Advanced Nickel-Catalyzed Synthesis of Benzyl Sulfides for Pharmaceutical Intermediates
Advanced Nickel-Catalyzed Synthesis of Benzyl Sulfides for Pharmaceutical Intermediates
The development of efficient and cost-effective methodologies for constructing carbon-sulfur bonds remains a pivotal challenge in modern organic synthesis, particularly for the production of high-value pharmaceutical intermediates and agrochemical active ingredients. Patent CN115477600A discloses a groundbreaking preparation method for benzyl sulfide compounds that leverages a nickel-catalyzed desulfitative cross-coupling strategy. This innovative approach utilizes benzylsulfonyl chloride and arylsulfonyl chloride as the primary building blocks, effectively transforming two readily available sulfonyl chloride species into a valuable thioether scaffold without the need for pre-functionalized thiols. The significance of this technology lies in its ability to bypass traditional limitations associated with sulfur sources, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. By employing an earth-abundant nickel catalyst system, this process not only enhances reaction efficiency but also aligns with the growing industry demand for sustainable and economically viable synthetic routes.
In this novel synthetic architecture, the structural diversity is achieved through the modulation of the aryl groups attached to the sulfonyl chloride moieties. The benzylsulfonyl chloride component, represented generally as structure (II), serves as the electrophilic partner, while the arylsulfonyl chloride, denoted as structure (III), acts as the sulfur source. 
 The convergence of these two distinct sulfonyl chloride derivatives results in the formation of the target benzyl sulfide compound, illustrated as structure (I), which features a versatile benzyl-thio-aryl linkage.
The convergence of these two distinct sulfonyl chloride derivatives results in the formation of the target benzyl sulfide compound, illustrated as structure (I), which features a versatile benzyl-thio-aryl linkage.  This specific connectivity is ubiquitous in bioactive molecules, making the ability to synthesize high-purity benzyl sulfides with broad substrate tolerance a critical capability for any reliable pharmaceutical intermediate supplier aiming to support drug discovery pipelines.
This specific connectivity is ubiquitous in bioactive molecules, making the ability to synthesize high-purity benzyl sulfides with broad substrate tolerance a critical capability for any reliable pharmaceutical intermediate supplier aiming to support drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C–S bonds has predominantly relied on the transition metal-catalyzed cross-coupling of organic halides with sulfur-containing nucleophiles, such as thiols or thiolates. However, these conventional methodologies suffer from significant drawbacks that hinder their application in large-scale fine chemical manufacturing. Thiols are notoriously malodorous, toxic, and prone to oxidation, creating severe handling and environmental safety challenges in industrial settings. Furthermore, sulfur nucleophiles have a strong tendency to coordinate with and poison transition metal catalysts, often necessitating the use of expensive palladium or copper systems in high loadings to maintain catalytic turnover. While alternative sulfur sources like disulfides or sulfonyl hydrazides have been explored, they often introduce additional synthetic steps or generate stoichiometric amounts of difficult-to-remove byproducts. The reliance on precious metals further exacerbates the cost burden, making cost reduction in pharmaceutical intermediate manufacturing difficult to achieve when using these legacy technologies.
The Novel Approach
The methodology described in CN115477600A represents a paradigm shift by utilizing sulfonyl chlorides as both the electrophile and the sulfur source in a single transformative event. This desulfitative cross-coupling reaction eliminates the need for unstable thiols entirely, thereby removing the associated odor and catalyst deactivation issues. The core of this innovation is the use of a nickel catalyst system, specifically bis(tricyclohexylphosphine)nickel dichloride, which facilitates the extrusion of sulfur dioxide (SO2) from the sulfonyl chloride moiety to generate the reactive sulfur species in situ. 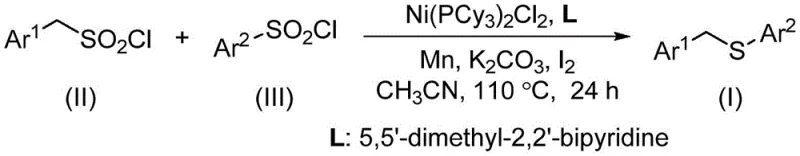 As depicted in the reaction scheme, the combination of benzylsulfonyl chloride and arylsulfonyl chloride under optimized conditions yields the desired benzyl sulfide with high efficiency. This approach not only simplifies the operational procedure but also utilizes starting materials that are cheap, stable, and widely available in the chemical market. By replacing precious metals with nickel, the process inherently supports cost reduction in fine chemical manufacturing, offering a scalable solution that addresses both economic and environmental concerns simultaneously.
As depicted in the reaction scheme, the combination of benzylsulfonyl chloride and arylsulfonyl chloride under optimized conditions yields the desired benzyl sulfide with high efficiency. This approach not only simplifies the operational procedure but also utilizes starting materials that are cheap, stable, and widely available in the chemical market. By replacing precious metals with nickel, the process inherently supports cost reduction in fine chemical manufacturing, offering a scalable solution that addresses both economic and environmental concerns simultaneously.
Mechanistic Insights into Nickel-Catalyzed Desulfitative Coupling
The mechanistic underpinning of this transformation involves a sophisticated interplay between the nickel catalyst, the ligand system, and the reducing agents. The reaction initiates with the activation of the nickel(II) precatalyst, likely reduced to a lower oxidation state nickel(0) or nickel(I) species by the manganese powder present in the reaction mixture. This active nickel species undergoes oxidative addition into the S–Cl or C–S bond of the sulfonyl chloride, followed by the extrusion of SO2 gas, which acts as the driving force for the reaction. The resulting nickel-sulfur intermediate then engages in a transmetallation or radical recombination process with the benzyl fragment derived from the second sulfonyl chloride molecule. The presence of iodine and the bipyridine ligand (5,5'-dimethyl-2,2'-bipyridine) plays a crucial role in stabilizing the catalytic cycle and facilitating the reductive elimination step that forms the final C–S bond. This intricate mechanism ensures that the reaction proceeds smoothly even with sterically hindered or electronically diverse substrates.
From an impurity control perspective, this mechanism offers distinct advantages over thiol-based routes. Since no free thiol is introduced, the formation of disulfide byproducts, which are common impurities in traditional syntheses, is effectively suppressed. The use of manganese as a reductant and potassium carbonate as a base ensures a clean reaction profile, minimizing the formation of homocoupling products or dehalogenated side products. The broad functional group tolerance observed in the patent data, accommodating groups such as nitro, trifluoromethyl, and various halogens, suggests that the catalytic cycle is robust against competitive coordination or side reactions. This level of purity is essential for clients seeking high-purity OLED material precursors or drug candidates where strict impurity profiles are mandated by regulatory bodies.
How to Synthesize Benzyl Sulfide Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and minimize waste. The patented process outlines a straightforward protocol where the nickel catalyst, ligand, base, and additives are combined with the sulfonyl chloride substrates in acetonitrile. The reaction is typically conducted at elevated temperatures, around 110°C, for a duration of approximately 24 hours to ensure complete conversion. Detailed standardized synthetic steps for replicating this high-efficiency route are provided in the technical guide below, ensuring reproducibility for process chemists.
- Charge a reaction vessel with bis(tricyclohexylphosphine)nickel dichloride, 5,5'-dimethyl-2,2'-bipyridine, potassium carbonate, manganese powder, and iodine under inert atmosphere.
- Add benzylsulfonyl chloride and arylsulfonyl chloride substrates along with acetonitrile solvent to the mixture.
- Heat the reaction mixture to 110°C for 24 hours, then filter, purify via silica gel chromatography to isolate the target benzyl sulfide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed technology presents a compelling value proposition centered on cost stability and operational simplicity. The primary driver for cost reduction in pharmaceutical intermediate manufacturing is the substitution of expensive precious metal catalysts with abundant nickel, which drastically lowers the raw material cost per kilogram of product. Additionally, the use of sulfonyl chlorides eliminates the need for specialized containment systems required for handling volatile and toxic thiols, thereby reducing capital expenditure on safety infrastructure and waste treatment facilities. The simplicity of the workup procedure, involving basic filtration and chromatography, further streamlines the production timeline, allowing for faster turnaround times and improved asset utilization.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily driven by the replacement of palladium or ruthenium catalysts with low-cost nickel complexes. Since the starting materials, benzylsulfonyl chloride and arylsulfonyl chloride, are commodity chemicals available from multiple global suppliers, the supply risk is minimized, and pricing remains stable. The elimination of thiol reagents also removes the hidden costs associated with odor control scrubbers and hazardous waste disposal, leading to significant overall operational savings without compromising on yield or quality.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of robust, shelf-stable reagents that do not require cold chain logistics or inert atmosphere storage prior to use. Unlike thiols which can degrade or oxidize upon exposure to air, sulfonyl chlorides maintain their integrity over long periods, ensuring consistent batch-to-bquality. This stability simplifies inventory management and reduces the risk of production delays caused by reagent spoilage, making it easier to maintain continuous supply for downstream customers requiring reliable pharmaceutical intermediate supplier partnerships.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in sealed tube reactions that can be readily adapted to standard stainless steel reactors used in commercial plants. The generation of sulfur dioxide as a byproduct is manageable with standard scrubbing systems, and the absence of heavy metal contamination simplifies the purification process, reducing the environmental footprint. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the commercial scale-up of complex pharmaceutical intermediates more feasible and environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic benefits for potential partners evaluating this technology for their supply chains.
Q: What are the advantages of using sulfonyl chlorides over thiols for C-S bond formation?
A: Sulfonyl chlorides are odorless, stable, and commercially available, avoiding the catalyst poisoning and unpleasant handling issues associated with volatile thiols.
Q: Is this nickel-catalyzed method suitable for large-scale manufacturing?
A: Yes, the use of inexpensive nickel catalysts instead of precious metals like palladium or ruthenium significantly reduces raw material costs, making it highly viable for scale-up.
Q: What is the functional group tolerance of this desulfitative coupling reaction?
A: The method exhibits excellent tolerance for various substituents including methyl, nitro, methoxy, halogens (F, Cl, Br), and trifluoromethyl groups on both aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of benzyl sulfide meets the exacting standards required for API synthesis and advanced material applications.
We invite you to collaborate with us to leverage this cutting-edge nickel-catalyzed technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce your overall manufacturing costs.
