Advanced Synthetic Route for Lycibarbarine A and B: Enabling Commercial Scale Production
The pharmaceutical industry is constantly seeking robust methods to access bioactive natural products that were previously limited by scarce natural sources. Patent CN114957284B introduces a groundbreaking efficient synthetic method for Lycibarbarine A and B, two promising alkaloids isolated from Lycium barbarum (wolfberry) known for their neuroprotective and antidepressant properties. Historically, obtaining these compounds relied on tedious extraction processes with extremely low yields, creating a bottleneck for drug development. This new technology disrupts that paradigm by establishing a concise, high-efficiency total synthesis route that transforms readily available starting materials into complex spiroketal alkaloids. By leveraging modern organic synthesis strategies, including selective protection, oxidation, and biomimetic reduction, this method provides a reliable supply chain solution for researchers and manufacturers aiming to explore the therapeutic potential of these molecules without the constraints of agricultural variability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of Lycibarbarines has been plagued by significant logistical and economic hurdles inherent to natural product isolation. The content of these bioactive alkaloids in wolfberry fruit is exceptionally low, necessitating the processing of massive quantities of raw plant material to obtain mere milligrams of the target compound. This extraction process is not only time-consuming and labor-intensive but also results in inconsistent purity profiles due to the complex matrix of plant metabolites. Furthermore, the seasonal dependence of crop harvesting introduces severe volatility into the supply chain, making it impossible to guarantee the continuous availability required for preclinical and clinical trials. The high cost associated with purification chromatography to separate these trace compounds from structurally similar impurities further exacerbates the financial burden, rendering large-scale pharmacological studies economically unfeasible for many research institutions.
The Novel Approach
In stark contrast, the synthetic methodology disclosed in the patent offers a transformative solution by constructing the Lycibarbarine skeleton from simple, commercially abundant precursors. The route strategically converges two distinct fragments: a sugar-derived spiroketal core and a functionalized tetrahydroquinoline moiety. By utilizing D-2-deoxyribose as a chiral pool starting material, the synthesis inherently establishes the necessary stereochemistry without requiring complex asymmetric catalysis in later stages. The process is designed for operational simplicity, featuring a short step count and the potential for one-pot multi-step reactions that drastically minimize intermediate isolation. This approach not only bypasses the limitations of plant extraction but also allows for the precise tuning of reaction conditions to optimize yield and purity, thereby laying a solid material foundation for the extensive biological evaluation of these promising antidepressant candidates.
Mechanistic Insights into the Convergent Synthesis Strategy
The core of this synthetic breakthrough lies in the meticulous construction of the spiroketal linkage and the selective reduction of the quinoline ring. The process begins with the acid-catalyzed protection of D-2-deoxyribose using acetone and 2-methoxypropene to form the acetonide intermediate, which locks the sugar conformation and protects sensitive hydroxyl groups. Subsequent oxidation with iodine and potassium carbonate elegantly converts the protected sugar into a lactone, setting the stage for nucleophilic attack. A critical mechanistic step involves the generation of an iodomethyl nucleophile at cryogenic temperatures (-78°C) using diiodomethane and methyllithium. This highly reactive species attacks the lactone carbonyl, creating the extended carbon framework necessary for the final spirocyclic structure. The precision required at this low temperature ensures that side reactions are minimized, preserving the integrity of the chiral centers derived from the starting sugar.
Parallel to the sugar fragment preparation, the aromatic component is synthesized through a classic yet optimized formylation of 8-hydroxyquinoline followed by a sophisticated reduction step. The use of Hantzsch ester (HE) catalyzed by boric acid represents a green chemistry approach to reducing the quinoline ring to a tetrahydroquinoline derivative. This biomimetic reduction is highly selective, avoiding the over-reduction or decomposition often seen with harsher metal hydride reagents. The final convergence involves coupling the iodomethyl sugar intermediate with the phenolic tetrahydroquinoline under basic conditions using cesium carbonate. This etherification links the two halves of the molecule, followed by a final acid-mediated deprotection and intramolecular cyclization that spontaneously forms the characteristic spiroketal ring system of Lycibarbarine A and B.
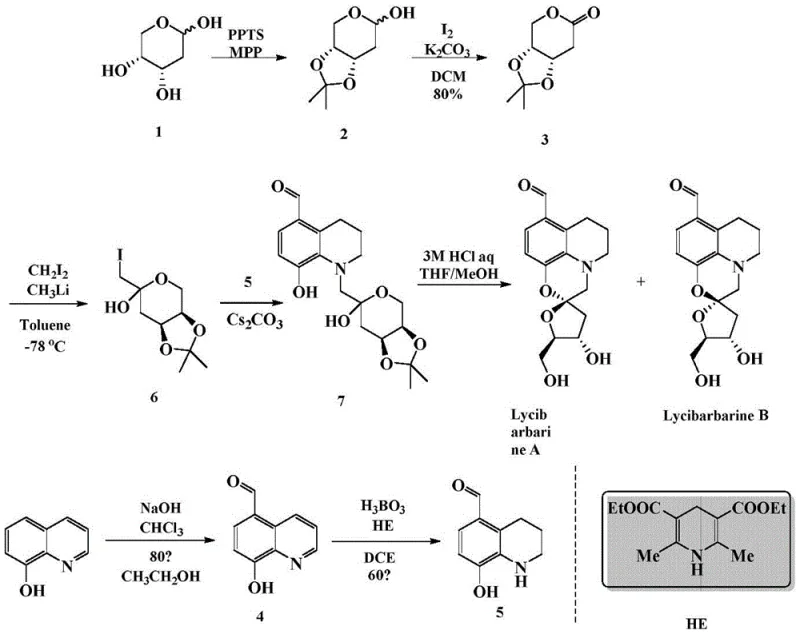
How to Synthesize Lycibarbarine Efficiently
The synthesis of these complex alkaloids requires strict adherence to anhydrous conditions and precise temperature control during the nucleophilic addition steps to ensure high stereochemical fidelity. The protocol outlined in the patent demonstrates that the intermediate coupling can be performed in a telescoped manner, where the crude iodomethyl species is reacted directly with the quinoline fragment, thereby saving significant time and solvent usage. For detailed operational parameters, reagent stoichiometry, and workup procedures that guarantee reproducible results, please refer to the standardized synthesis guide below.
- Protect D-2-deoxyribose with acetonide groups and oxidize to form the key lactone intermediate.
- Perform formylation on 8-hydroxyquinoline followed by selective biomimetic reduction using Hantzsch ester.
- Couple the sugar-derived nucleophile with the quinoline fragment and finalize with acid-mediated cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial strategic advantages over traditional extraction methods. The reliance on commodity chemicals such as D-2-deoxyribose, 8-hydroxyquinoline, and common solvents like toluene and ethyl acetate ensures that raw material sourcing is stable and cost-effective. Unlike botanical extracts which are subject to harvest failures and price fluctuations, these chemical feedstocks are produced on a multi-ton scale globally, providing a secure foundation for long-term manufacturing planning. The elimination of complex purification steps associated with natural product isolation further streamlines the production workflow, reducing the overall consumption of silica gel and chromatography solvents which are significant cost drivers in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to minimize unit operations, which directly translates to lower operational expenditures. By enabling one-pot transformations and avoiding the use of expensive transition metal catalysts or exotic reagents, the process significantly lowers the cost of goods sold (COGS). The use of inexpensive oxidants like iodine and bases like potassium carbonate instead of precious metal catalysts removes the need for costly metal scavenging steps, simplifying the downstream processing and waste treatment requirements. This economic efficiency makes the commercial production of Lycibarbarine intermediates viable for broad-scale pharmaceutical applications.
- Enhanced Supply Chain Reliability: Adopting this chemical synthesis route decouples the supply of Lycibarbarine from the agricultural sector, mitigating risks associated with climate change, pests, or geopolitical issues affecting crop yields. Manufacturers can maintain consistent inventory levels and meet Just-In-Time delivery schedules because the production timeline is determined by reactor capacity rather than growing seasons. This reliability is crucial for pharmaceutical partners who require guaranteed batch-to-batch consistency and uninterrupted supply to support regulatory filings and clinical trial timelines without the risk of raw material shortages.
- Scalability and Environmental Compliance: The process exhibits excellent scalability characteristics, having been designed with industrial feasibility in mind. The reaction conditions utilize standard equipment and do not require extreme pressures or temperatures that would necessitate specialized infrastructure. Furthermore, the reduction in solvent usage and the avoidance of heavy metal waste align with increasingly stringent environmental regulations and green chemistry principles. This facilitates easier permitting and compliance with environmental, health, and safety (EHS) standards, allowing for smoother technology transfer from laboratory scale to multi-ton commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Lycibarbarine intermediates. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of this synthetic approach for potential partners and stakeholders evaluating this technology for their pipeline.
Q: What are the primary advantages of this synthetic route over plant extraction?
A: This chemical synthesis eliminates the reliance on low-yield plant extraction from Lycium barbarum, offering consistent supply, higher purity, and significantly reduced production costs suitable for industrial scaling.
Q: How does the process ensure high stereochemical purity?
A: The route utilizes chiral pool starting materials like D-2-deoxyribose and employs highly selective reduction conditions with Hantzsch esters, ensuring the correct stereochemistry required for biological activity.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the protocol features short reaction steps, utilizes economical reagents like iodine and carbonates, and supports one-pot multi-step operations, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lycibarbarine Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of novel natural product derivatives in the field of neuroprotection and mental health therapeutics. As a leading CDMO partner, we possess the technical expertise to adapt and optimize complex synthetic pathways like the one described in CN114957284B for industrial application. Our facilities are equipped to handle diverse chemical transformations, ranging from cryogenic organolithium reactions to sensitive biomimetic reductions, ensuring that the transition from gram-scale research to commercial manufacturing is seamless. We pride ourselves on our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, coupled with our stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards.
We invite pharmaceutical innovators and research institutions to collaborate with us to accelerate the development of Lycibarbarine-based therapies. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall R&D expenditure. Please contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in turning promising chemical discoveries into life-saving medicines.
