Advanced Synthesis of 2,7-bis-(N-phenothiazinyl)fluorenone for High-Efficiency OLED Displays
Introduction to Next-Generation Blue Host Materials
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only emit pure colors but also possess exceptional thermal stability and charge transport properties. Patent CN103193733A introduces a breakthrough in this domain with the synthesis of 2,7-bis-(N-phenothiazinyl)fluorenone, often abbreviated as DPhFO. This compound represents a significant leap forward as a blue light host material, leveraging the unique electron-rich nature of the phenothiazine moiety combined with the rigid fluorenone core. Unlike traditional planar structures that suffer from aggregation-caused quenching, the non-planar geometry of the phenothiazine units in DPhFO effectively suppresses intermolecular interactions, leading to superior luminescence efficiency. As a reliable OLED material supplier, understanding the structural nuances of such compounds is critical for developing next-generation displays that offer longer lifespans and lower power consumption.
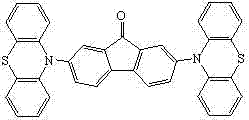
The strategic placement of phenothiazine groups at the 2 and 7 positions of the fluorenone backbone creates a highly conjugated system that facilitates hole injection and transport. This structural design is not merely theoretical; it addresses the fundamental bottleneck in OLED efficiency where exciton management is paramount. By fine-tuning the electronic structure through these specific substituents, the material achieves a narrow optical bandgap suitable for blue emission, a spectrum notoriously difficult to stabilize in organic electronics. For R&D directors seeking high-purity OLED material solutions, this patent provides a validated pathway to a compound that balances electronic performance with synthetic feasibility, setting a new benchmark for blue host materials in the competitive display market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex N-substituted heterocyclic compounds for electronic applications has relied heavily on palladium-catalyzed cross-coupling reactions, such as the Buchwald-Hartwig amination. While effective, these conventional methods present significant drawbacks for large-scale manufacturing, primarily due to the prohibitive cost of palladium catalysts and the stringent requirement for removing trace metal residues to parts-per-billion levels. Furthermore, traditional routes often involve multiple protection and deprotection steps to manage the reactivity of the amine groups, leading to reduced overall atom economy and increased waste generation. The reliance on expensive ligands and inert atmosphere conditions throughout multi-step sequences further inflates the production cost, making cost reduction in electronic chemical manufacturing a persistent challenge for procurement teams. Additionally, the thermal instability of some intermediates in these older pathways can lead to inconsistent batch quality, posing risks to supply chain continuity.
The Novel Approach
In stark contrast, the methodology outlined in CN103193733A employs a direct copper-catalyzed coupling strategy that dramatically simplifies the synthetic landscape. By utilizing cuprous iodide (CuI) in conjunction with 1,10-phenanthroline and pyridine as ligands, the process achieves efficient C-N bond formation without the need for precious metals. The use of 1,3-dimethylpropylene urea (DMPU) as a co-solvent or additive plays a pivotal role in solubilizing the inorganic base and stabilizing the catalytic species at elevated temperatures of 180-200°C. This novel approach eliminates the need for complex protecting group chemistry, allowing for a direct one-pot reaction between phenothiazine and 2,7-dibromofluorenone. The result is a streamlined process that not only reduces raw material costs but also minimizes the environmental footprint by cutting down on solvent usage and purification steps, aligning perfectly with modern green chemistry principles.

Mechanistic Insights into Copper-Catalyzed C-N Coupling
The core of this synthesis lies in the Ullmann-type condensation mechanism, facilitated by the CuI/phenanthroline catalytic system. In this cycle, the copper(I) species initially coordinates with the nitrogen atom of the phenothiazine, activating it for nucleophilic attack. Simultaneously, the oxidative addition of the aryl bromide (2,7-dibromofluorenone) to the copper center generates a transient copper(III) intermediate, although the exact oxidation state dynamics can vary depending on the ligand environment. The presence of DMPU is crucial here, as it acts as a polar aprotic solvent that enhances the solubility of the potassium carbonate base, ensuring a steady supply of the deprotonated phenothiazine anion. This high-temperature environment (180-200°C) provides the necessary activation energy to overcome the kinetic barriers typically associated with unactivated aryl halides in copper catalysis. For technical teams, understanding this mechanism highlights the robustness of the catalyst system, which tolerates the electron-rich nature of the phenothiazine substrate without undergoing premature deactivation.
From an impurity control perspective, the symmetry of the reaction and the specific choice of reagents contribute to a clean product profile. The use of a slight excess of phenothiazine (molar ratio up to 1:0.3-0.5 relative to dibromofluorenone) ensures that the dibromo precursor is fully consumed, minimizing the presence of mono-substituted byproducts which could act as trap sites in the final OLED device. The subsequent purification via silica gel chromatography using a petroleum ether and dichloromethane gradient effectively removes copper residues and unreacted starting materials. This rigorous control over the impurity profile is essential for achieving the high purity specifications required for electronic grade materials, where even trace contaminants can drastically reduce device lifetime. The resulting DPhFO exhibits a thermal decomposition temperature exceeding 380°C, a testament to the structural integrity achieved through this optimized synthetic route.
How to Synthesize 2,7-bis-(N-phenothiazinyl)fluorenone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the careful weighing of reagents under an inert atmosphere to prevent oxidation of the sensitive phenothiazine and copper catalyst. The reaction mixture is subjected to prolonged heating in the dark to prevent any potential photo-degradation of the intermediates, a detail often overlooked in standard protocols but critical for this specific chemistry. Following the reaction, the workup involves a hot filtration and crystallization strategy that leverages the solubility differences of the product in dichloromethane, followed by column chromatography to achieve electronic-grade purity. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different manufacturing scales.
- Mix phenothiazine, 2,7-dibromofluorenone, potassium carbonate, and cuprous iodide in a molar ratio of 1: (0.3-0.5):(2-3):(1-1.2) under nitrogen protection.
- Add pyridine and 1,10-phenanthroline (0.2-0.3% mass of phenothiazine each) and 1,3-dimethylpropylene urea (DMPU, 5-7.5% mass).
- Stir the mixture without light at 180-200°C for 60-90 hours, then purify via silica gel chromatography using petroleum ether and dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed route offers tangible strategic benefits beyond mere technical performance. The shift away from palladium-based chemistry immediately alleviates the pressure associated with the volatility of precious metal markets, providing a more stable cost structure for long-term contracts. Furthermore, the use of commodity chemicals like phenothiazine and simple inorganic bases ensures that raw material sourcing is not bottlenecked by specialized suppliers, thereby enhancing supply chain resilience. The simplified workflow, characterized by fewer unit operations and the elimination of protection/deprotection cycles, translates directly into reduced manufacturing lead times and lower operational expenditures. These factors collectively position DPhFO as a commercially viable candidate for mass adoption in the OLED industry.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with abundant copper salts results in substantial cost savings on catalyst procurement and recovery systems. Additionally, the high atom economy of the direct coupling reaction minimizes waste disposal costs, while the ability to run the reaction at high concentrations improves reactor throughput. By eliminating the need for cryogenic conditions or ultra-dry solvents often required in alternative methods, utility costs for heating and cooling are also significantly optimized. This holistic reduction in processing complexity allows for a more competitive pricing model for the final high-purity OLED material without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as potassium carbonate and cuprous iodide mitigates the risk of supply disruptions that often plague specialty reagent markets. The robustness of the reaction conditions, which tolerate a range of stoichiometric ratios without significant yield loss, adds a layer of operational flexibility that is invaluable during raw material shortages. Moreover, the scalability of the process from gram to kilogram scales has been demonstrated with consistent yields greater than 70%, ensuring that production targets can be met reliably. This stability is crucial for maintaining continuous production lines in the fast-paced consumer electronics sector where display panel demand fluctuates rapidly.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry initiatives by reducing the E-factor (mass of waste per mass of product) through simplified purification steps. The absence of toxic tin or boron reagents, commonly found in Stille or Suzuki couplings, simplifies wastewater treatment and regulatory compliance. The high thermal stability of the final product also implies better handling safety and storage characteristics, reducing logistical hazards. As global regulations on chemical manufacturing tighten, adopting such environmentally benign synthetic routes future-proofs the supply chain against potential regulatory hurdles and carbon taxes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,7-bis-(N-phenothiazinyl)fluorenone. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear picture of the material's capabilities. Understanding these aspects helps stakeholders make informed decisions about integrating this material into their existing product portfolios or R&D pipelines.
Q: What are the key advantages of using phenothiazine derivatives in OLEDs?
A: Phenothiazine derivatives offer a non-planar structure that prevents π-bond aggregation and exciplex formation, thereby enhancing quantum efficiency and device stability compared to planar carbazole analogs.
Q: How does the CuI/DMPU catalytic system improve yield?
A: The combination of CuI with DMPU and nitrogen ligands facilitates efficient C-N bond formation at high temperatures, achieving yields greater than 70% while avoiding expensive palladium catalysts.
Q: Is this material suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials and standard high-temperature stirring equipment, making it highly scalable for industrial manufacturing of display materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-bis-(N-phenothiazinyl)fluorenone Supplier
The synthesis of 2,7-bis-(N-phenothiazinyl)fluorenone represents a convergence of advanced organic synthesis and practical industrial engineering, offering a pathway to high-performance blue host materials. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and thermal stability testing, which are critical for electronic grade materials. We understand that consistency is key in the display industry, and our process controls are designed to deliver batch-to-batch reproducibility that meets the exacting standards of global panel manufacturers.
We invite you to collaborate with us to optimize your supply chain for next-generation OLED components. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient synthesis route can lower your total cost of ownership. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us help you secure a stable, high-quality supply of this critical blue light host material.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
