Advanced One-Step Rhodium Catalysis for Commercial Polyaryl Naphthol Derivatives Production
Advanced One-Step Rhodium Catalysis for Commercial Polyaryl Naphthol Derivatives Production
The development of efficient synthetic routes for highly conjugated aromatic systems is a critical priority for the modern electronic materials and fine chemicals sectors. Patent CN107382782B introduces a groundbreaking methodology for the synthesis of polyaryl substituted naphthol derivatives, leveraging transition metal-catalyzed carbon-hydrogen bond activation. This technology addresses a significant gap in the prior art, where direct functionalization of phenol derivatives into complex naphthalene scaffolds was previously unreported or required multi-step sequences with poor atom economy. By utilizing a robust rhodium catalytic system, this invention enables the direct coupling of phenol derivatives with diaryl acetylenes in a single operational step. For R&D directors and procurement strategists, this represents a pivotal shift towards more streamlined manufacturing processes that reduce waste and lower the overall cost of goods sold for high-value optoelectronic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polyaryl-substituted naphthalene cores has relied on indirect strategies that often involve pre-functionalized substrates or specific directing groups that are not native to simple phenols. Prior art, such as the work by the Miura group in 2008, utilized phenylpyrazole compounds as raw materials to access naphthyl pyrazole derivatives, necessitating the installation and subsequent removal of the pyrazole directing group. Similarly, reports from the Wu topic group in 2010 described palladium-catalyzed reactions of acetanilides, which limited the substrate scope to amine derivatives rather than the more versatile and abundant phenol class. These conventional approaches suffer from several inherent drawbacks: they require additional synthetic steps to install directing groups, generate stoichiometric amounts of chemical waste, and often exhibit limited tolerance for sensitive functional groups. Furthermore, the reliance on specific heterocyclic scaffolds restricts the structural diversity of the final products, making it difficult to fine-tune the photoelectric properties required for advanced display or semiconductor applications.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN107382782B employs a direct C-H activation strategy that transforms readily available phenol derivatives and diaryl acetylenes into complex polyaryl naphthols in a single pot. This novel approach eliminates the need for pre-functionalization of the phenol substrate beyond a simple protecting group strategy, significantly enhancing the atom economy of the process. The reaction proceeds under the synergistic action of a rhodium catalyst, a silver additive, and a copper oxidant, facilitating the simultaneous formation of multiple carbon-carbon bonds to construct the naphthalene core. As illustrated in the specific transformation below, the method tolerates a wide range of substituents, including alkyl, alkoxy, and halogen groups, allowing for precise modulation of the electronic properties of the final material. This direct route not only simplifies the synthetic workflow but also opens new avenues for the rapid discovery of novel organic photoelectric materials with tailored conductivity and fluorescence characteristics.
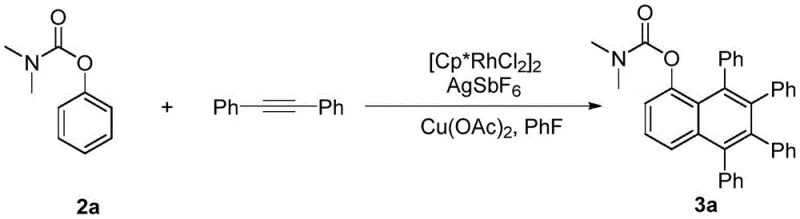
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this technological breakthrough lies in the sophisticated mechanism of rhodium(III)-catalyzed C-H bond activation, which serves as the driving force for the annulation reaction. The catalytic cycle initiates with the generation of a cationic rhodium species through the abstraction of chloride ligands by the silver additive, typically AgSbF6. This active catalyst then coordinates with the oxygen atom of the carbamate-protected phenol, directing the metal center to the ortho-position of the aromatic ring. Through a concerted metalation-deprotonation (CMD) pathway, the rhodium species activates the inert C-H bond, forming a stable five-membered rhodacycle intermediate. This step is critical as it determines the regioselectivity of the reaction, ensuring that the subsequent insertion of the alkyne occurs exclusively at the desired position adjacent to the oxygen substituent. The use of fluorobenzene as the solvent further stabilizes these cationic intermediates, promoting higher turnover frequencies and minimizing catalyst deactivation pathways that often plague transition metal catalysis in polar media.
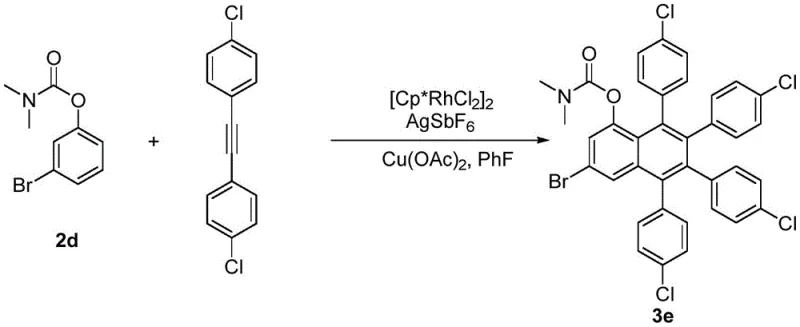
Following the C-H activation event, the diaryl acetylene substrate undergoes migratory insertion into the rhodium-carbon bond, extending the conjugated system and setting the stage for ring closure. The presence of the copper oxidant, such as Cu(OAc)2, is essential for the reductive elimination step that releases the final polyaryl naphthol product and regenerates the active Rh(III) catalyst. This redox-neutral cycle ensures that the reaction can proceed efficiently without the accumulation of inactive Rh(I) species. From an impurity control perspective, the specificity of the rhodium catalyst for the ortho-C-H bond minimizes the formation of regioisomers, while the mild reaction conditions (110-120 °C) prevent thermal degradation of the sensitive polyaryl framework. The versatility of this mechanism is further evidenced by its ability to accommodate electron-withdrawing groups on the alkyne, as seen in the synthesis of chlorophenyl-substituted derivatives, demonstrating the robustness of the catalytic system against electronic variations in the substrate.
How to Synthesize Polyaryl Substituted Naphthol Derivatives Efficiently
The practical implementation of this synthesis route is designed for scalability and reproducibility, making it highly attractive for industrial adoption. The process begins with the protection of the phenol starting material, a straightforward nucleophilic substitution that yields the dimethylcarbamoyl protected intermediate in high purity. This intermediate is then subjected to the key cyclization reaction under optimized conditions involving precise stoichiometric ratios of the rhodium catalyst (1-5 mol%) and silver additive (1-1.5 equivalents). The reaction is typically conducted in fluorobenzene at elevated temperatures to ensure complete conversion of the starting materials. Post-reaction workup is remarkably simple, involving filtration to remove metal residues followed by solvent evaporation and standard column chromatography. This streamlined protocol reduces the operational complexity typically associated with transition metal catalysis, allowing for seamless translation from laboratory benchtop to pilot plant scale. For detailed standardized synthesis steps, please refer to the guide below.
- Protect the phenol starting material using dimethylcarbamoyl chloride and potassium carbonate in acetonitrile to form the carbamate intermediate.
- Perform the key cyclization reaction by mixing the protected phenol with diaryl acetylene, [Cp*RhCl2]2 catalyst, AgSbF6 additive, and Cu(OAc)2 oxidant in fluorobenzene at 110 °C.
- Isolate the final polyaryl naphthol derivative through filtration, solvent removal, and purification via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this rhodium-catalyzed methodology offers substantial strategic advantages over traditional multi-step syntheses. The primary benefit lies in the drastic reduction of process steps; by consolidating the construction of the naphthalene core into a single transformation, manufacturers can significantly lower labor costs, energy consumption, and equipment occupancy time. The elimination of pre-functionalization steps means fewer raw materials need to be sourced and managed, simplifying the supply chain and reducing the risk of bottlenecks associated with specialized reagents. Furthermore, the high atom economy of the C-H activation process translates to less chemical waste generation, which directly correlates to lower disposal costs and a reduced environmental footprint. This aligns perfectly with modern green chemistry initiatives and regulatory compliance standards, positioning companies that adopt this technology as leaders in sustainable manufacturing practices within the fine chemicals sector.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the simplification of the synthetic route. By avoiding the need for expensive pre-functionalized building blocks and reducing the total number of unit operations, the overall cost of production is significantly optimized. The use of relatively inexpensive oxidants like copper acetate and the ability to recover and recycle the organic solvent further contribute to margin improvement. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, reducing the burden on downstream purification processes and increasing the overall yield of the valuable API intermediate or electronic material precursor.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as phenol derivatives and diaryl acetylenes ensures a stable and resilient supply chain. Unlike specialized heterocyclic directing groups that may have limited suppliers and long lead times, the starting materials for this process are widely available from multiple global sources. This diversification of supply mitigates the risk of production delays due to raw material shortages. Moreover, the robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor variations in temperature or reagent quality compared to more fragile enzymatic or low-temperature organometallic reactions.
- Scalability and Environmental Compliance: Scaling this process from grams to kilograms is facilitated by the homogeneous nature of the catalytic system and the use of standard organic solvents. The reaction does not require extreme pressures or cryogenic temperatures, allowing it to be run in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. From an environmental standpoint, the high efficiency of the C-H activation reduces the E-factor (mass of waste per mass of product), making it easier to meet stringent environmental regulations. The simplified workup procedure also reduces the volume of aqueous waste streams, lowering the cost and complexity of wastewater treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing portfolios and for procurement specialists assessing the long-term viability of the supply chain.
Q: What catalyst system is required for this naphthol synthesis?
A: The process utilizes a cationic rhodium(III) system generated in situ from [Cp*RhCl2]2 and AgSbF6, with Cu(OAc)2 serving as the terminal oxidant to drive the C-H activation cycle.
Q: Can this method tolerate halogen substituents on the phenol ring?
A: Yes, the protocol demonstrates excellent tolerance for halogens. Examples in the patent show successful synthesis of 3-chloro and 3-bromo substituted naphthol derivatives with yields up to 67%.
Q: What is the optimal solvent for maximizing reaction conversion?
A: Fluorobenzene (PhF) is identified as the preferred solvent, providing higher reaction conversion rates and fewer side reactions compared to alternatives like 1,2-dichloroethane.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyaryl Substituted Naphthol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in the production of high-performance organic materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of rhodium-catalyzed reactions, including rigorous QC labs that enforce stringent purity specifications to meet the demanding standards of the electronic and pharmaceutical industries. We are committed to delivering consistent quality and reliability, making us the ideal partner for your complex synthesis needs.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your production costs and accelerate your time to market. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your project volume. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for advanced organic intermediates.
