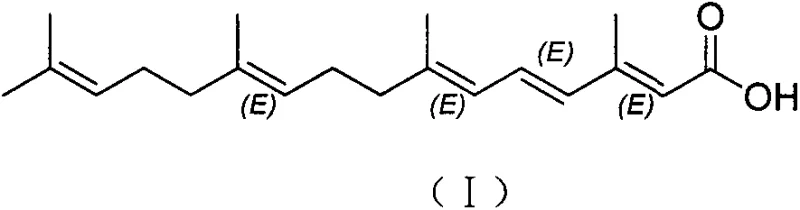
Advanced Manufacturing of Peretinoin: A Scalable Route for Hepatocellular Carcinoma Therapy
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex oncology therapeutics, and the synthesis of Peretinoin (Pelretin A acid) represents a critical area of development for treating hepatocellular carcinoma recurrence. Patent CN103626649A introduces a transformative methodology that shifts the paradigm from expensive, harsh synthetic routes to a streamlined process utilizing trans-farnesol as the primary feedstock. This innovation addresses the longstanding challenges of isomer control and purification that have plagued previous manufacturing attempts. By leveraging mild oxidation conditions and a novel mixed-base Wittig-Horner coupling strategy, the process achieves crude product purity exceeding 85 percent, which drastically simplifies downstream processing. For R&D directors and supply chain managers, this represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering high-quality active ingredients with improved economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
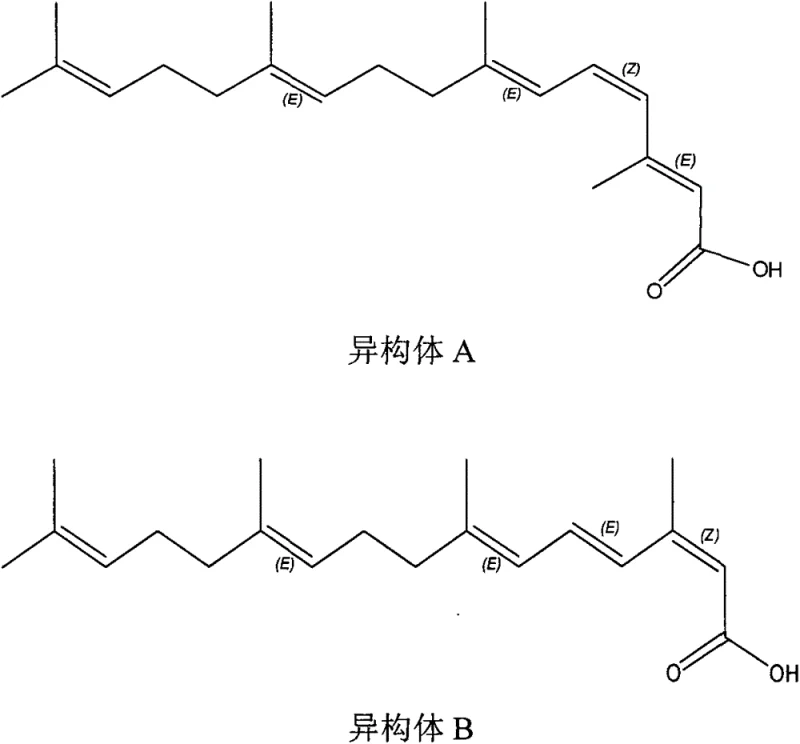
Historically, the industrial preparation of Peretinoin has been hindered by reliance on sophisticated and costly starting materials such as trimethylammonium ketones or toluenesulfonyl derivatives, as detailed in earlier patents like US4988732. These legacy processes often necessitate extreme reaction conditions, including cryogenic temperatures down to minus 50 degrees Celsius under strict nitrogen protection, which imposes severe energy burdens and operational complexity on manufacturing facilities. Furthermore, traditional methods frequently employ sodium ethoxide as a singular base in the Wittig-Horner step, a condition known to promote the formation of multiple cis-trans isomers. These geometric isomers, particularly the stubborn Isomer B, possess physical properties similar to the target molecule, making their removal via recrystallization exceptionally difficult and resulting in substantial yield losses. The cumulative effect of these factors is a process that is neither economically viable nor easily scalable for meeting global demand.
The Novel Approach
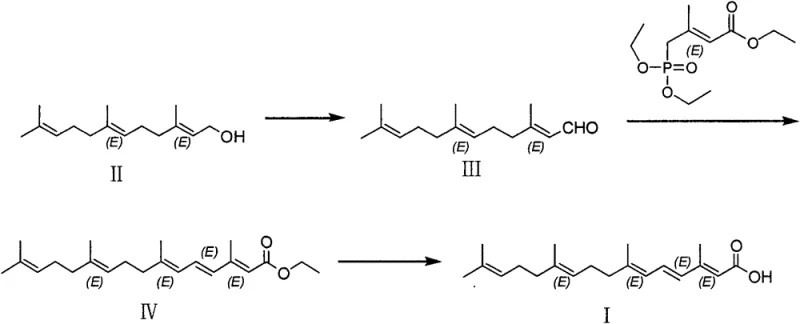
The disclosed invention fundamentally re-engineers the synthetic pathway by initiating the sequence with trans-farnesol, a commercially abundant and inexpensive terpene alcohol. As illustrated in the reaction scheme below, the process flows through a gentle oxidation to an aldehyde, followed by a highly selective chain extension and final hydrolysis. This approach eliminates the need for cryogenic reactors, operating instead at manageable temperatures ranging from minus 30 to 10 degrees Celsius, or even room temperature for the oxidation step. The strategic implementation of a mixed base system during the carbon-carbon bond-forming step effectively suppresses the generation of unwanted geometric isomers at the source. Consequently, the crude product emerges with significantly higher purity, reducing the burden on purification units and enabling a more straightforward path to the greater than 99 percent purity required for bulk drug substances.
Mechanistic Insights into Mixed-Base Wittig-Horner Coupling
The core technical breakthrough of this methodology lies in the precise modulation of the Wittig-Horner reaction environment to control stereochemistry. In standard protocols, the use of strong alkoxides like sodium ethoxide creates a highly basic environment that can facilitate thermodynamic equilibration towards less desirable cis-isomers. The inventors discovered that introducing lithium tert-butoxide in conjunction with sodium ethoxide creates a synergistic effect that kinetically favors the formation of the desired all-trans configuration. This mixed base system appears to stabilize the transition state or alter the aggregation of the enolate species, thereby minimizing the production of Isomer A and, more critically, Isomer B. By optimizing the molar ratio of these bases, specifically favoring a 1:4 ratio of sodium ethoxide to lithium tert-butoxide in THF, the process achieves a dramatic reduction in impurity profiles compared to single-base alternatives.
Beyond the coupling step, the selection of oxidants and hydrolysis solvents plays a pivotal role in maintaining structural integrity throughout the synthesis. The use of 2-iodoxybenzoic acid (IBX) or Dess-Martin periodinane for the initial oxidation of trans-farnesol ensures selective conversion to the aldehyde without over-oxidation to the carboxylic acid, a common side reaction with harsher oxidants like PCC. Furthermore, the hydrolysis step avoids the pitfalls of transesterification observed when using isopropanol as a sole solvent. By employing a mixed solvent system of tetrahydrofuran and water, or ethanol and water, the reaction proceeds cleanly to the free acid. This solvent engineering prevents the formation of iso-propyl ester byproducts that would otherwise complicate the final isolation and reduce the overall yield of the high-purity API intermediate.
How to Synthesize Peretinoin Efficiently
The synthesis of Peretinoin via this patented route involves three distinct operational stages that can be seamlessly integrated into existing multipurpose reactor trains. The process begins with the oxidation of trans-farnesol, followed by the critical Wittig-Horner coupling using the optimized mixed base, and concludes with a controlled hydrolysis. Each step has been refined to maximize yield while minimizing the generation of hazardous waste or difficult-to-separate impurities. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for process chemists aiming to implement this technology. Detailed standardized synthesis steps are provided in the guide below.
- Oxidize trans-farnesol using IBX or Dess-Martin reagent to form the corresponding aldehyde intermediate.
- Perform Wittig-Horner reaction using a mixed base system (Sodium Ethoxide and Lithium tert-butoxide) to couple the aldehyde with the phosphonate ester.
- Hydrolyze the resulting ester using potassium hydroxide in a THF-water mixed solvent to obtain high-purity Peretinoin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this trans-farnesol-based route offers compelling economic and logistical benefits that directly impact the bottom line. The substitution of exotic, low-volume starting materials with commodity-grade trans-farnesol significantly de-risks the supply chain, ensuring consistent availability of raw materials even during market fluctuations. Moreover, the elimination of cryogenic requirements (-50°C) translates to substantial energy savings and allows the reaction to be performed in standard glass-lined steel reactors without specialized cooling infrastructure. This simplification of equipment requirements lowers the barrier to entry for contract manufacturing organizations and facilitates faster technology transfer between sites.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the drastic reduction in raw material costs and the simplification of purification workflows. By utilizing trans-farnesol, which is widely produced for the fragrance and flavor industries, manufacturers can avoid the premium pricing associated with specialized ketone or sulfone precursors. Additionally, the high selectivity of the mixed-base reaction means that less material is lost during recrystallization steps, leading to higher overall throughput. The avoidance of expensive heavy metal catalysts or complex protecting group strategies further contributes to a leaner cost structure, allowing for competitive pricing in the global pharmaceutical intermediates market.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions, which are far less sensitive to minor variations in temperature or moisture compared to the fragile cryogenic processes of the past. The use of common solvents like THF, ethanol, and water ensures that solvent supply chains remain stable and cost-effective. Furthermore, the ability to achieve high crude purity (>85%) reduces the dependency on extensive chromatographic purification, which is often a bottleneck in large-scale production. This reliability ensures that delivery schedules for critical oncology drugs can be met consistently, minimizing the risk of stockouts for downstream drug product manufacturers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route offers a greener profile by avoiding the use of highly toxic reagents and reducing the volume of organic waste generated per kilogram of product. The mild reaction temperatures reduce the thermal load on the facility, enhancing operational safety. The process is inherently scalable, having been designed with industrial feasibility in mind, moving from gram-scale experiments to potential multi-ton production without fundamental changes to the chemistry. This scalability ensures that as demand for Peretinoin grows, the manufacturing capacity can be expanded rapidly to meet clinical and commercial needs without compromising quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is essential for process development teams evaluating the feasibility of adopting this technology for commercial production. The answers below clarify the specific advantages regarding isomer control and solvent selection.
Q: How does the new method control cis-trans isomers in Peretinoin synthesis?
A: The method utilizes a specific mixed base system (Sodium Ethoxide combined with Lithium tert-butoxide) during the Wittig-Horner reaction. Experimental data indicates this combination significantly suppresses the formation of difficult-to-remove Isomer B compared to traditional single-base systems like Sodium Ethoxide alone.
Q: Why is trans-farnesol preferred as a starting material?
A: Trans-farnesol is commercially readily available and cost-effective compared to the complex ketones or sulfones used in prior art. Additionally, its oxidation to the aldehyde under mild conditions (using IBX or Dess-Martin) preserves the stereochemistry without generating significant impurities.
Q: What prevents transesterification during the hydrolysis step?
A: Unlike prior methods that used isopropanol which led to transesterification and purity loss, this process employs a mixed solvent system of Tetrahydrofuran (THF) and water (or ethanol and water). This solvent choice ensures clean hydrolysis of the ester to the acid without altering the molecular structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Peretinoin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of complex oncology intermediates like Peretinoin. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications. Our facility is equipped to handle the specific solvent systems and mixed-base reagents required for this advanced synthesis, positioning us as a strategic partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that align with your corporate goals for cost efficiency and product quality. Let us collaborate to bring this life-saving medication to patients faster and more affordably.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
