Advanced Synthesis of Quinazoline-2,4-dione via CO2 Fixation for Commercial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking sustainable pathways to construct privileged heterocyclic scaffolds, and patent CN112778219A presents a groundbreaking approach to synthesizing 2,4-(1H,3H)-quinazoline diketone compounds. This specific patent details a novel methodology that utilizes carbon dioxide as a C1 synthon in conjunction with 2-aminobenzonitrile derivatives, mediated by a specialized 2-hydroxypyridine ionic liquid system. The significance of this technology lies in its ability to transform a greenhouse gas into a valuable building block under relatively mild conditions, thereby addressing both economic and environmental challenges simultaneously. For R&D directors and procurement managers, this represents a shift away from traditional, hazardous reagents towards a greener, more cost-effective paradigm that does not compromise on yield or purity. The technical robustness of this CO2 fixation strategy suggests a high potential for commercial scale-up, offering a reliable supply chain solution for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazoline-2,4-dione derivatives has relied heavily on the use of anthranilic acid reacting with urea or, more problematically, the use of anthranilamide with phosgene. These traditional routes are fraught with significant industrial drawbacks, primarily concerning safety and environmental compliance. The use of phosgene, a highly toxic and regulated gas, necessitates expensive containment infrastructure and rigorous safety protocols that drastically increase capital expenditure and operational complexity. Furthermore, reactions involving urea often require harsh thermal conditions that can lead to the formation of unwanted by-products and impurities, complicating the downstream purification process. These legacy methods also suffer from poor atom economy, generating substantial waste streams that require costly treatment before disposal. For supply chain heads, the reliance on such hazardous materials introduces volatility and risk, as regulatory changes can abruptly impact the availability and cost of key reagents.
The Novel Approach
In stark contrast, the method disclosed in CN112778219A leverages the unique properties of 2-hydroxypyridine ionic liquids to activate carbon dioxide effectively at atmospheric or low pressures. This innovative approach bypasses the need for toxic phosgene entirely, replacing it with an abundant, inexpensive, and non-toxic carbon source. The ionic liquid serves a dual function as both solvent and catalyst, creating a homogeneous reaction environment that enhances mass transfer and reaction kinetics. This results in a streamlined process where reaction conditions are significantly milder, typically ranging from 30°C to 150°C, which reduces energy consumption and thermal stress on the equipment. The simplicity of the work-up procedure, often involving merely the addition of water to precipitate the product, further underscores the operational efficiency of this novel route. By eliminating complex separation steps and hazardous reagents, this technology offers a compelling value proposition for manufacturers seeking to optimize their production lines.
Mechanistic Insights into Ionic Liquid Catalyzed CO2 Cycloaddition
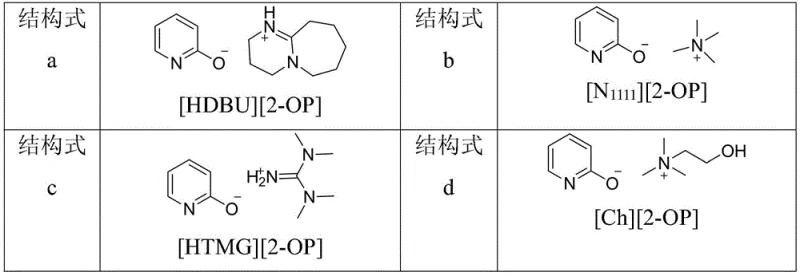
The core of this technological advancement lies in the specific interaction between the 2-hydroxypyridine ionic liquid and the carbon dioxide molecule. The ionic liquid, characterized by its high polarity and low vapor pressure, acts as a powerful activator for the thermodynamically stable CO2. The basic anion or the hydroxyl group within the ionic liquid structure facilitates the nucleophilic attack on the carbon atom of CO2, forming a reactive carbamate or carbonate intermediate. This activated species then undergoes cyclization with the amino and cyano groups of the 2-aminobenzonitrile substrate. The tunability of the ionic liquid allows for precise optimization of the electronic environment, ensuring that the activation energy barrier is sufficiently lowered to permit the reaction to proceed under mild conditions. This mechanistic pathway is not only efficient but also highly selective, minimizing the occurrence of side reactions that typically plague high-temperature syntheses.

Furthermore, the choice of the cation in the ionic liquid plays a critical role in stabilizing the transition states and intermediates throughout the catalytic cycle. Structures such as [HDBU][2-OP] or [Ch][2-OP] provide a specific steric and electronic environment that promotes the intramolecular cyclization step. The presence of the 2-hydroxypyridine moiety is crucial, as it likely participates in hydrogen bonding networks that orient the reactants favorably for bond formation. This level of mechanistic control translates directly into higher purity profiles for the final API intermediate, as fewer impurities are generated at the source. For quality control teams, this means a more robust specification sheet and reduced risk of batch failure due to trace contaminants. The ability to fine-tune the catalyst structure offers a pathway to further optimize the process for specific substrate derivatives, enhancing the versatility of the platform.
How to Synthesize 2,4-(1H,3H)-quinazoline Diketone Efficiently
The implementation of this synthesis route in a laboratory or pilot plant setting follows a logical sequence designed to maximize yield and safety. The process begins with the preparation of the ionic liquid catalyst, which can be synthesized simply by stirring 2-hydroxypyridine with an organic base in ethanol, a procedure that is scalable and straightforward. Once the catalyst is ready, the 2-aminobenzonitrile substrate is introduced into the reaction vessel under a carbon dioxide atmosphere. The reaction is then heated to the optimal temperature, which varies depending on the specific ionic liquid and substrate used, typically falling within the 60°C to 100°C range for best results. Detailed standardized synthesis steps see the guide below.
- Prepare the 2-hydroxypyridine ionic liquid catalyst by stirring 2-hydroxypyridine with an organic base in ethanol.
- React 2-aminobenzonitrile with carbon dioxide in the presence of the ionic liquid at 30-150°C.
- Isolate the product by adding water to the reaction mixture, filtering the precipitate, and washing with organic solvents.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this CO2 fixation technology offers substantial strategic advantages for procurement and supply chain management. The primary benefit is the drastic reduction in raw material costs associated with replacing expensive and hazardous reagents like phosgene with carbon dioxide. This shift not only lowers the direct cost of goods sold but also reduces the indirect costs related to safety compliance, waste disposal, and insurance. Additionally, the mild reaction conditions imply lower energy consumption, contributing to a smaller carbon footprint and aligning with corporate sustainability goals. For supply chain heads, the use of readily available starting materials ensures a more stable and resilient supply chain, less susceptible to market fluctuations of specialty chemicals. The simplicity of the purification process also means faster turnaround times from reaction to finished product, enhancing overall operational efficiency.
- Cost Reduction in Manufacturing: The elimination of phosgene and the use of a reusable ionic liquid catalyst system fundamentally alter the cost structure of quinazoline production. By removing the need for specialized phosgene handling equipment and the associated safety infrastructure, capital expenditure is significantly reduced. Furthermore, the high yields achieved under mild conditions mean less raw material is wasted, improving the overall material efficiency of the process. The ionic liquid can potentially be recovered and recycled, further driving down the recurring cost of catalysts. These factors combine to create a manufacturing process that is inherently more economical, allowing for competitive pricing in the global market without sacrificing quality margins.
- Enhanced Supply Chain Reliability: Relying on carbon dioxide as a key reagent provides a significant buffer against supply chain disruptions. Unlike complex organic reagents that may have limited suppliers or long lead times, CO2 is a commodity chemical available globally in vast quantities. This abundance ensures that production schedules can be maintained consistently, even during periods of market volatility. The robustness of the ionic liquid catalyst also contributes to reliability, as it is less sensitive to moisture and air compared to many traditional organometallic catalysts. This stability simplifies storage and handling logistics, reducing the risk of material degradation before use and ensuring a steady flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: The transition from batch to continuous flow or large-scale batch production is facilitated by the mild and safe nature of this reaction. The absence of highly toxic gases simplifies the regulatory approval process for new manufacturing sites, accelerating time-to-market for new products. Environmental compliance is significantly easier to achieve, as the process generates minimal hazardous waste and utilizes a greenhouse gas as a feedstock. This aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against potential regulatory crackdowns on traditional chemical processes. The scalability is further supported by the simple work-up procedure, which can be easily adapted to large-scale filtration and drying equipment without complex modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed CO2 fixation technology. These answers are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines. The information covers aspects ranging from catalyst recovery to substrate compatibility, ensuring a comprehensive overview of the technology's capabilities and limitations.
Q: What are the advantages of using CO2 over phosgene for quinazoline synthesis?
A: Using CO2 eliminates the need for highly toxic phosgene gas, significantly improving process safety and reducing environmental hazards associated with traditional synthesis routes.
Q: Can this ionic liquid catalyst be reused in industrial production?
A: Yes, the ionic liquid system demonstrates high stability and can potentially be recovered and reused, contributing to lower operational costs and waste generation.
Q: What is the substrate scope for this CO2 fixation reaction?
A: The method accommodates various substituents on the benzene ring, including alkyl, halogen, alkoxy, and trifluoromethyl groups, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline-2,4-dione Supplier
The technological potential of CO2 fixation for heterocycle synthesis is immense, and NINGBO INNO PHARMCHEM stands ready to leverage this innovation for your commercial needs. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of quinazoline-2,4-dione meets the highest international standards. We understand the critical nature of API intermediates in your supply chain and are committed to delivering consistent quality and reliability.
We invite you to discuss how this green synthesis route can optimize your manufacturing costs and sustainability profile. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us collaborate to build a more efficient and sustainable supply chain for your pharmaceutical and agrochemical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
