Advanced Synthesis of 5-Substituted Hydantoins for Commercial ACE Inhibitor Production
Introduction to Novel 5-Substituted Hydantoins Technology
The pharmaceutical industry constantly seeks robust and scalable pathways to access high-value chiral intermediates, particularly for cardiovascular medications. Patent CN101679295A introduces a groundbreaking class of 5-substituted hydantoins that serve as versatile building blocks for the synthesis of enantiomerically enriched alpha-amino acids. These novel compounds address a long-standing gap in chemical literature where specific O-acyl, S-acyl, or cyclic alkenyl substituted hydantoins were either unknown or difficult to synthesize with high purity. By leveraging readily available starting materials such as L-serine and L-cysteine, this technology enables the efficient production of precursors for blockbuster drugs like Ramipril and Perindopril. The strategic design of these molecules allows for precise enzymatic resolution, ensuring the high optical purity required for modern active pharmaceutical ingredients (APIs).
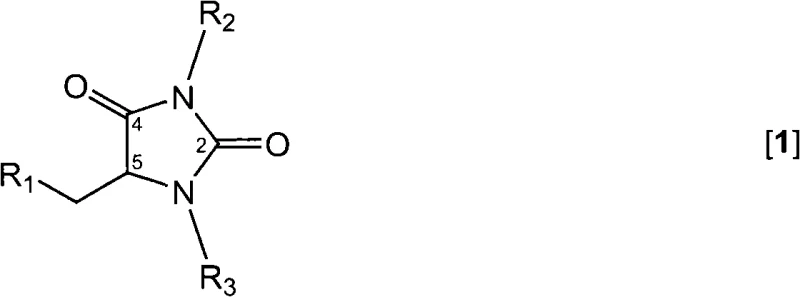
This technological advancement represents a significant leap forward for reliable pharmaceutical intermediates supplier networks, offering a route that bypasses the limitations of traditional racemic synthesis. The ability to introduce specific leaving groups at the 5-position opens up new avenues for downstream functionalization, making these hydantoins indispensable tools in the medicinal chemist's arsenal. Furthermore, the process is designed with industrial scalability in mind, utilizing standard reagents and manageable reaction conditions that facilitate seamless technology transfer from laboratory to pilot plant.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-substituted hydantoins has been constrained by the availability of suitable starting materials and the harsh conditions often required to install functional groups at the 5-position. Conventional methods frequently struggle to introduce specific O-based or S-based leaving groups without compromising the integrity of the hydantoin ring or causing unwanted side reactions. Many existing protocols rely on expensive chiral catalysts or complex resolution steps that drastically increase the cost of goods sold (COGS) and extend lead times. Additionally, the lack of accessible cyclic alkenyl or cyclic keto-alkyl substituted hydantoins has limited the structural diversity available for drug discovery programs targeting enzyme-mediated pathways. These bottlenecks often result in lower overall yields and generate significant chemical waste, posing challenges for both economic viability and environmental compliance in large-scale manufacturing.
The Novel Approach
The methodology disclosed in CN101679295A overcomes these hurdles by utilizing a mild and highly selective acylation or sulfonylation strategy on amino acid hydantoins. Instead of forcing difficult transformations, this approach starts with stable precursors like serine hydantoin or cysteine hydantoin and modifies the side chain under controlled conditions. The use of common acylating agents such as acetyl chloride or sulfonylating agents like tosyl chloride allows for the precise installation of leaving groups with excellent regioselectivity. This novel pathway not only expands the chemical space of available hydantoins but also sets the stage for efficient enzymatic conversion into optically pure amino acids. By avoiding extreme temperatures and hazardous reagents, the process aligns perfectly with the goals of cost reduction in pharmaceutical intermediates manufacturing, offering a cleaner and more direct route to high-value targets.
Mechanistic Insights into Acylation and Enamine Addition
The core of this synthetic strategy involves the nucleophilic attack of the hydroxyl or thiol group on the side chain of the amino acid hydantoin by an electrophilic acylating or sulfonylating agent. In the presence of an organic base such as pyridine or triethylamine, the reaction proceeds smoothly at temperatures ranging from -30°C to 50°C, ensuring that the sensitive hydantoin ring remains intact. The choice of solvent, typically acetonitrile or dichloromethane, plays a crucial role in solubilizing the intermediates and managing the exotherm during reagent addition. Following the formation of the activated 5-substituted hydantoin, the process employs a sophisticated enamine addition or ketoester coupling step. This transformation utilizes cyclic enamines or beta-keto esters to construct the complex cyclic side chains found in ACE inhibitor precursors. The subsequent hydrolysis step reveals the ketone functionality, setting up the molecule for the critical biotransformation phase.
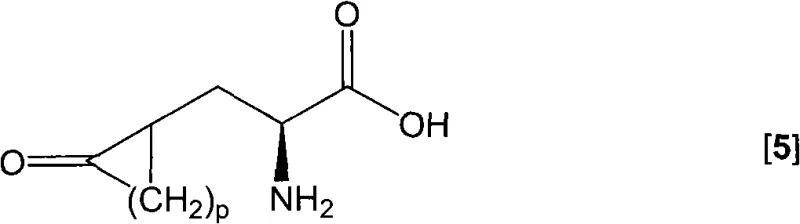
Impurity control is meticulously managed through the selection of stoichiometric ratios and reaction temperatures. The patent specifies that the molar amounts of acylating agent and base should be closely matched, with a preference for slight excesses to drive the reaction to completion without generating excessive byproducts. The use of enzymatic resolution in the downstream processing further enhances purity, as enzymes like hydantoinase and carbamoylase exhibit high stereoselectivity, effectively filtering out unwanted enantiomers. This dual approach of chemical precision followed by biological specificity ensures that the final alpha-amino acid products meet the stringent purity specifications demanded by regulatory agencies for human therapeutics.
How to Synthesize 5-Substituted Hydantoins Efficiently
Executing this synthesis requires careful attention to reaction parameters, particularly temperature control and reagent addition rates, to maximize yield and minimize impurity formation. The process begins with the suspension of the amino acid hydantoin in a dry inert solvent, followed by the slow addition of the activating agent to manage heat generation. Detailed standard operating procedures for each step, including workup and purification techniques, are essential for reproducibility. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and isolation methods described in the patent, please refer to the standardized synthesis guide below.
- Prepare the starting amino acid hydantoin (e.g., from L-serine or L-cysteine) and suspend it in a dry inert solvent like acetonitrile.
- React the suspension with an acylating or sulfonylating agent (e.g., acetyl chloride or tosyl chloride) in the presence of an organic base at controlled low temperatures.
- Perform enamine addition or ketoester coupling followed by hydrolysis and enzymatic resolution to yield the optically pure alpha-amino acid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers substantial strategic benefits beyond mere technical feasibility. The reliance on commodity chemicals like L-serine and L-cysteine as starting materials ensures a stable and predictable supply chain, reducing the risk of raw material shortages that often plague specialty chemical manufacturing. Furthermore, the elimination of expensive transition metal catalysts in favor of organic bases and enzymatic steps significantly lowers the input costs associated with catalyst recovery and heavy metal removal. This shift not only reduces the environmental footprint of the production process but also simplifies the regulatory filing process by minimizing residual metal concerns in the final API.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive and readily available acylating agents and solvents, avoiding the need for proprietary or high-cost chiral auxiliaries. The mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity of the enzymatic resolution step minimizes the loss of valuable material to unwanted isomers. By streamlining the synthesis into fewer, more efficient steps, manufacturers can achieve a lower cost per kilogram, enhancing the overall profitability of the supply chain for ACE inhibitor production.
- Enhanced Supply Chain Reliability: Sourcing strategies are strengthened by the use of bulk chemicals that are widely produced globally, mitigating the risks associated with single-source suppliers for exotic reagents. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor variations in ambient conditions compared to cryogenic or high-pressure alternatives. This reliability translates to shorter lead times for high-purity pharmaceutical intermediates, enabling pharmaceutical companies to maintain leaner inventory levels while ensuring continuous production of life-saving medications.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been designed with commercial scale-up of complex pharmaceutical intermediates in mind. The use of aqueous workups and standard organic solvents facilitates easy waste treatment and solvent recovery, aligning with modern green chemistry principles. The avoidance of toxic heavy metals and the use of biocatalysis in the final steps demonstrate a commitment to sustainable manufacturing practices, which is increasingly becoming a key criterion for vendor selection in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydantoin synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical aspects of adopting this route. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this chemistry into their existing pipelines.
Q: What are the primary applications of these novel 5-substituted hydantoins?
A: These compounds serve as critical intermediates for the synthesis of enantiomerically enriched alpha-amino acids, which are precursors for major ACE inhibitors like Ramipril and Perindopril.
Q: How does this process improve upon conventional hydantoin synthesis?
A: The patented method allows for the introduction of specific O-acyl, S-acyl, or cyclic alkenyl leaving groups that were previously difficult to access, facilitating downstream enzymatic resolution.
Q: Is the process scalable for industrial manufacturing?
A: Yes, the reaction conditions utilize common solvents like acetonitrile and moderate temperatures ranging from -30°C to 50°C, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Substituted Hydantoins Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation cardiovascular therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-substituted hydantoins meets the highest international standards. Our commitment to quality and consistency makes us the preferred partner for pharmaceutical companies seeking to secure their supply chains for essential ACE inhibitor precursors.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can optimize your production costs and timelines. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of switching to this novel methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project requirements, ensuring a successful and profitable partnership.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
