Advanced Metal-Free Cyanation of 8-Acylaminoquinolines for High-Purity Pharma Intermediates
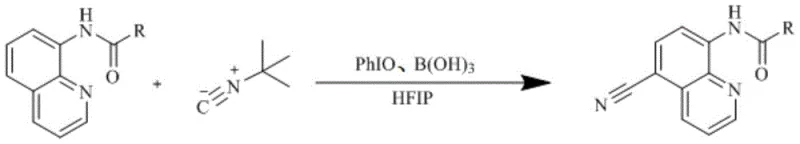
The landscape of quinoline functionalization is undergoing a significant transformation driven by the urgent demand for greener, metal-free synthetic methodologies in the fine chemical sector. Patent CN113773250A introduces a groundbreaking preparation method for 5-cyano-8-acylaminoquinoline compounds, addressing critical limitations associated with traditional transition metal-catalyzed cyanation. This technology leverages a hypervalent iodine-mediated radical process to install a cyano group at the C-5 position of the quinoline scaffold with remarkable regioselectivity. For R&D directors and procurement specialists, this represents a pivotal shift away from expensive palladium and nickel systems towards a more sustainable, cost-effective, and operationally simple protocol. The ability to synthesize these valuable intermediates without heavy metal residues directly translates to streamlined downstream processing and enhanced supply chain reliability for high-purity pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-cyanoquinoline derivatives has been heavily reliant on cross-coupling reactions such as the Negishi coupling or nickel-catalyzed cyanation, which present substantial operational and environmental hurdles. These conventional pathways typically necessitate the use of stoichiometric amounts of toxic transition metal catalysts, such as palladium or nickel complexes, along with hazardous reagents like zinc dust or potassium phosphate under rigorous inert atmospheres. The requirement for high temperatures, often exceeding 150°C in toluene, not only consumes significant energy but also poses safety risks regarding thermal runaway and solvent volatility. Furthermore, the presence of residual heavy metals in the final product is a major compliance issue for pharmaceutical applications, mandating complex and costly purification steps involving metal scavengers or repeated recrystallization to meet stringent regulatory limits. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113773250A utilizes a metal-free system comprising iodosobenzene (PhIO), boric acid, and tert-butyl isonitrile in hexafluoroisopropanol (HFIP). This innovative route operates under significantly milder conditions, specifically between 60-70°C, thereby reducing energy consumption and thermal stress on the substrate. The absence of transition metals eliminates the risk of heavy metal contamination, fundamentally altering the purification workflow and reducing the reliance on expensive scavenging resins. By employing a hypervalent iodine oxidant, the reaction achieves efficient C-H activation through a radical mechanism that is both atom-economical and environmentally benign. This approach not only simplifies the reaction setup by removing the need for strict inert gas protection but also enhances the overall process safety profile, making it an ideal candidate for reliable agrochemical intermediate and pharmaceutical intermediate supplier operations seeking to optimize their manufacturing footprint.
Mechanistic Insights into Hypervalent Iodine-Mediated Radical Cyanation
The mechanistic pathway of this transformation is rooted in a single-electron transfer (SET) process initiated by the interaction between the hypervalent iodine species and the electron-rich quinoline ring. Upon heating to 60-70°C, iodosobenzene undergoes homolytic cleavage or SET with the 8-acylaminoquinoline substrate to generate a delocalized aryl carbon radical at the C-5 position. This regioselectivity is governed by the electronic directing effect of the 8-acylamino group, which stabilizes the radical intermediate through resonance, ensuring that the cyanation occurs exclusively at the desired site rather than other positions on the quinoline ring. The presence of boric acid is crucial, as it likely coordinates with the iodine species or the solvent to modulate the oxidation potential, facilitating the smooth generation of the radical species without over-oxidation of the sensitive amide functionality.
Once the aryl radical is formed, it reacts rapidly with tert-butyl isonitrile, which serves as the cyanide source in a non-toxic, easy-to-handle form. The addition of the isonitrile to the radical center forms a new imidoyl radical intermediate, which subsequently undergoes fragmentation or further oxidation to release the tert-butyl group and establish the stable carbon-nitrogen triple bond of the cyano group. The use of HFIP as the solvent is not merely incidental; its strong hydrogen-bond donating capability stabilizes the polar transition states and radical intermediates, effectively lowering the activation energy of the rate-determining steps. This sophisticated interplay of reagents ensures high selectivity and avoids the formation of competitive by-products, such as acetoxy-substituted impurities, which are observed when nucleophilic iodine reagents like iodosobenzene diacetate are employed instead of pure PhIO.
How to Synthesize 5-Cyano-8-Acylaminoquinoline Efficiently
The practical execution of this synthesis is designed for scalability and ease of operation, requiring standard glassware and common laboratory equipment without the need for specialized high-pressure reactors. The process begins with the sequential addition of the solid and liquid reagents into a reaction vessel, followed by a controlled heating period that allows the radical cascade to proceed to completion. The simplicity of the workup procedure, involving a straightforward aqueous quench and organic extraction, underscores the industrial viability of this method for the commercial scale-up of complex polymer additives and fine chemicals. Operators can expect a robust process window where minor fluctuations in stirring speed or addition order do not critically compromise the outcome, provided the temperature is maintained within the optimal range.
- Charge the reactor with 8-acylaminoquinoline, tert-butyl isonitrile, iodosobenzene, boric acid, and hexafluoroisopropanol (HFIP).
- Stir the reaction mixture at a controlled temperature of 60-70°C for a duration of 12 to 16 hours to ensure complete conversion.
- Quench with water, extract the organic phase with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this metal-free cyanation technology offers profound benefits for cost reduction in pharmaceutical intermediates manufacturing. The elimination of precious metal catalysts removes a significant variable cost component, as palladium and nickel prices are subject to high market volatility and supply constraints. Moreover, the avoidance of heavy metals drastically simplifies the quality control (QC) burden, as extensive testing for residual metals (e.g., ICP-MS analysis) and the associated remediation steps are no longer required. This streamlining of the production workflow leads to substantial cost savings by reducing the consumption of auxiliary materials like metal scavengers and shortening the overall batch cycle time, thereby increasing the throughput capacity of existing manufacturing facilities without capital expenditure.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated purification infrastructure results in a leaner cost structure. By bypassing the need for metal scavenging resins and complex filtration units, manufacturers can achieve significant operational expenditure (OpEx) reductions. Furthermore, the use of commercially available and relatively inexpensive reagents like iodosobenzene and boric acid ensures that raw material costs remain stable and predictable, shielding the supply chain from the price shocks often associated with rare earth and precious metal markets.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized, potentially import-restricted catalysts enhances the resilience of the supply chain. Tert-butyl isonitrile and hypervalent iodine reagents are widely available from multiple global suppliers, reducing the risk of single-source dependency. The mild reaction conditions also allow for the use of standard stainless steel or glass-lined reactors, which are ubiquitous in fine chemical plants, ensuring that production can be easily transferred between sites or scaled up rapidly to meet surging demand for high-purity OLED material or API precursors without lengthy equipment qualification processes.
- Scalability and Environmental Compliance: The metal-free nature of this process aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge and waste management. By generating less hazardous waste streams, facilities can reduce their environmental compliance costs and simplify waste disposal logistics. The moderate temperature range of 60-70°C is energy-efficient compared to high-temperature alternatives, contributing to a lower carbon footprint for the manufacturing process. This sustainability profile is a key differentiator for companies aiming to meet corporate social responsibility (CSR) goals while maintaining high production standards for reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this novel cyanation protocol. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear understanding of the critical process parameters. Understanding these nuances is essential for process chemists aiming to replicate the high yields and selectivity reported in the intellectual property, ensuring a smooth technology transfer from the laboratory to the pilot plant.
Q: Why is the metal-free approach superior for quinoline cyanation?
A: Traditional methods rely on palladium or nickel catalysts which leave toxic metal residues requiring expensive removal steps. This metal-free protocol eliminates heavy metal contamination, simplifying purification and ensuring higher purity for pharmaceutical applications.
Q: What is the role of Hexafluoroisopropanol (HFIP) in this reaction?
A: HFIP acts as a specialized solvent that stabilizes radical intermediates through hydrogen bonding and enhances the electrophilicity of the hypervalent iodine oxidant, facilitating the C-H activation at the 5-position under mild conditions.
Q: How does temperature affect the yield of 5-cyano-8-acylaminoquinoline?
A: Optimal yields (~50-54%) are achieved at 60-70°C. Deviating significantly, such as lowering to 30°C or raising to 150°C, drastically reduces yield to below 16% due to insufficient radical generation or thermal decomposition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Cyano-8-Acylaminoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic routes in modernizing the production of high-value heterocyclic intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our infrastructure is uniquely positioned to adopt green chemistry innovations like the hypervalent iodine-mediated cyanation, allowing us to offer clients a sustainable and cost-competitive supply solution.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for quinoline-based building blocks. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this advanced metal-free technology can be integrated into your existing manufacturing portfolio to drive efficiency and reduce total landed costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
