Revolutionizing Avanafil Production via Boric Porphyrin Catalysis for Global Pharmaceutical Supply Chains
Revolutionizing Avanafil Production via Boric Porphyrin Catalysis for Global Pharmaceutical Supply Chains
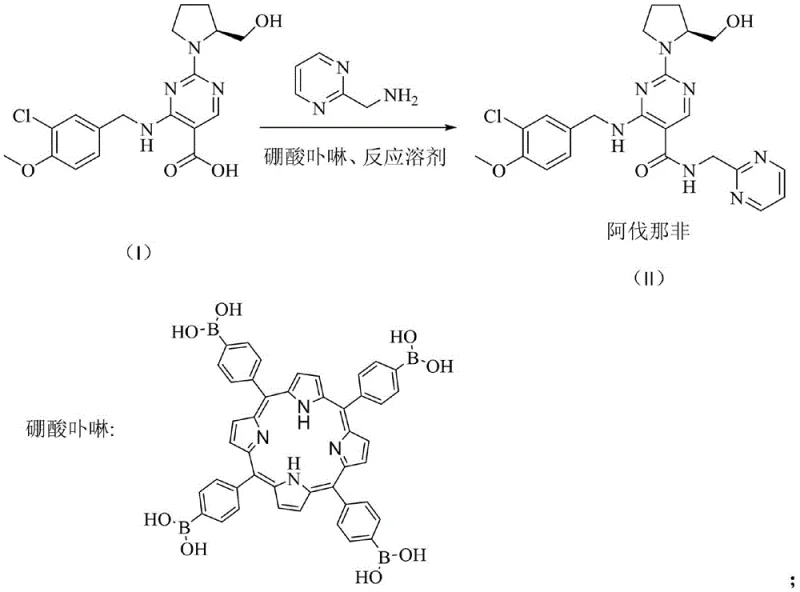
The pharmaceutical industry is constantly seeking more efficient, sustainable, and cost-effective pathways for the synthesis of critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in patent CN109776505B, which discloses a novel preparation method for avanafil, a potent PDE5 inhibitor used in the treatment of erectile dysfunction. This patent introduces a transformative approach by utilizing a boric acid porphyrin catalyst to directly catalyze the amidation reaction between a key carboxylic acid intermediate and an aminomethyl pyrimidine derivative. Unlike traditional methods that rely on stoichiometric amounts of expensive coupling agents, this innovation leverages the unique electronic properties of porphyrin borates to activate the carboxyl group, thereby streamlining the synthetic route. For global procurement leaders and R&D directors, this technology represents a pivotal shift towards greener chemistry, offering a pathway to reduce waste generation while simultaneously enhancing the overall yield and purity profile of the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of avanafil and similar amide-containing pharmaceutical intermediates has relied heavily on carbodiimide-mediated condensation reactions. Standard protocols typically employ reagents such as Dicyclohexylcarbodiimide (DCC) or 1-(3-dimethylaminopropyl)-3-Ethylcarbodiimide (EDCI), often in conjunction with auxiliary acylation activating agents like 1-hydroxybenzotriazole (HOBt). While effective on a small laboratory scale, these conventional methods present severe drawbacks when translated to industrial manufacturing. The primary issue lies in the stoichiometric nature of these reagents; for every mole of product formed, a mole of urea byproduct is generated, leading to poor atom economy. Furthermore, these urea byproducts are notoriously difficult to separate from the target amide, often requiring cumbersome purification steps such as column chromatography or multiple recrystallizations. This not only increases the consumption of solvents and energy but also introduces risks of residual impurities that can compromise the safety profile of the final API, creating significant bottlenecks for supply chain reliability.
The Novel Approach
In stark contrast to the wasteful conventional routes, the method disclosed in patent CN109776505B utilizes a boric acid porphyrin catalyst to drive the amidation reaction directly. This innovative approach eliminates the need for stoichiometric coupling reagents entirely. By dissolving the starting materials, specifically 4-[(3-chloro-4-methoxyphenyl)methylamino]-2-[(S)-2-hydroxymethylpyrrol-1-yl]pyrimidine-5-carboxylic acid and 2-aminomethyl pyrimidine, in a solvent like toluene, the reaction proceeds efficiently under reflux conditions with only a catalytic loading of the porphyrin complex. The most striking advantage of this novel approach is the ease of downstream processing; the catalyst can be easily recovered by filtration after the reaction is quenched with dilute hydrochloric acid. This simplicity drastically reduces the complexity of the workup procedure, minimizes solvent usage, and allows for the recycling of the valuable catalyst, thereby addressing both economic and environmental concerns inherent in traditional amide bond formation strategies.
Mechanistic Insights into Boric Porphyrin-Catalyzed Amidation
To fully appreciate the technical superiority of this method, one must understand the underlying mechanistic principles that govern the catalytic cycle. The core of this transformation lies in the formation of a monoacyloxyboronic acid reactive intermediate. When the aryl boronic acid moiety within the porphyrin structure interacts with the carboxylic acid substrate, it forms a mixed anhydride-like species. This intermediate is electronically distinct because the electron-deficient boron atom, coupled with intramolecular hydrogen bonding, effectively activates the carbonyl carbon of the carboxylic acid. This activation lowers the energy barrier for nucleophilic attack by the amine component, facilitating the formation of the amide bond without the need for external activation by carbodiimides. The porphyrin macrocycle serves as a robust scaffold that stabilizes the boron center, ensuring high catalytic activity and longevity throughout the reaction duration, which typically spans 8 to 16 hours under reflux.
From an impurity control perspective, this mechanism offers profound benefits for R&D teams focused on quality by design. Because the reaction does not generate stable urea byproducts or require auxiliary additives like HOBt, the impurity profile of the crude reaction mixture is significantly cleaner. The absence of these hard-to-remove side products means that the risk of genotoxic impurities or persistent organic contaminants is minimized. Furthermore, the specific interaction between the catalyst and the substrate ensures high chemoselectivity, preventing unwanted side reactions at other sensitive functional groups within the complex avanafil molecule. This high level of selectivity translates directly into higher isolated yields, with experimental data from the patent indicating yields reaching up to 91.2% and purity levels exceeding 99.5%, demonstrating the robustness of this catalytic system in maintaining stringent quality standards.
How to Synthesize Avanafil Efficiently
Implementing this advanced catalytic route requires precise control over reaction parameters to maximize efficiency and reproducibility. The process begins with the careful selection of solvent and molar ratios, where toluene serves as the optimal medium for the reflux reaction. The protocol dictates a molar ratio of the carboxylic acid intermediate to the amine of approximately 1:1.0 to 1.2, ensuring complete conversion while minimizing excess reagent waste. The catalyst loading is remarkably low, ranging from 0.01 to 0.1 equivalents, which underscores the high turnover number of the boric porphyrin complex. Following the reaction, the workup involves a strategic acid-base extraction sequence that leverages the solubility differences between the product and the catalyst. Detailed standardized operating procedures for this synthesis are essential for technology transfer and scale-up.
- Dissolve 4-[(3-chloro-4-methoxyphenyl)methylamino]-2-[(S)-2-hydroxymethylpyrrol-1-yl]pyrimidine-5-carboxylic acid and 2-aminomethyl pyrimidine in toluene, add boric porphyrin catalyst, and reflux for 8-16 hours.
- Cool the reaction mixture, add dilute hydrochloric acid to dissolve the product, filter to recover the catalyst, separate layers, and adjust pH to 7.0 with NaOH to precipitate crude avanafil.
- Recrystallize the crude product from anhydrous methanol with activated carbon decolorization to obtain high-purity avanafil finished product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this boric porphyrin catalyzed process offers tangible strategic advantages that extend beyond mere technical novelty. The shift from stoichiometric reagents to a catalytic system fundamentally alters the cost structure of avanafil manufacturing. By eliminating the need for large quantities of expensive coupling agents like EDCI and HOBt, the raw material costs are significantly reduced. Moreover, the ability to recover and recycle the catalyst further drives down the cost per kilogram of the final product. This economic efficiency is compounded by the simplified purification process, which reduces the consumption of solvents and energy associated with extensive chromatographic separations, leading to substantial cost savings in utility and waste disposal expenditures.
- Cost Reduction in Manufacturing: The elimination of stoichiometric coupling reagents represents a major financial benefit. Traditional methods consume expensive reagents that end up as waste, whereas this catalytic method uses minute amounts of a reusable catalyst. This structural change in the bill of materials leads to a drastic reduction in direct material costs. Additionally, the simplified workup reduces the labor hours and equipment time required for purification, contributing to a lower overall cost of goods sold (COGS) for the manufactured intermediate.
- Enhanced Supply Chain Reliability: Relying on complex supply chains for specialized coupling reagents can introduce vulnerabilities. This new method utilizes widely available and stable starting materials, reducing dependency on niche chemical suppliers. The robustness of the reaction conditions and the ease of catalyst recovery ensure consistent batch-to-batch quality, minimizing the risk of production delays caused by failed batches or purification bottlenecks. This reliability is crucial for maintaining continuous supply to downstream API manufacturers.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental impact intensify, green chemistry solutions are becoming mandatory. This process generates significantly less chemical waste due to improved atom economy and the absence of urea byproducts. The reduced solvent load and the potential for solvent recycling align perfectly with modern environmental, social, and governance (ESG) goals. This makes the process highly scalable and compliant with stringent environmental regulations, facilitating smoother regulatory approvals for commercial production facilities.
Frequently Asked Questions (FAQ)
Understanding the nuances of this new technology is vital for stakeholders evaluating its potential for integration into existing manufacturing portfolios. The following questions address common inquiries regarding the technical feasibility and operational benefits of the boric porphyrin catalyzed synthesis of avanafil. These answers are derived directly from the experimental data and technical disclosures found in the relevant patent literature, providing a factual basis for decision-making.
Q: What are the advantages of using boric porphyrin catalyst over traditional carbodiimide reagents?
A: Unlike traditional carbodiimide reagents like DCC or EDCI which produce stoichiometric urea byproducts that are difficult to separate, the boric porphyrin catalyst operates in catalytic amounts (0.01 to 0.1 eq), allows for easy recovery via filtration, and significantly improves atom economy and product purity.
Q: How does this method impact the purification process of avanafil?
A: The method simplifies purification by allowing the catalyst to be filtered off directly after acid treatment. The product is then extracted and precipitated by pH adjustment, avoiding complex chromatographic separations often required to remove coupling reagent byproducts in conventional methods.
Q: Is this catalytic process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It uses common solvents like toluene and methanol, operates under standard reflux conditions, and the catalyst is recyclable, making it cost-effective and environmentally friendly for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avanafil Supplier
The transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering comprehensive support for the commercialization of advanced pharmaceutical intermediates like avanafil. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent examples can be reliably reproduced on an industrial scale. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to explore how this catalytic technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into the economic benefits tailored to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your avanafil supply.
