Optimizing Disodium 5'-Ribonucleotide Production: A Technical Analysis of Steric Control Phosphorylation
Optimizing Disodium 5'-Ribonucleotide Production: A Technical Analysis of Steric Control Phosphorylation
The global demand for high-purity nucleotides, particularly disodium 5'-ribonucleotide, has surged due to their critical applications in the pharmaceutical, food, and agricultural sectors. As a key flavor enhancer and metabolic regulator, the quality of this compound directly impacts the efficacy of therapeutic formulations and the sensory profile of food products. Patent CN101891772A introduces a transformative chemical synthesis route that addresses long-standing challenges in regioselectivity and yield. Unlike traditional enzymatic or direct chemical phosphorylation methods which often suffer from complex impurity profiles, this invention leverages a steric hindrance strategy to achieve superior control over the reaction pathway. The core innovation lies in the use of a pre-formed phosphoryl dihalide intermediate, which acts as a highly selective phosphorylating agent. This approach not only simplifies the purification process but also significantly enhances the overall economic viability of the manufacturing process.
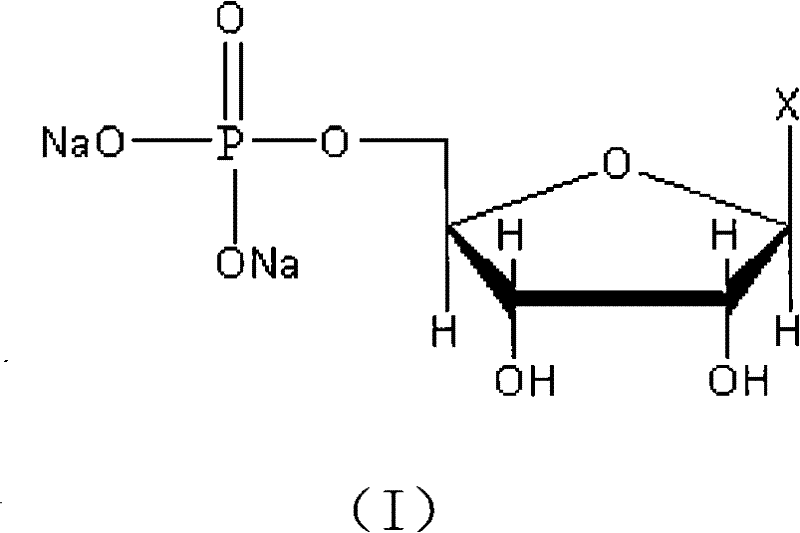
For procurement managers and supply chain directors seeking a reliable disodium 5'-ribonucleotide supplier, understanding the underlying chemistry is crucial for assessing long-term supply stability. The structural integrity of the final product, as depicted in the molecular diagram, relies heavily on the precise placement of the phosphate group at the 5' position of the ribose ring. Any deviation leading to 2' or 3' isomers constitutes a difficult-to-remove impurity that can compromise regulatory compliance. The method described in CN101891772A ensures that the final structure matches the stringent requirements for high-purity food additive intermediates and pharmaceutical ingredients. By shifting the paradigm from direct phosphorylation to a stepped, protected intermediate approach, the technology offers a robust solution for cost reduction in nucleotide manufacturing while maintaining exceptional quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of 5'-nucleotides has been plagued by the inherent lack of selectivity of phosphorus oxychloride (POCl3). When nucleosides such as inosine or guanosine are reacted directly with POCl3, the phosphorylation occurs indiscriminately at the 2', 3', and 5' hydroxyl groups. This results in a complex mixture containing the desired 5'-monophosphate alongside significant quantities of 2'- and 3'-phosphoric acid esters, bisphosphates, and free purine bases. The separation of these closely related isomers is notoriously difficult and expensive, often requiring multiple chromatographic steps that drastically reduce the overall yield. Furthermore, conventional chemical methods typically achieve yields in the range of 85% to 90%, leaving a substantial portion of valuable starting material wasted as byproducts. This inefficiency not only drives up raw material costs but also creates significant waste disposal challenges, complicating environmental compliance for large-scale facilities.
The Novel Approach
The methodology presented in the patent data circumvents these issues by introducing a steric control mechanism prior to the phosphorylation event. Instead of using naked POCl3, the process first converts it into a phosphoryl dihalide species bearing a bulky alkyl or substituted alkyl group (R-group). This modified reagent possesses significant steric bulk that physically hinders access to the more crowded 2' and 3' hydroxyl positions on the ribose sugar. Consequently, the reaction is funneled almost exclusively toward the more accessible 5'-hydroxyl group. This strategic modification transforms a messy, non-selective reaction into a highly precise transformation. The result is a crude reaction mixture that is far cleaner than that produced by conventional methods, thereby reducing the burden on downstream purification units. This shift represents a fundamental improvement in process chemistry, enabling the production of high-purity intermediates with minimal effort.
Mechanistic Insights into Steric Hindrance Phosphorylation
The success of this synthesis hinges on the careful design of the phosphorylating agent. The mechanism begins with the reaction of phosphorus oxychloride with a specific alcohol (R-OH) at low temperatures, typically below 0°C, to generate the key intermediate, phosphoryl dihalide (II). The choice of the R-group is critical; groups such as tert-butyl or trityl provide the necessary spatial volume to enforce selectivity. Once formed, this intermediate reacts with the nucleoside in the presence of an acid scavenger like triethylamine. The steric bulk of the R-group ensures that the nucleophilic attack by the ribose hydroxyls occurs primarily at the 5' position. Following this selective phosphorylation, the R-group is removed via hydrolysis—either acidic or basic depending on the specific nature of the R-group—to reveal the free phosphate, which is then converted to the disodium salt.
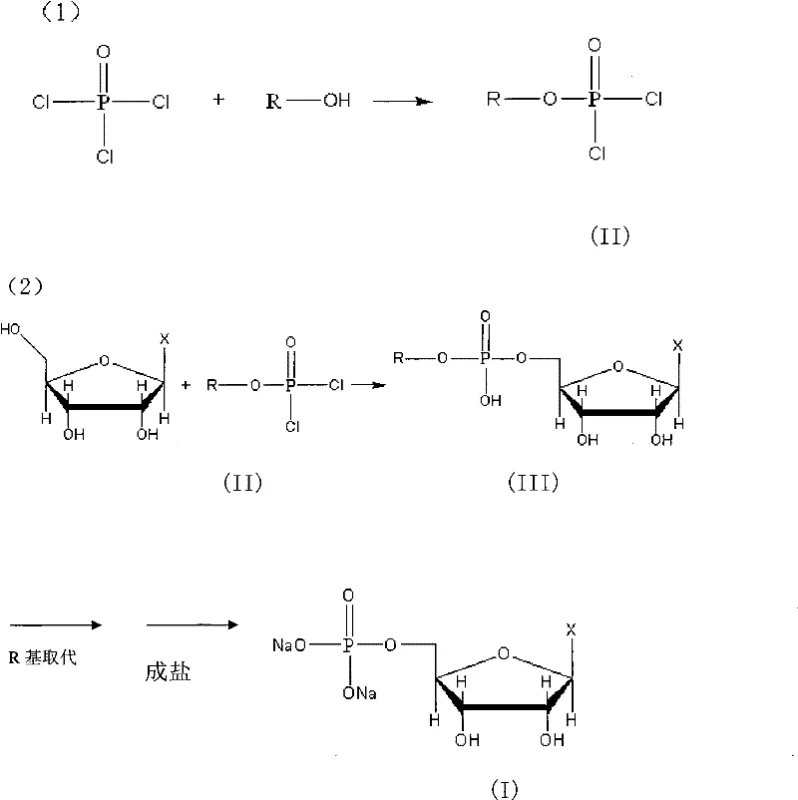
From an impurity control perspective, this mechanism is superior because it prevents the formation of isomeric byproducts at the source rather than trying to remove them later. In traditional synthesis, the 2' and 3' isomers are structurally very similar to the 5' product, making separation energy-intensive. By preventing their formation through steric exclusion, the process inherently guarantees a higher purity profile. The patent data highlights that this method can achieve phosphorylation yields of 95% to 98%, a marked improvement that speaks to the efficiency of the mechanistic design. For R&D directors, this implies a more predictable reaction profile and a reduced risk of batch failure due to out-of-specification impurity levels. The ability to tune the R-group allows for further optimization of reaction kinetics and solubility, offering a versatile platform for synthesizing various 5'-ribonucleotides beyond just guanylic acid.
How to Synthesize Disodium 5'-Ribonucleotide Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable pathway for producing high-quality nucleotides. The process is divided into three distinct operational stages: the preparation of the phosphorylating agent, the coupling reaction with the nucleoside, and the final deprotection and salification. Each step is optimized for temperature control and stoichiometry to maximize conversion while minimizing side reactions. The use of common inert solvents such as methylene dichloride ensures that the process can be easily integrated into existing chemical infrastructure without requiring specialized equipment. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-efficiency route.
- Preparation of Phosphoryl Dihalide: React phosphorus oxychloride with a bulky alcohol (R-OH) at sub-zero temperatures to form the sterically hindered phosphorylating agent.
- Selective Phosphorylation: React the nucleoside with the phosphoryl dihalide intermediate in the presence of an acid scavenger to selectively target the 5'-hydroxyl group.
- Deprotection and Salification: Remove the protecting R-group via hydrolysis (acidic or basic depending on R) and neutralize with sodium base to obtain the final disodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this advanced synthesis route offers tangible benefits that extend beyond simple chemistry. The primary advantage lies in the drastic simplification of the purification workflow. Because the reaction generates significantly fewer byproducts, the need for extensive chromatographic purification is reduced or potentially eliminated in favor of simpler crystallization or extraction techniques. This reduction in processing complexity translates directly into lower operational expenditures and shorter production cycles. Furthermore, the high conversion rates mean that less raw material is required to produce the same amount of finished goods, providing a buffer against volatility in nucleoside pricing. This efficiency makes the supply chain more resilient and cost-effective.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the increase in reaction yield fundamentally alter the cost structure of production. By avoiding the loss of material to 2' and 3' isomers, the effective cost per kilogram of the active ingredient is significantly lowered. Additionally, the reduction in solvent usage and waste generation associated with extensive purification lowers environmental compliance costs. The process utilizes readily available reagents and standard solvents, avoiding the need for exotic or prohibitively expensive catalysts often found in enzymatic routes. This economic efficiency allows for competitive pricing strategies in the global market for food additives and pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Chemical synthesis routes are generally less susceptible to the biological variabilities that plague fermentation or enzymatic processes. There is no risk of strain degeneration or enzyme deactivation, ensuring consistent batch-to-batch quality and reliable delivery schedules. The robustness of the chemical conditions means that production can be scaled up or down rapidly in response to market demand without the long lead times associated with biological system optimization. This reliability is critical for customers who require just-in-time delivery for their own manufacturing lines, reducing the need for excessive safety stock.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing standard unit operations such as distillation, extraction, and filtration. The reduction in byproduct formation inherently reduces the chemical oxygen demand (COD) of the waste stream, simplifying wastewater treatment requirements. The ability to operate at moderate temperatures and pressures further enhances safety and reduces energy consumption. These factors combined make the technology highly attractive for large-scale commercial production, ensuring that supply can meet the growing global demand for nucleotides in a sustainable manner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of disodium 5'-ribonucleotide using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility and advantages of integrating this supply source into their procurement strategies.
Q: How does this method improve purity compared to traditional POCl3 phosphorylation?
A: Traditional methods using direct POCl3 often result in significant formation of 2'- and 3'-phosphate isomers due to lack of selectivity. This novel method utilizes a bulky R-group on the phosphorylating agent to create steric hindrance, effectively blocking reaction at the 2' and 3' positions and ensuring high regioselectivity for the 5'-position.
Q: What are the typical yields for this synthesis route?
A: The patent data indicates that this specific pathway achieves phosphorylation yields ranging from 95% to 98%, which is a significant improvement over conventional chemical synthesis methods that typically hover around 85% to 90%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes standard inert solvents like methylene dichloride and avoids complex enzymatic membrane systems, making it robust for commercial scale-up with simplified post-processing and purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disodium 5'-Ribonucleotide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity nucleotides play in the formulation of advanced pharmaceuticals and premium food products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the highest international standards. Our commitment to process excellence allows us to deliver materials that support your R&D and manufacturing goals without compromise.
We invite you to discuss how our advanced manufacturing capabilities can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our efficient synthesis methods can enhance your product quality and bottom line.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
