Advanced Iodine-Catalyzed Perester Synthesis for Scalable Pharmaceutical Manufacturing
Advanced Iodine-Catalyzed Perester Synthesis for Scalable Pharmaceutical Manufacturing
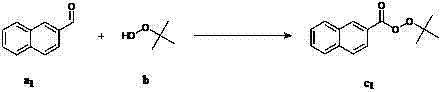
The landscape of organic peroxide synthesis is undergoing a significant transformation driven by the urgent demand for greener, safer, and more economically viable manufacturing processes. Patent CN102153470A introduces a groundbreaking methodology for the preparation of perester compounds, specifically leveraging an iodine-catalyzed oxidation system that directly converts aldehydes into valuable peresters using t-butyl hydroperoxide. This technological advancement represents a paradigm shift away from traditional, hazardous routes that rely on acyl chlorides and strong bases, offering instead a mild, atom-economical pathway that aligns perfectly with modern Green Chemistry principles. For R&D directors and process chemists, this patent provides a robust framework for synthesizing high-purity perester intermediates with exceptional functional group tolerance, while supply chain leaders will appreciate the simplified operational requirements and reduced waste treatment burdens associated with this catalytic system.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of perester compounds has been dominated by the reaction of acyl chlorides with t-butyl hydroperoxide in the presence of strong bases. This conventional approach suffers from several critical drawbacks that hinder its efficiency and sustainability in modern chemical manufacturing. Firstly, acyl chlorides are not always readily available starting materials and often require their own energy-intensive synthesis from carboxylic acids, adding unnecessary steps and cost to the overall process. Secondly, the requirement for excessive amounts of strong bases to neutralize the hydrochloric acid generated during the reaction creates significant environmental liabilities, including the production of large volumes of saline wastewater that require costly treatment. Furthermore, the harsh conditions often associated with acyl chloride chemistry can lead to compatibility issues with sensitive functional groups, limiting the scope of substrates that can be effectively utilized in complex molecule synthesis.
The Novel Approach
In stark contrast, the novel methodology disclosed in patent CN102153470A utilizes a direct oxidative esterification strategy that bypasses the need for activated acyl derivatives entirely. By employing a catalytic system based on inexpensive and readily available iodine species—such as potassium iodide, sodium iodide, or various quaternary ammonium iodides—the process activates the formyl C-H bond of aldehydes directly. This allows for a smooth reaction with t-butyl hydroperoxide under mild thermal conditions ranging from 25°C to 100°C, often conducted simply under an air atmosphere. The elimination of strong bases and corrosive reagents not only enhances operator safety but also drastically simplifies the downstream purification process, typically requiring only standard extraction and silica gel filtration. This approach exemplifies a true leap forward in process intensification, enabling the synthesis of diverse perester structures with high selectivity and minimal environmental impact.
Mechanistic Insights into Iodine-Catalyzed Aldehyde Oxidation
The core of this innovative synthesis lies in the unique ability of iodine catalysts to mediate the oxidation of aldehydes to peresters through a mechanism that likely involves the generation of reactive iodine species in situ. When an iodide salt interacts with t-butyl hydroperoxide, it can generate electrophilic iodine intermediates or radical species that facilitate the abstraction of the aldehydic hydrogen. This activation step is crucial as it renders the carbonyl carbon more susceptible to nucleophilic attack by the hydroperoxide anion or neutral molecule. The subsequent rearrangement and elimination steps lead to the formation of the peroxide bond (O-O) while regenerating the active iodine catalyst, thereby sustaining the catalytic cycle. This mechanistic pathway is distinct from traditional nucleophilic acyl substitution, offering a lower energy barrier for reaction initiation and allowing for much milder operating temperatures that preserve the integrity of thermally labile perester products.
From an impurity control perspective, the mildness of this iodine-catalyzed system offers substantial advantages for producing high-purity pharmaceutical intermediates. Traditional methods involving strong bases often promote side reactions such as aldol condensations or hydrolysis of sensitive ester groups present on the substrate. However, the neutral to slightly acidic nature of the iodine/TBHP system minimizes these degradation pathways. The patent data demonstrates excellent compatibility with a wide array of functional groups, including halogens, ethers, esters, and even unsaturated bonds like alkenes and alkynes, as evidenced by the successful synthesis of complex derivatives. This high level of chemoselectivity ensures that the final perester product requires less rigorous purification, directly translating to higher overall yields and reduced solvent consumption during the isolation phase, which is a key metric for process efficiency.
How to Synthesize t-Butyl Peresters Efficiently
To implement this cutting-edge synthesis route in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize yield while ensuring safety. The process begins with the careful configuration of the reaction system, where the stoichiometry of the oxidant is critical; typically, three equivalents of t-butyl hydroperoxide are employed relative to the aldehyde substrate to drive the equilibrium towards completion. The choice of solvent is also flexible, with options ranging from polar protic solvents like ethanol to aprotic solvents like ethyl acetate or dichloromethane, allowing for optimization based on substrate solubility. Following the reaction period, which can vary from 2 to 48 hours depending on the electronic nature of the aldehyde, the workup involves a quenching step with sodium bisulfite to reduce any residual peroxides, followed by standard organic extraction techniques.
- Configure the reaction system by mixing the aldehyde substrate, t-butyl hydroperoxide (3 equivalents), and an iodine-based catalyst (e.g., KI, NaI, or quaternary ammonium iodides) in a suitable solvent such as ethyl acetate or dichloromethane.
- Stir the reaction mixture under air atmosphere at temperatures ranging from 25°C to 100°C for a duration of 2 to 48 hours, monitoring conversion until the aldehyde is fully consumed.
- Quench the reaction with saturated sodium bisulfite solution, extract the organic phase with ethyl acetate, and purify the crude product via silica gel adsorption or column chromatography to obtain the pure perester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed perester synthesis technology presents a compelling value proposition centered around cost reduction and operational resilience. The primary economic driver is the substitution of expensive and hazardous acyl chlorides with commodity aldehydes, which are widely available from global chemical suppliers at competitive price points. Additionally, the catalyst system relies on simple iodine salts or quaternary ammonium compounds, which are significantly cheaper than transition metal catalysts often used in oxidation chemistry. The elimination of strong bases removes the need for specialized corrosion-resistant equipment and reduces the volume of hazardous waste generated, leading to substantial savings in waste disposal fees and regulatory compliance costs. These factors combine to create a manufacturing process that is not only cheaper per kilogram but also more robust against supply chain disruptions affecting specialty reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the simplification of the synthetic route and the use of low-cost reagents. By avoiding the multi-step preparation of acyl chlorides and eliminating the consumption of stoichiometric amounts of strong bases, the overall material cost is drastically lowered. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified workup procedure minimizes solvent usage and labor hours required for purification. This holistic reduction in operational expenditure makes the production of perester intermediates significantly more margin-friendly for large-scale commercial applications.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by the reliance on universally available starting materials. Aldehydes and t-butyl hydroperoxide are bulk chemicals produced by numerous manufacturers worldwide, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate air and moisture better than many organometallic processes, means that production can continue with fewer interruptions due to environmental controls. This reliability ensures consistent delivery schedules for downstream customers, a critical factor for pharmaceutical companies managing tight production timelines for active pharmaceutical ingredients.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of exothermic hazards associated with strong base neutralization. The reaction can be safely conducted in standard glass-lined or stainless steel reactors without the need for exotic materials. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding volatile organic compounds and saline wastewater discharge. The atom economy of directly converting aldehydes to peresters minimizes byproduct formation, supporting corporate sustainability goals and reducing the carbon footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-catalyzed perester synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN102153470A, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the advantages of using iodine catalysts over traditional acyl chloride methods for perester synthesis?
A: The iodine-catalyzed method described in patent CN102153470A eliminates the need for acyl chlorides, which are often corrosive and require harsh preparation steps. Furthermore, this novel approach operates under mild conditions without the necessity for excessive strong bases, significantly reducing environmental pollution and simplifying the workup procedure compared to conventional methods.
Q: Is this synthesis method compatible with sensitive functional groups?
A: Yes, the protocol demonstrates excellent functional group tolerance. It is effective for a wide range of substrates including aryl aldehydes, heterocyclic aldehydes, and alkyl aldehydes bearing sensitive groups such as halogens, esters, alkenes, and alkynes, making it highly suitable for complex pharmaceutical intermediate synthesis.
Q: What are the typical reaction conditions and catalyst loadings?
A: The reaction typically proceeds at temperatures between 25°C and 100°C under air atmosphere. The catalyst loading is optimized to be greater than or equal to 20% of the molar amount of the aldehyde substrate, ensuring high conversion rates while maintaining economic feasibility for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perester Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-catalyzed perester synthesis route detailed in patent CN102153470A and have integrated similar green chemistry principles into our own CDMO operations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of perester intermediate meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality and safety makes us an ideal partner for organizations seeking to leverage this advanced synthetic technology.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your project volume. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-quality perester compounds with the reliability and speed your supply chain demands.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
