Advanced Two-Step Synthesis of Biphenyl Alcohol Acid for Commercial Scale-Up
The pharmaceutical landscape for hepatoprotective agents continues to evolve, driven by the demand for higher purity intermediates and more sustainable manufacturing processes. Patent CN113754627A introduces a transformative approach to synthesizing biphenyl alcohol acid, a critical precursor for the liver-protecting drug Bicyclol. Traditionally, the production of this chiral biaryl scaffold has been plagued by multi-step sequences involving hazardous reagents and energy-intensive conditions. This new methodology disrupts the status quo by condensing the synthesis into a highly efficient two-step sequence that bypasses the formation of unstable anhydride intermediates. By leveraging catalytic partial hydrolysis followed by selective reduction, the process not only enhances overall yield but also aligns with modern green chemistry principles, offering a robust solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
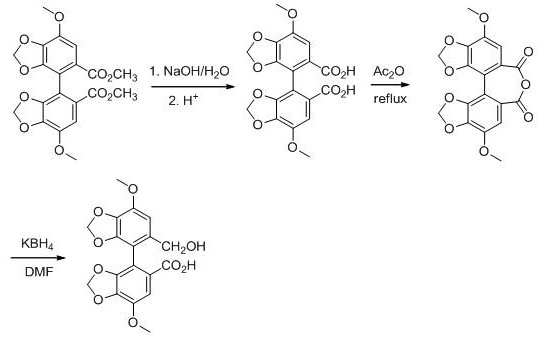
Historically, the industrial preparation of biphenyl alcohol acid from biphenyl dicarboxylate has relied on a cumbersome three-step trajectory that poses significant operational and regulatory challenges. The conventional route necessitates the complete hydrolysis of the diester to the diacid, followed by a dehydration step using acetic anhydride to form the cyclic anhydride, which is subsequently subjected to alcoholysis or selective reduction. This reliance on acetic anhydride is particularly problematic as it is a strictly regulated chemical due to its potential misuse, complicating procurement and storage logistics for manufacturing facilities. Furthermore, the dehydration step typically requires high-temperature reflux conditions, leading to excessive energy consumption and the generation of difficult-to-treat aromatic solvent waste streams. The complexity of isolating the anhydride intermediate often results in material loss and increased production costs, creating a bottleneck for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
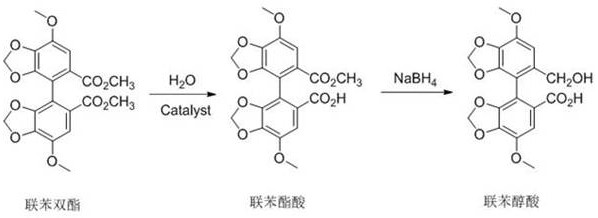
In stark contrast to the legacy methods, the innovative strategy disclosed in the patent streamlines the synthesis by directly converting the biphenyl diester into the target alcohol-acid functionality through a telescoped two-step protocol. The process initiates with a controlled partial hydrolysis, where a catalytic amount of acid or base selectively cleaves one ester group to yield the biphenyl ester acid, effectively skipping the diacid and anhydride stages entirely. This intermediate is then immediately subjected to reduction using a borohydride salt, which selectively reduces the remaining ester moiety to a hydroxymethyl group while preserving the carboxylic acid functionality. This direct transformation eliminates the need for hazardous dehydrating agents and high-temperature conditions, drastically simplifying the reaction setup and workup procedures. The result is a cleaner reaction profile with fewer byproducts, enabling cost reduction in API manufacturing through reduced solvent usage and simplified purification protocols.
Mechanistic Insights into Catalytic Partial Hydrolysis and Selective Reduction
The core chemical innovation lies in the precise control of the hydrolysis equilibrium to prevent over-reaction to the diacid, which would complicate the subsequent selective reduction. By employing a solid acid catalyst such as magnetic sulfonic acid (Fe3O4@SiO2-SO3H), the reaction environment promotes the nucleophilic attack of water on one ester carbonyl while the steric and electronic properties of the biaryl system help stabilize the mono-ester intermediate. The use of a mixed solvent system, typically methanol and water in a specific ratio like 9:1, fine-tunes the solubility and reactivity, ensuring that the hydrolysis stops at the desired mono-acid stage. Real-time monitoring or strict time-temperature controls are essential here, as allowing the reaction to proceed too long would generate the diacid, which is unreactive towards the subsequent selective reduction conditions intended for the ester group. This mechanistic precision ensures high atom economy and minimizes the formation of hard-to-separate impurities.
Following the hydrolysis, the selective reduction mechanism utilizes the differential reactivity of borohydride species towards esters versus carboxylic acids under controlled conditions. In the presence of solvents like tetrahydrofuran, the borohydride anion attacks the carbonyl carbon of the methyl ester group, forming a tetrahedral intermediate that collapses to release methoxide and form the aldehyde, which is rapidly reduced further to the primary alcohol. Crucially, the carboxylic acid group formed in the first step remains largely intact or forms a stable salt that does not undergo reduction under these mild temperatures (-10 to 20°C). This chemoselectivity is vital for producing high-purity biphenyl alcohol acid without the need for protecting group strategies, thereby enhancing the feasibility of the process for large-scale production. The use of magnetic catalysts further aids in impurity control by allowing for the physical removal of the acid source before the reduction step, preventing acid-catalyzed side reactions.
How to Synthesize Biphenyl Alcohol Acid Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize the yield of the mono-hydrolyzed intermediate. The process begins by dispersing the biphenyl diester in a methanol-water mixture and adding the magnetic catalyst, followed by stirring at ambient temperature until conversion is optimized. Once the mono-acid is isolated or prepared in situ, it is dissolved in an anhydrous solvent like THF and treated with a slight excess of sodium or potassium borohydride at low temperatures to ensure selectivity. For detailed operational parameters, stoichiometric ratios, and specific workup procedures, please refer to the standardized synthesis guide below.
- Perform partial hydrolysis of biphenyl diester using a catalytic amount of acid or base (e.g., magnetic sulfonic acid) in a methanol/water solvent system at 0-40°C to obtain biphenyl ester acid.
- React the resulting biphenyl ester acid with a borohydride reducing agent (such as sodium borohydride or potassium borohydride) in an organic solvent like tetrahydrofuran at -10 to 20°C.
- Quench the reaction with water, adjust pH to below 2, and isolate the final biphenyl alcohol acid product through filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from a three-step to a two-step synthesis represents a substantial opportunity to de-risk the supply of critical liver drug intermediates. By eliminating the requirement for acetic anhydride, companies can bypass the stringent regulatory hurdles and security protocols associated with purchasing and storing this controlled substance, thereby smoothing the procurement workflow. The removal of high-temperature dehydration steps also translates to lower energy utility costs and reduced wear on reactor equipment, contributing to a more sustainable and cost-effective manufacturing footprint. Furthermore, the simplified workflow reduces the total cycle time per batch, allowing for greater flexibility in production scheduling and faster response to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of the dehydration step and the associated reagents like acetic anhydride removes a significant cost center from the bill of materials. Additionally, the ability to recover and reuse the magnetic sulfonic acid catalyst through simple magnetic separation reduces the consumption of fresh catalyst, leading to cumulative savings over large production campaigns. The simplified purification process, which avoids complex distillations or extractions associated with anhydride handling, further lowers the operational expenditure related to solvent recovery and waste disposal.
- Enhanced Supply Chain Reliability: Relying on commodity reagents such as borohydrides and common alcohols instead of specialized dehydrating agents enhances the resilience of the supply chain against raw material shortages. The robust nature of the two-step process ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed quality control tests or off-spec intermediates. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who depend on just-in-time delivery models for their own production schedules.
- Scalability and Environmental Compliance: The process operates under mild conditions with aqueous workups, making it inherently safer and easier to scale from pilot plant to multi-ton commercial production. The avoidance of aromatic solvents and toxic anhydrides aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste generation. This green chemistry profile not only reduces the environmental impact but also minimizes the regulatory burden on the manufacturing site, facilitating smoother audits and inspections by global health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical principles underlying the technology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines.
Q: Why is the elimination of acetic anhydride significant in this synthesis?
A: Acetic anhydride is a regulated precursor often associated with safety hazards and complex waste treatment. Eliminating it removes the need for high-temperature dehydration steps, significantly improving process safety and environmental compliance.
Q: How does the new method achieve selectivity without forming the diacid?
A: The process utilizes controlled partial hydrolysis with real-time monitoring or specific catalytic conditions (e.g., magnetic sulfonic acid) to stop the reaction at the mono-ester/mono-acid stage, preventing full hydrolysis to the diacid.
Q: What are the advantages of using magnetic sulfonic acid as a catalyst?
A: Magnetic sulfonic acid allows for rapid and efficient separation from the reaction mixture using an external magnet, facilitating catalyst recycling and simplifying downstream purification compared to traditional homogeneous acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Alcohol Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity biphenyl alcohol acid that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in catalytic hydrogenation and selective reduction allows us to optimize this specific two-step route for maximum yield and minimal environmental impact.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall production costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
