Advanced Synthesis of N-Boc Piperazine: Technical Upgrade and Commercial Scalability
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for key heterocyclic intermediates that balance high purity with economic feasibility. A significant breakthrough in this domain is detailed in patent CN108033931B, which outlines a novel three-step synthesis method for N-Boc piperazine, a critical building block for numerous active pharmaceutical ingredients (APIs). This patent describes a process that starts from diethanolamine, undergoing chlorination, Boc protection, and aminolysis cyclization to yield the target compound with exceptional efficiency. The strategic value of this methodology lies in its ability to bypass the traditional reliance on expensive anhydrous piperazine, instead utilizing widely available and cost-effective starting materials. For R&D directors and procurement managers alike, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols. The structural integrity of the final product, N-tert-butoxycarbonylpiperazine, is paramount for downstream drug synthesis, and this method ensures the preservation of the piperazine ring structure while introducing the necessary protecting group with high fidelity.
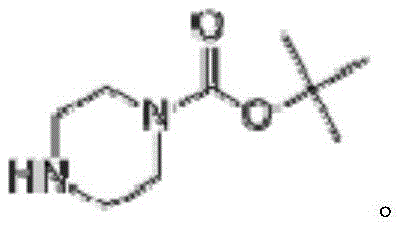
N-Boc piperazine belongs to the class of piperazine monosubstituted compounds, which serve as indispensable scaffolds in the construction of complex therapeutic molecules. Its utility spans across various therapeutic areas, including the synthesis of glucocorticoids and kinase inhibitors such as palbociclib and ranolazine. The economic and pharmaceutical value of synthesizing this intermediate efficiently cannot be overstated, as it directly impacts the cost of goods sold (COGS) for the final drug products. By adopting the route disclosed in the patent data, manufacturers can secure a reliable supply of high-purity intermediates while mitigating the risks associated with volatile raw material markets. This approach not only enhances the supply chain resilience for pharmaceutical intermediates but also aligns with the growing industry demand for greener chemical processes that minimize environmental footprints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-Boc piperazine has been plagued by significant inefficiencies and safety concerns inherent to traditional synthetic routes. One common method involves the direct reaction of anhydrous piperazine with di-tert-butyl dicarbonate; however, this approach suffers from poor selectivity, often resulting in di-substituted byproducts that are difficult to separate. The purification process typically requires extensive washing with large volumes of water, which drastically reduces the overall yield and generates substantial wastewater, thereby inflating the cost of three-waste treatment. Another prevalent method utilizes glacial acetic acid to form a salt with piperazine before acylation, followed by extraction with toluene. While this improves selectivity, the reliance on anhydrous piperazine as a starting material imposes a heavy financial burden due to its high market price. Furthermore, the use of toluene, a toxic aromatic solvent, poses serious health risks to workers and complicates regulatory compliance regarding solvent residues in pharmaceutical products.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent data offers a transformative solution by reimagining the synthetic pathway from the ground up. By selecting diethanolamine as the starting material, the process leverages a commodity chemical that is significantly more affordable and readily available than anhydrous piperazine. The three-step sequence of chlorination, protection, and cyclization is designed to maximize atom economy and minimize waste generation. The reaction conditions are notably mild, avoiding the extreme temperatures or pressures that often characterize less efficient processes. This new route effectively eliminates the need for toxic solvents like toluene, replacing them with safer alternatives such as ethyl acetate and water. For supply chain heads, this translates to a more stable production schedule with fewer interruptions caused by regulatory hurdles or raw material shortages, ensuring a continuous flow of high-quality intermediates to downstream customers.
Mechanistic Insights into Diethanolamine-Based Cyclization
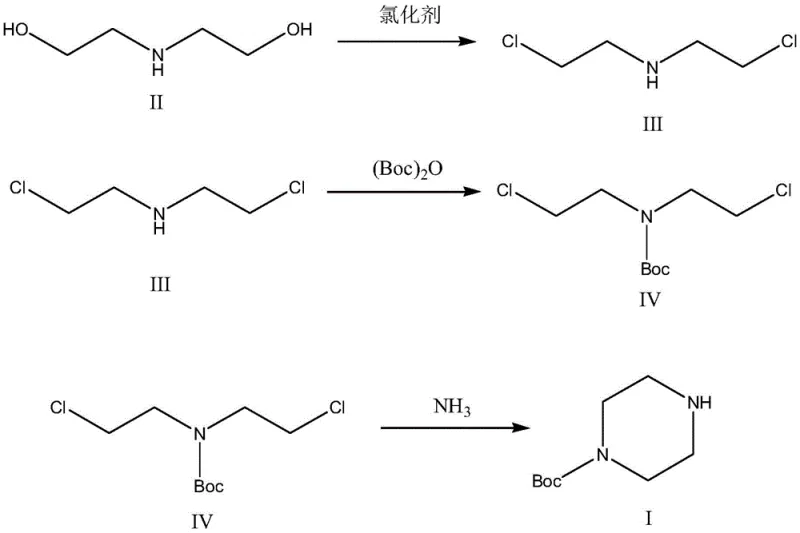
The core of this synthetic innovation lies in the precise control of the reaction mechanism, which ensures high conversion rates and minimal impurity formation. The process begins with the chlorination of diethanolamine using a chlorinating agent such as thionyl chloride (SOCl2), converting the hydroxyl groups into chloro groups to form bis(2-chloroethyl)amine. This intermediate is then subjected to Boc protection using Boc anhydride under alkaline conditions, which selectively protects the nitrogen atom without affecting the chloro groups. The final and most critical step is the aminolysis cyclization, where the protected intermediate reacts with ammonia water to close the piperazine ring. The use of ammonia water at controlled temperatures facilitates the nucleophilic attack required for ring closure while maintaining the integrity of the Boc protecting group. This mechanistic pathway is robust and reproducible, making it ideal for large-scale manufacturing where consistency is key.
Impurity control is another critical aspect where this method excels, particularly for R&D directors focused on the quality of API intermediates. The stepwise nature of the synthesis allows for intermediate purification, ensuring that impurities from the chlorination step do not carry over into the final product. The alkaline conditions maintained during the Boc protection step prevent the formation of acidic byproducts that could degrade the product quality. Furthermore, the cyclization step is optimized to minimize the formation of oligomers or polymeric side products, which are common pitfalls in piperazine synthesis. The result is a final product with purity levels consistently exceeding 99.4%, as demonstrated in the patent examples. This high level of purity reduces the need for extensive downstream purification, saving both time and resources in the overall manufacturing process.
How to Synthesize N-Boc Piperazine Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters to ensure optimal yield and safety. The process is designed to be straightforward, utilizing standard chemical engineering equipment and common reagents. The initial chlorination step is performed under reflux, followed by a controlled quench with water to manage exothermic heat. The subsequent protection and cyclization steps are carried out at moderate temperatures, reducing energy consumption and equipment stress. For technical teams looking to adopt this method, the patent provides detailed guidance on molar ratios, reaction times, and temperature profiles. The following guide outlines the standardized steps derived from the patent data to facilitate a smooth technology transfer.
- Chlorination of diethanolamine using thionyl chloride under reflux to form bis(2-chloroethyl)amine.
- Protection of the amine group using Boc anhydride under alkaline conditions to form the protected intermediate.
- Cyclization reaction with ammonia water at 55-65°C to close the piperazine ring and yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers substantial advantages that directly address the pain points of procurement managers and supply chain leaders. The primary benefit is the significant reduction in raw material costs achieved by substituting expensive anhydrous piperazine with low-cost diethanolamine. This switch not only lowers the direct material cost but also simplifies the sourcing strategy, as diethanolamine is a bulk chemical with a stable global supply. Additionally, the elimination of toxic solvents like toluene reduces the costs associated with solvent recovery, waste disposal, and environmental compliance. These factors combine to create a more cost-effective manufacturing process that enhances the overall competitiveness of the supply chain.
- Cost Reduction in Manufacturing: The shift to diethanolamine as a starting material fundamentally alters the cost structure of N-Boc piperazine production. By avoiding the premium pricing of anhydrous piperazine, manufacturers can achieve substantial cost savings that can be passed down to customers or reinvested in process optimization. The high yield of the process, reported to be over 93.5%, further contributes to cost efficiency by maximizing the output from each batch of raw materials. Moreover, the simplified workup procedure, which avoids complex extraction and washing steps, reduces labor and utility costs. These cumulative savings make the process highly attractive for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly improved due to the use of widely available and stable raw materials. Diethanolamine and thionyl chloride are commodity chemicals with multiple global suppliers, reducing the risk of supply disruptions compared to specialized reagents. The mild reaction conditions also mean that the process can be easily scaled up or down based on market demand without requiring specialized high-pressure or high-temperature equipment. This flexibility allows manufacturers to respond quickly to changes in customer orders, ensuring timely delivery of high-purity pharmaceutical intermediates. The robustness of the process minimizes batch failures, further stabilizing the supply chain and building trust with long-term partners.
- Scalability and Environmental Compliance: The environmental profile of this synthesis method is superior to conventional routes, aligning with the increasing regulatory pressure for greener manufacturing. The absence of toxic solvents like toluene simplifies waste treatment and reduces the environmental footprint of the production facility. The use of water and ethyl acetate, which are easier to handle and dispose of, ensures compliance with strict environmental regulations. This environmental compliance is not just a regulatory necessity but also a competitive advantage, as more pharmaceutical companies prefer suppliers with sustainable practices. The scalability of the process is proven by the successful demonstration of multi-gram to kilogram scale reactions in the patent examples, indicating readiness for industrial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of N-Boc piperazine. These answers are derived directly from the patent data and practical manufacturing experience, providing clarity for stakeholders evaluating this technology. Understanding these details is crucial for making informed decisions about process adoption and supply chain integration. The information covers aspects ranging from raw material selection to quality control standards.
Q: What are the primary advantages of using diethanolamine over anhydrous piperazine?
A: Using diethanolamine significantly reduces raw material costs compared to expensive anhydrous piperazine. It also eliminates the need for toxic solvents like toluene, improving workplace safety and reducing waste treatment expenses.
Q: What purity levels can be achieved with this synthesis method?
A: The patented process consistently achieves purity levels exceeding 99.4%, with yields typically above 93.5%, making it suitable for high-specification pharmaceutical applications without extensive recrystallization.
Q: Is this process scalable for industrial production?
A: Yes, the reaction conditions are mild (55-65°C) and utilize common reagents like thionyl chloride and ammonia water, ensuring excellent scalability from pilot plants to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Boc Piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development and manufacturing. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering N-Boc piperazine that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced synthesis methods like the one described in CN108033931B, we can offer you a product that combines technical excellence with commercial viability.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific requirements. Whether you need specific COA data to verify quality or route feasibility assessments to explore new applications, we are here to support your goals. Contact us today to discuss how we can become your trusted partner in the production of high-purity pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
