Advanced Enzymatic Synthesis of Rapamycin 42-Esters for Commercial Pharmaceutical Manufacturing
Advanced Enzymatic Synthesis of Rapamycin 42-Esters for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and environmentally benign methodologies for the production of complex immunosuppressant intermediates. Patent CN1942476A introduces a groundbreaking regiospecific synthesis of rapamycin 42-ester derivatives, addressing a longstanding challenge in the functionalization of the rapamycin macrocycle. This technology leverages the power of biocatalysis, specifically utilizing microbial lipases to achieve high-yield acylation exclusively at the 42-position, bypassing the notorious selectivity issues associated with the chemically similar 31-hydroxyl group. By employing acyl donors such as vinyl esters, isopropenyl esters, or acid anhydrides in suitable organic solvents, this method delivers exceptional purity and efficiency under mild reaction conditions. For R&D directors and process chemists, this represents a paradigm shift from traditional multi-step chemical protections to a streamlined, single-step enzymatic transformation that aligns perfectly with modern green chemistry principles and commercial manufacturing demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical modification of rapamycin to produce 42-ester derivatives has been fraught with significant synthetic hurdles, primarily stemming from the presence of two secondary hydroxyl groups at the 31 and 42 positions which exhibit similar reactivity profiles. Conventional chemical acylation strategies often suffer from poor regioselectivity, resulting in complex mixtures of 31-monoesters, 42-monoesters, and diesters that necessitate costly and time-consuming purification techniques such as preparative high-performance liquid chromatography (HPLC). To circumvent this lack of selectivity, prior art methods frequently resorted to tedious protection-deprotection sequences, such as silyl protection of the 31-hydroxyl group prior to acylation, which inherently increases the number of unit operations, reduces overall atom economy, and escalates production costs. Furthermore, traditional chemical catalysts often require harsh acidic or basic conditions that can compromise the stability of the sensitive rapamycin macrocycle, leading to degradation products and further complicating the impurity profile. These inefficiencies create substantial bottlenecks in the supply chain, extending lead times and increasing the cost of goods sold for critical immunosuppressant intermediates.
The Novel Approach
In stark contrast to these legacy methods, the enzymatic approach detailed in patent CN1942476A offers a remarkably elegant solution by exploiting the inherent stereospecificity of microbial lipases to differentiate between the 31 and 42 hydroxyl groups without the need for protecting groups. This novel biocatalytic route utilizes robust enzymes, such as Candida antarctica lipase B or Pseudomonas cepacia lipase, to catalyze the acylation reaction with high fidelity, ensuring that the acyl group is installed exclusively at the desired 42-position. The process operates under exceptionally mild conditions, typically ranging from room temperature to 45°C, which preserves the structural integrity of the rapamycin scaffold and minimizes the formation of thermal degradation byproducts. By eliminating the need for protection-deprotection steps and harsh chemical reagents, this method drastically simplifies the synthetic workflow, reduces solvent consumption, and enhances the overall throughput of the manufacturing process. The result is a highly efficient, cost-effective, and scalable pathway that provides reliable access to high-purity rapamycin 42-ester derivatives essential for the production of next-generation therapeutics.
Mechanistic Insights into Lipase-Catalyzed Regiospecific Acylation
The success of this regiospecific synthesis lies in the precise molecular recognition capabilities of the lipase enzyme, which acts as a chiral template to orient the bulky rapamycin molecule within its active site. When rapamycin binds to the lipase, the steric environment of the enzyme's catalytic pocket selectively exposes the 42-hydroxyl group to the acyl-enzyme intermediate while effectively shielding the 31-hydroxyl group from nucleophilic attack. This "lock-and-key" interaction ensures that the transesterification or acylation reaction proceeds with absolute regiocontrol, a feat that is nearly impossible to achieve with small-molecule chemical catalysts. The use of vinyl esters as acyl donors further enhances the reaction kinetics through an irreversible mechanism; as the ester bond is cleased, the leaving group (vinyl alcohol) rapidly tautomerizes to acetaldehyde, preventing the reverse hydrolysis reaction and driving the equilibrium strongly towards product formation. This thermodynamic sink allows the reaction to proceed to near-completion even with stoichiometric amounts of reagents, maximizing yield and minimizing waste.

The versatility of this biocatalytic system is demonstrated by its compatibility with a wide range of acyl donors, including vinyl esters, isopropenyl esters, and acid anhydrides, allowing for the synthesis of diverse 42-ester derivatives tailored for specific pharmacological profiles. For instance, the use of vinyl chloroacetate facilitates the introduction of reactive chloromethyl functionalities, while vinyl benzoate allows for the installation of aromatic moieties, all with consistently high yields exceeding 90% in many embodiments. The choice of solvent also plays a critical role in maintaining enzyme activity and substrate solubility, with tert-butyl methyl ether (TBME) emerging as a preferred medium due to its ability to suspend the immobilized enzyme effectively while dissolving the hydrophobic rapamycin substrate. This mechanistic robustness ensures that the process remains stable and reproducible across different batches, providing a solid foundation for commercial scale-up and regulatory compliance in pharmaceutical manufacturing.
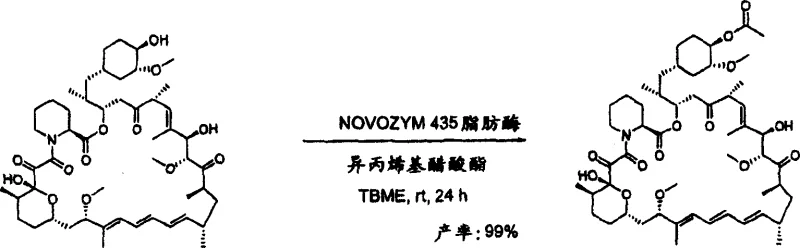
Furthermore, the utilization of acid anhydrides as acyl donors offers an alternative pathway that maintains high regioselectivity while potentially offering cost advantages due to the lower price of anhydrides compared to specialized vinyl esters. In these reactions, the lipase catalyzes the nucleophilic attack of the 42-hydroxyl group on the anhydride, releasing a carboxylic acid byproduct which can be easily removed during workup. The patent data indicates that even with acid anhydrides, the enzymatic system maintains its exquisite selectivity, producing the desired 42-monoester in yields as high as 96% without detectable formation of the 31-isomer. This flexibility in acyl donor selection empowers process chemists to optimize the economic and environmental parameters of the synthesis without sacrificing product quality or purity, making it a highly adaptable platform technology for the production of various rapamycin analogues.

How to Synthesize Rapamycin 42-Ester Derivatives Efficiently
The practical implementation of this enzymatic acylation process is straightforward and designed for ease of operation in both laboratory and pilot plant settings. The general procedure involves suspending the rapamycin starting material and the selected acyl donor in a dry organic solvent such as TBME, followed by the addition of the immobilized lipase catalyst under an inert nitrogen atmosphere to prevent moisture-induced hydrolysis. The reaction mixture is then stirred at a controlled temperature, typically between room temperature and 45°C, until monitoring by thin-layer chromatography (TLC) or HPLC confirms the complete consumption of the starting material. Following the reaction, the solid enzyme catalyst is simply removed by filtration, allowing for potential recycling, and the solvent is evaporated under reduced pressure to yield the crude product which can be purified by standard chromatographic techniques or recrystallization. For a detailed, step-by-step breakdown of the standardized synthesis protocol including specific molar ratios and workup procedures, please refer to the technical guide below.
- Dissolve rapamycin and the chosen acyl donor (such as vinyl acetate, isopropenyl acetate, or acid anhydride) in a suitable organic solvent like tert-butyl methyl ether (TBME).
- Add the lipase catalyst, preferably Candida antarctica lipase B (e.g., Novozym 435) or Pseudomonas cepacia lipase, to the reaction mixture under a nitrogen atmosphere.
- Stir the mixture at mild temperatures ranging from room temperature to 45°C until conversion is complete, then filter off the enzyme and purify the product via chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this lipase-catalyzed synthesis route offers profound advantages for procurement managers and supply chain heads tasked with optimizing the cost and reliability of pharmaceutical intermediate sourcing. By transitioning from multi-step chemical syntheses to a direct enzymatic acylation, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing through the elimination of expensive protecting group reagents, the reduction of solvent volumes, and the minimization of waste disposal costs associated with hazardous chemical byproducts. The mild reaction conditions also translate to lower energy consumption, as there is no need for cryogenic cooling or high-temperature heating, further contributing to a leaner and more sustainable production cost structure. Additionally, the high regioselectivity of the enzyme removes the need for complex and yield-eroding purification steps like preparative HPLC, thereby increasing the overall mass balance and throughput of the facility.
- Cost Reduction in Manufacturing: The enzymatic process eliminates the need for stoichiometric amounts of harsh chemical activators and protecting groups, which are often costly and generate significant waste streams requiring expensive treatment. By streamlining the synthesis into a single catalytic step with high atom economy, the overall cost of goods is drastically lowered, allowing for more competitive pricing in the global market for immunosuppressant ingredients. The ability to use readily available and inexpensive acyl donors like acid anhydrides further enhances the economic viability of the process, ensuring long-term margin stability for suppliers.
- Enhanced Supply Chain Reliability: Reliance on specialized chemical reagents with long lead times is minimized, as the key inputs—rapamycin, common vinyl esters, and commercially available immobilized lipases—are sourced from robust and diversified supply chains. The simplicity of the reaction setup reduces the risk of batch failures due to operator error or equipment malfunction, ensuring consistent on-time delivery of high-purity rapamycin esters to downstream customers. Furthermore, the stability of the immobilized enzyme allows for extended storage and transport without significant loss of activity, mitigating risks associated with raw material degradation during logistics.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently in organic solvents like TBME which are widely accepted in pharmaceutical manufacturing and easier to recover and recycle than many alternative solvents. The "green" nature of the biocatalytic route, characterized by mild conditions and non-toxic catalysts, simplifies regulatory compliance and environmental permitting, accelerating the timeline for commercial scale-up of complex immunosuppressant intermediates. This alignment with sustainability goals not only reduces regulatory friction but also enhances the brand value of the final pharmaceutical product in an increasingly eco-conscious market.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed technical disclosures within patent CN1942476A. These answers address common concerns regarding enzyme selection, substrate scope, and process scalability, providing a clear roadmap for integrating this biocatalytic method into existing production workflows. Understanding these technical nuances is critical for R&D directors aiming to implement reducing lead time for high-purity rapamycin esters in their development pipelines while maintaining strict quality control standards.
Q: How does the enzymatic method distinguish between the 31-OH and 42-OH groups on rapamycin?
A: The lipase enzyme, particularly Candida antarctica lipase B, possesses a specific active site geometry that sterically favors the acylation of the 42-hydroxyl group over the 31-hydroxyl group. This intrinsic regioselectivity eliminates the need for cumbersome protection-deprotection strategies required in traditional chemical synthesis.
Q: What are the preferred acyl donors for this biocatalytic process?
A: The patent highlights vinyl esters (such as vinyl acetate, vinyl chloroacetate, and vinyl benzoate), isopropenyl esters, and acid anhydrides as highly effective acyl donors. Vinyl esters are particularly advantageous because the byproduct (acetaldehyde) tautomerizes, driving the reaction equilibrium forward irreversibly.
Q: Can this enzymatic process be scaled for industrial production of immunosuppressant intermediates?
A: Yes, the process operates under mild conditions (room temperature to 45°C) in common organic solvents like TBME, making it highly amenable to scale-up. The use of immobilized enzymes allows for easy filtration and potential reuse, significantly enhancing process economics and supply chain reliability for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rapamycin 42-Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route described in patent CN1942476A for the production of high-value immunosuppressant intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory benchtop to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of rapamycin 42-ester derivative we supply meets the highest standards of quality and consistency required by global regulatory agencies. We are committed to leveraging our technical expertise to help you navigate the complexities of biocatalytic manufacturing and secure a stable supply of critical intermediates for your drug development programs.
We invite you to engage with our technical procurement team to discuss how this advanced enzymatic technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of switching to this regiospecific synthesis method for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and efficiency in your pharmaceutical manufacturing operations. Let us be your partner in turning complex chemical challenges into commercial successes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
