regulatory approval process for new drug applications, as there is no need to validate complex metal clearance protocols. The one-pot nature of the reaction reduces solvent consumption and energy usage compared to multi-step sequences, contributing to a lower carbon footprint for the manufacturing site. Scaling this process from kilogram to tonnage levels is straightforward due to the lack of exothermic hazards associated with metal catalysis and the use of standard heating equipment. This ease of commercial scale-up of complex pharmaceutical intermediates ensures that the technology can grow alongside the clinical development of the drug candidate, providing a seamless path from lab to plant.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived from the specific experimental data and advantages outlined in the patent literature. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines. The answers provided reflect the proven capabilities of the method as demonstrated in the cited examples, offering a realistic view of its performance and limitations. We encourage stakeholders to review these insights to facilitate informed decision-making regarding process adoption and partnership opportunities.
Q: Does this synthesis require expensive noble metal catalysts?
A: No, the patented method utilizes inexpensive inorganic bases like potassium carbonate or cesium carbonate, completely eliminating the need for costly palladium or platinum catalysts.
Q: What are the typical reaction conditions for this cyclization?
A: The reaction proceeds under mild conditions, starting at room temperature for 20 minutes and then heating to 80°C for 1.5 hours in solvents like acetonitrile.
Q: Is this method suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process features simple operation, high yields, and easy purification via flash column chromatography, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 2-Cyclopentenyl Ethyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology in advancing the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from bench-scale optimization to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this base-catalyzed cyclization, while our rigorous QC labs enforce stringent purity specifications to guarantee product quality that meets global regulatory standards. We are committed to leveraging this innovative chemistry to deliver cost-effective and reliable solutions for your complex synthesis needs.
We invite you to engage with our technical procurement team to discuss how this novel route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this process offers compared to your current supply chain. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to become your long-term partner in the successful commercialization of these critical cyclopentene building blocks.
